Abstract
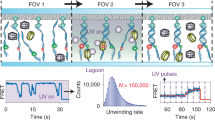
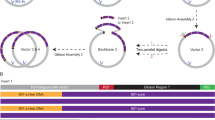
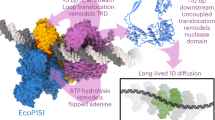
This protocol describes how to monitor individual naturally supercoiled circular DNA plasmids bound via peptide nucleic acid (PNA) handles between a bead and a surface. The protocol was developed for single-molecule investigation of the dynamics of supercoiled DNA, and it allows the investigation of both the dynamics of the molecule itself and of its interactions with a regulatory protein. Two bis-PNA clamps designed to bind with extremely high affinity to predetermined homopurine sequence sites in supercoiled DNA are prepared: one conjugated with digoxigenin for attachment to an anti-digoxigenin-coated glass cover slide, and one conjugated with biotin for attachment to a submicron-sized streptavidin-coated polystyrene bead. Plasmids are constructed, purified and incubated with the PNA handles. The dynamics of the construct is analyzed by tracking the tethered bead using video microscopy: less supercoiling results in more movement, and more supercoiling results in less movement. In contrast to other single-molecule methodologies, the current methodology allows for studying DNA in its naturally supercoiled state with constant linking number and constant writhe. The protocol has potential for use in studying the influence of supercoils on the dynamics of DNA and its associated proteins, e.g., topoisomerase. The procedure takes ∼4 weeks.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout









Similar content being viewed by others
Change history
12 September 2014
In the version of this article initially published, the source from which Figures 2, 4, 8 and 9 was adapted was not cited and credited correctly. The error has been corrected in the HTML and PDF versions of the article.
References
Bustamante, C., Bryant, Z. & Smith, S.B. Ten years of tension: single-molecule DNA mechanics. Nature 421, 423–427 (2003).
Lee, W.M., Reece, P.J., Marchington, R.F., Metzger, N.K. & Dholakia, K. Construction and calibration of an optical trap on a fluorescence optical microscope. Nat. Protoc. 2, 3226–3238 (2007).
Strick, T., Allemand, J.F., Croquette, V. & Bensimon, D. Twisting and stretching single DNA molecules. Prog. Biophys. Mol. Biol. 74, 115–140 (2000).
Wang, M.D., Yin, H., Landick, R., Gelles, J. & Block, S.M. Stretching DNA with optical tweezers. Biophys. J. 72, 1335–1346 (1997).
Gross, P. et al. Quantifying how DNA stretches, melts and changes twist under tension. Nat. Phys. 7, 731–736 (2011).
Stratmann, S.A. & van Oijen, A.M. DNA replication at the single-molecule level. Chem. Soc. Rev. 43, 1201–1220 (2014).
Dulin, D., Lipfert, J., Moolman, M.C. & Dekker, N.H. Studying genomic processes at the single-molecule level: introducing the tools and applications. Nat. Rev. Genet. 14, 9–22 (2013).
Herbert, K.M., Greenleaf, W.J. & Block, S.M. Single-molecule studies of RNA polymerase: motoring along. Annu. Rev. Biochem. 77, 149–176 (2008).
Chaurasiya, K.R., Geertsema, H., Cristofari, G., Darlix, J.L. & Williams, M.C. A single zinc finger optimizes the DNA interactions of the nucleocapsid protein of the yeast retrotransposon Ty3. Nucleic Acids Res. 40, 751–760 (2012).
Paramanathan, T., Vladescu, I., McCauley, M.J., Rouzina, I. & Williams, M.C. Force spectroscopy reveals the DNA structural dynamics that govern the slow binding of actinomycin D. Nucleic Acids Res. 40, 4925–4932 (2012).
Forget, A.L., Dombrowski, C.C., Amitani, I. & Kowalczykowski, S.C. Exploring protein-DNA interactions in 3D using in situ construction, manipulation and visualization of individual DNA dumbbells with optical traps, microfluidics and fluorescence microscopy. Nat. Protoc. 8, 525–538 (2013).
Zurla, C. et al. Direct demonstration and quantification of long-range DNA looping by the λ bacteriophage repressor. Nucleic Acids Res. 37, 2789–2795 (2009).
Manzo, C., Zurla, C., Dunlap, D.D. & Finzi, L. The effect of nonspecific binding of λ repressor on DNA looping dynamics. Biophys. J. 103, 1753–1761 (2012).
Priest, D.G. et al. Quantitation of the DNA tethering effect in long-range DNA looping in vivo and in vitro using the Lac and λ repressors. Proc. Natl. Acad. Sci. USA 111, 349–354 (2014).
Normanno, D., Vanzi, F. & Pavone, F.S. Single-molecule manipulation reveals supercoiling-dependent modulation of lac repressor-mediated DNA looping. Nucleic Acids Res. 36, 2505–2513 (2008).
Norregaard, K. et al. DNA supercoiling enhances cooperativity and efficiency of an epigenetic switch. Proc. Natl. Acad. Sci. USA 110, 17386–17391 (2013).
Jian, H.M., Schlick, T. & Vologodskii, A. Internal motion of supercoiled DNA: Brownian dynamics simulations of site juxtaposition. J. Mol. Biol. 284, 287–296 (1998).
Bauer, M. & Metzler, R. In vivo facilitated diffusion model. PLoS ONE 8, e53956 (2013).
Bentin, T. & Nielsen, P.E. In vitro transcription of a torsionally constrained template. Nucleic Acids Res. 30, 803–809 (2002).
Bentin, T. & Nielsen, P.E. Enhanced peptide nucleic acid binding to supercoiled DNA: possible implications for DNA 'breathing' dynamics. Biochemistry 35, 8863–8869 (1996).
Norregaard, K. et al. Effect of supercoiling on the λ switch. Bacteriophage 4, e27517 (2014).
Lia, G. et al. The antiparallel loops in gal DNA. Nucleic Acids Res. 36, 4204–4210 (2008).
Lia, G. et al. Supercoiling and denaturation in Gal repressor/heat unstable nucleoid protein (HU)-mediated DNA looping. Proc. Natl. Acad. Sci. USA 100, 11373–11377 (2003).
Strick, T.R., Croquette, V. & Bensimon, D. Single-molecule analysis of DNA uncoiling by a type II topoisomerase. Nature 404, 901–904 (2000).
Lindner, M., Nir, G., Vivante, A., Young, I.T. & Garini, Y. Dynamic analysis of a diffusing particle in a trapping potential. Phy. Rev. E Stat. Nonlin. Soft Matter Phys. 87, 022716 (2013).
Mearini, G., Nielsen, P.E. & Fackelmayer, F.O. Localization and dynamics of small circular DNA in live mammalian nuclei. Nucleic Acids Res. 32, 2642–2651 (2004).
Norregaard, K., Jauffred, L., Berg-Sørensen, K. & Oddershede, L.B. Optical manipulation of single molecules in the living cell. Phys. Chem. Chem. Phys. 16, 12614–12624 (2014).
Nelson, P.C. et al. Tethered particle motion as a diagnostic of DNA tether length. J. Phys. Chem. B 110, 17260–17267 (2006).
Baek, K., Svenningsen, S., Eisen, H., Sneppen, K. & Brown, S. Single-cell analysis of λ immunity regulation. J. Mol. Biol. 334, 363–372 (2003).
Egholm, M. et al. Efficient pH-independent sequence-specific DNA binding by pseudoisocytosine-containing bis-PNA. Nucleic Acids Res. 23, 217–222 (1995).
Nielsen, P.E. Sequence-selective targeting of duplex DNA by peptide nucleic acids. Curr. Opin. Mol. Ther. 12, 184–191 (2010).
Griffith, M.C. et al. Single and bis peptide nucleic acids as triplexing agents: binding and stoichiometry. J. Am. Chem. Soc. 117, 831–832 (1995).
Yin, H., Landick, R. & Gelles, J. Tethered particle motion method for studying transcript elongation by a single RNA-polymerase molecule. Biophys. J. 67, 2468–2478 (1994).
Pardee, A.B., Jacob, F. & Monod, J. Genetic control and cytoplasmic expression of inducibility in the synthesis of β-galactosidase by E. coli. J. Mol. Biol. 1, 165–178 (1959).
Miller, J.H. Experiments in Molecular Genetics (Cold Spring Harbor Laboratory, 1972).
Norgard, M.V., Emigholz, K. & Monahan, J.J. Increased amplification of Pbr322 plasmid deoxyribonucleic-acid in Escherichia coli K-12 strain-Rr1 and Strain-Chi-1776 grown in the presence of high-concentrations of nucleoside. J. Bacteriol. 138, 270–272 (1979).
Cheezum, M.K., Walker, W.F. & Guilford, W.H. Quantitative comparison of algorithms for tracking single fluorescent particles. Biophys. J. 81, 2378–2388 (2001).
Birnboim, H.C. & Doly, J. A rapid alkaline extraction procedure for screening recombinant plasmid DNA. Nucleic Acids Res. 7, 1513–1523 (1979).
Radloff, R., Bauer, W. & Vinograd, J. A dye-buoyant-density method for the detection and isolation of closed circular duplex DNA: the closed circular DNA in HeLa cells. Proc. Natl. Acad. Sci. USA 57, 1514–1521 (1967).
Esposito, F. & Sinden, R.R. Supercoiling in prokaryotic and eukaryotic DNA: changes in response to topological perturbation of plasmids in E. coli and SV40 in vitro, in nuclei and in CV-1 cells. Nucleic Acids Res. 15, 5105–5124 (1987).
Hansen, G.I., Bentin, T., Larsen, H.J. & Nielsen, P.E. Structural isomers of bis-PNA bound to a target in duplex DNA. J. Mol. Biol. 307, 67–74 (2001).
Andersson, M., Czerwinski, F. & Oddershede, L.B. Optimizing active and passive calibration of optical tweezers. J. Opt. 13, 044020 (2011).
Czaerwinski, F., Richardson, A.C. & Oddershede, L.B. Quantifying noise in optical tweezers by Allan variance. Opt. Express 17, 13255–13269 (2009).
Tolic-Norrelykke, S.F., Rasmussen, M.B., Pavone, F.S., Berg-Sorensen, K. & Oddershede, L.B. Stepwise bending of DNA by a single TATA-box binding protein. Biophys. J. 90, 3694–3703 (2006).
Blumberg, S., Gajraj, A., Pennington, M.W. & Meiners, J.C. Three-dimensional characterization of tethered microspheres by total internal reflection fluorescence microscopy. Biophys. J. 89, 1272–1281 (2005).
Han, L. et al. Concentration and length dependence of DNA looping in transcriptional regulation. PLoS ONE 4, e5621 (2009).
Acknowledgements
The CI protein was a generous gift from D. Lewis and S. Adhya, Laboratory of Molecular Biology, Center for Cancer Research, National Cancer Institute, US National Institutes of Health. We acknowledge financial support from a University of Copenhagen center of excellence and from the Swedish Research Council.
Author information
Authors and Affiliations
Contributions
P.E.N., S.B. and L.B.O. designed the study; K.N., M.A. and S.B. performed the experiments; P.E.N. and S.B. contributed new reagents; K.N., M.A., S.B. and L.B.O. analyzed data; all authors wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Purification and identity of PNA product.
(a) HPLC analysis of purified digoxigenin labelled bis-PNA. (b) MALDI-TOF spectrum of digoxigenin labelled bis-PNA (calculated mass is 7187 and the found mass is within the accuracy of the instrument).
Supplementary information
Supplementary Figure 1
Purification and identity of PNA product. (PDF 499 kb)
Rights and permissions
About this article
Cite this article
Norregaard, K., Andersson, M., Nielsen, P. et al. Tethered particle analysis of supercoiled circular DNA using peptide nucleic acid handles. Nat Protoc 9, 2206–2223 (2014). https://doi.org/10.1038/nprot.2014.152
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.152
This article is cited by
-
DNA supercoiling during transcription
Biophysical Reviews (2016)
-
Erratum: Tethered particle analysis of supercoiled circular DNA using peptide nucleic acid handles
Nature Protocols (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.