Abstract
Although kinases are important regulators of many cellular processes, measuring their activity in live cells remains challenging. We have developed kinase translocation reporters (KTRs), which enable multiplexed measurements of the dynamics of kinase activity at a single-cell level. These KTRs are composed of an engineered construct in which a kinase substrate is fused to a bipartite nuclear localization signal (bNLS) and nuclear export signal (NES), as well as to a fluorescent protein for microscopy-based detection of its localization. The negative charge introduced by phosphorylation of the substrate is used to directly modulate nuclear import and export, thereby regulating the reporter's distribution between the cytoplasm and nucleus. The relative cytoplasmic versus nuclear fluorescence of the KTR construct (the C/N ratio) is used as a proxy for the kinase activity in living, single cells. Multiple KTRs can be studied in the same cell by fusing them to different fluorescent proteins. Here, we present a protocol to execute and analyze live-cell microscopy experiments using KTRs. We describe strategies for development of new KTRs and procedures for lentiviral expression of KTRs in a cell line of choice. Cells are then plated in a 96-well plate, from which multichannel fluorescent images are acquired with automated time-lapse microscopy. We provide detailed guidance for a computational analysis and parameterization pipeline. The entire procedure, from virus production to data analysis, can be completed in ∼10 d.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Manning, G., Whyte, D.B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Cohen, P. & Philip, C. The role of protein phosphorylation in human health and disease. Eur. J. Biochem. 268, 5001–5010 (2001).
Purvis, J.E. & Lahav, G. Encoding and decoding cellular information through signaling dynamics. Cell 152, 945–956 (2013).
Selimkhanov, J. et al. Accurate information transmission through dynamic biochemical signaling networks. Science 346, 1370–1373 (2014).
Shalek, A.K. et al. Single-cell RNA-seq reveals dynamic paracrine control of cellular variation. Nature 510, 363–369 (2014).
Zernicka-Goetz, M., Morris, S.A. & Bruce, A.W. Making a firm decision: multifaceted regulation of cell fate in the early mouse embryo. Nat. Rev. Genet. 10, 467–477 (2009).
Vlamakis, H., Aguilar, C., Losick, R. & Kolter, R. Control of cell fate by the formation of an architecturally complex bacterial community. Genes Dev. 22, 945–953 (2008).
Spencer, S.L. et al. The proliferation-quiescence decision is controlled by a bifurcation in CDK2 activity at mitotic exit. Cell 155, 369–383 (2013).
Albeck, J.G., Mills, G.B. & Brugge, J.S. Frequency-modulated pulses of ERK activity transmit quantitative proliferation signals. Mol. Cell 49, 249–261 (2013).
Regot, S., Hughey, J.J., Bajar, B.T., Carrasco, S. & Covert, M.W. High-sensitivity measurements of multiple kinase activities in live single cells. Cell 157, 1724–1734 (2014).
Kondoh, K., Terasawa, K., Morimoto, H. & Nishida, E. Regulation of nuclear translocation of extracellular signal-regulated kinase 5 by active nuclear import and export mechanisms. Mol. Cell. Biol. 26, 1679–1690 (2006).
Nardozzi, J.D., Lott, K. & Cingolani, G. Phosphorylation meets nuclear import: a review. Cell Commun. Signal. 8, 32 (2010).
Noursadeghi, M. et al. Quantitative imaging assay for NF-κB nuclear translocation in primary human macrophages. J. Immunol. Methods 329, 194–200 (2008).
Maryu, G., Matsuda, M. & Aoki, K. Multiplexed fluorescence imaging of ERK and Akt activities and cell-cycle progression. Cell Struct. Funct. 41, 81–92 (2016).
Oldach, L. & Zhang, J. Genetically encoded fluorescent biosensors for live-cell visualization of protein phosphorylation. Chem. Biol. 21, 186–197 (2014).
Lam, A.J. et al. Improving FRET dynamic range with bright green and red fluorescent proteins. Nat. Methods 9, 1005–1012 (2012).
Kosugi, S., Hasebe, M., Tomita, M. & Yanagawa, H. Nuclear export signal consensus sequences defined using a localization-based yeast selection system. Traffic 9, 2053–2062 (2008).
Kosugi, S., Hasebe, M., Tomita, M. & Yanagawa, H. Systematic identification of cell cycle-dependent yeast nucleocytoplasmic shuttling proteins by prediction of composite motifs. Proc. Natl. Acad. Sci. USA 106, 10171–10176 (2009).
Sieracki, N.A. & Komarova, Y.A. Studying cell signal transduction with biomimetic point mutations. In Manipulation of DNA and Protein –Examples from Current Research, 381–392. (In Tech, 2013).
Miyamoto, T. et al. Compartmentalized AMPK signaling illuminated by genetically encoded molecular sensors and actuators. Cell Rep. 11, 657–670 (2015).
Miura, H., Haruko, M., Michiyuki, M. & Kazuhiro, A. Development of a FRET biosensor with high specificity for Akt. Cell Struct. Funct. 39, 9–20 (2014).
Seong, J. et al. Detection of focal adhesion kinase activation at membrane microdomains by fluorescence resonance energy transfer. Nat. Commun. 2, 406 (2011).
Ubersax, J.A. & Ferrell, J.E. Jr. Mechanisms of specificity in protein phosphorylation. Nat. Rev. Mol. Cell Biol. 8, 530–541 (2007).
Pearce, L.R., Komander, D. & Alessi, D.R. The nuts and bolts of AGC protein kinases. Nat. Rev. Mol. Cell Biol. 11, 9–22 (2010).
Moritz, A. et al. Akt-RSK-S6 kinase signaling networks activated by oncogenic receptor tyrosine kinases. Sci. Signal. 3, ra64 (2010).
Hornbeck, P.V. et al. PhosphoSitePlus, 2014: mutations, PTMs and recalibrations. Nucleic Acids Res. 43, D512–D520 (2015).
McNulty, D.E., Sikorski, T.W. & Annan, R.S. Identification and analysis of protein phosphorylation by mass spectrometry. in Analysis of Protein Post-Translational Modifications by Mass Spectrometry 17–87 (2016).
Peck, S.C. Analysis of protein phosphorylation: methods and strategies for studying kinases and substrates. Plant J. 45, 512–522 (2006).
Van Valen, D.A. et al. Deep learning automates the quantitative analysis of individual cells in live-cell imaging experiments. PLoS Comput. Biol. 12, e1005177 (2016).
Schindelin, J. et al. Fiji: an open-source platform for biological-image analysis. Nat. Methods 9, 676–682 (2012).
Carpenter, A.E. et al. CellProfiler: image analysis software for identifying and quantifying cell phenotypes. Genome Biol. 7, R100 (2006).
Edelstein, A.D. et al. Advanced methods of microscope control using μManager software. J. Biol. Methods 1, e10 (2014).
Magidson, V. & Khodjakov, A. Circumventing photodamage in live-cell microscopy. Methods Cell Biol. 114, 545–560 (2013).
Basu, S., Campbell, H.M., Dittel, B.N. & Ray, A. Purification of specific cell population by fluorescence activated cell sorting (FACS). J. Vis. Exp. http://dx.doi.org/10.3791/1546 (2010).
Masip, M.E. et al. Reversible cryo-arrest for imaging molecules in living cells at high spatial resolution. Nat. Methods 13, 665–672 (2016).
Tay, S. et al. Single-cell NF-kappaB dynamics reveal digital activation and analogue information processing. Nature 466, 267–271 (2010).
Williams, R.G. et al. Generation and characterization of a stable MK2-EGFP cell line and subsequent development of a high-content imaging assay on the Cellomics ArrayScan platform to screen for p38 mitogen-activated protein kinase inhibitors. Methods Enzymol. 364–389 (2006).
Anton, R. et al. A p38 substrate-specific MK2-EGFP translocation assay for identification and validation of new p38 inhibitors in living cells: a comprising alternative for acquisition of cellular p38 inhibition data. PLoS ONE 9, e95641 (2014).
Reinhardt, H.C. et al. DNA damage activates a spatially distinct late cytoplasmic cell-cycle checkpoint network controlled by MK2-mediated RNA stabilization. Mol. Cell 40, 34–49 (2010).
Samsonov, A. et al. Tagging of genomic STAT3 and STAT1 with fluorescent proteins and insertion of a luciferase reporter in the cyclin D1 gene provides a modified A549 cell line to screen for selective STAT3 inhibitors. PLoS ONE 8, e68391 (2013).
Yissachar, N. et al. Dynamic response diversity of NFAT isoforms in individual living cells. Mol. Cell 49, 322–330 (2013).
Gross, S.M. & Rotwein, P. Akt signaling dynamics in individual cells. J. Cell Sci. 128, 2509–2519 (2015).
Katsura, Y. et al. An optogenetic system for interrogating the temporal dynamics of Akt. Sci. Rep. 5, 14589 (2015).
Gross, S.M. & Peter, R. Mapping growth-factor-modulated Akt signaling dynamics. Development 143, e1.2 (2016).
Schmierer, B. & Hill, C.S. Kinetic analysis of Smad nucleocytoplasmic shuttling reveals a mechanism for transforming growth factor-dependent nuclear accumulation of Smads. Mol. Cell. Biol. 25, 9845–9858 (2005).
Wang, C.-C. et al. A time- and matrix-dependent TGFBR3–JUND–KRT5 regulatory circuit in single breast epithelial cells and basal-like premalignancies. Nat. Cell Biol. 16, 345–356 (2014).
Hahn, A.T., Jones, J.T. & Meyer, T. Quantitative analysis of cell cycle phase durations and PC12 differentiation using fluorescent biosensors. Cell Cycle 8, 1044–1052 (2009).
Zhao, B. et al. Inactivation of YAP oncoprotein by the Hippo pathway is involved in cell contact inhibition and tissue growth control. Genes Dev. 21, 2747–2761 (2007).
Cohen-Saidon, C., Cohen, A.A., Sigal, A., Liron, Y. & Alon, U. Dynamics and variability of ERK2 response to EGF in individual living cells. Mol. Cell 36, 885–893 (2009).
Acknowledgements
We thank the members of Covert lab for advice and critical commentary on the manuscript, in particular K. Lane for supplying the images of primary macrophages in Supplementary Figure 1. We gratefully acknowledge funding from several sources, including a Distinguished Investigator award and a Discovery Center grant from the Paul Allen Family Foundation, as well as an NIH Pioneer Award (5DP1LM01150-05) to M.W.C., a Human Frontier Science Program (HFSP) postdoctoral fellowship (LT000529/ 2012-L) to S.R., the Nakajima Foundation Scholarship to T.K., a DOE CSGF grant (DE-FG02-97ER25308) to D.N.M., and a Systems Biology Center grant (P50 GM107615).
Author information
Authors and Affiliations
Contributions
S.R. developed the reporter strategy and designed the experiments. T.K. and D.N.M. developed the computational pipelines and prepared the protocols. J.J.H. and S.A. developed the general workflow for the modeling. T.K., S.J., and M.W.C. wrote the manuscript, with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
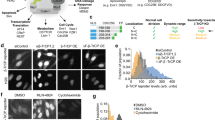
Supplementary Figure 1 KTR translocation in different cell lines.
Time relative to stimulation is shown on each images.
(A) HeLa cells expressing JNK KTR were stimulated with anisomycin 50 ng/ml.
(B) HEK293 cells expressing JNK KTR were stimulated with anisomycin 50 ng/ml.
(C) Raw 264.7 cells expressing JNK KTR were stimulated with LPS 100 ng/ml.
(D) PC-12 cells expressing ERK KTR were stimulated with NGF 25 ng/ml.
(E) Primary Bone Marrow-derived Macrophages (BMDMs) isolated from mice expressing JNK KTR were stimulated with LPS 100 ng/ml.
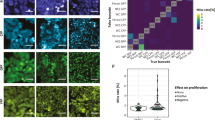
Supplementary Figure 2 KTR translocation and the estimated active JNK concentration.
(A) Heatmap illustrating the experimental C/N ratio, simulated C/N ratio and predicted active JNK concentration upon IL-1β stimulation. Each row denotes a color coded individual time course of an indicated property for a single cell. This is the final output from the jupyter notebook demonstration. (B) The population average traces of the C/N ratio (red), active JNK concentration (blue) and total phosphorylated KTR concentration (green). Note that the C/N ratio is acquired by direct analysis of the KTR image data, while the JNK and KTR concentrations are calculated by further analysis using a computational mechanistic model of KTR phosphorylation and dephosphorylation as well as nuclear or cytoplasmic translocation. The concentrations are normalized by minimum and maximum values.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1 and 2, Supplementary Methods, Supplementary Tables 1–3, Supplementary Notes 1 and 2, and Supplementary Data. (PDF 8160 kb)
Rights and permissions
About this article
Cite this article
Kudo, T., Jeknić, S., Macklin, D. et al. Live-cell measurements of kinase activity in single cells using translocation reporters. Nat Protoc 13, 155–169 (2018). https://doi.org/10.1038/nprot.2017.128
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2017.128
This article is cited by
-
DNA-functionalized artificial mechanoreceptor for de novo force-responsive signaling
Nature Chemical Biology (2024)
-
Characterizing heterogeneous single-cell dose responses computationally and experimentally using threshold inhibition surfaces and dose-titration assays
npj Systems Biology and Applications (2024)
-
TrackMate 7: integrating state-of-the-art segmentation algorithms into tracking pipelines
Nature Methods (2022)
-
Rapid signaling reactivation after targeted BRAF inhibition predicts the proliferation of individual melanoma cells from an isogenic population
Scientific Reports (2021)
-
Viral fibrotic scoring and drug screen based on MAPK activity uncovers EGFR as a key regulator of COVID-19 fibrosis
Scientific Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.