Abstract
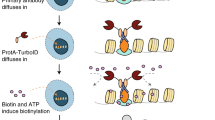
Solid-phase proximity ligation assays share properties with the classical sandwich immunoassays for protein detection. The proteins captured via antibodies on solid supports are, however, detected not by single antibodies with detectable functions, but by pairs of antibodies with attached DNA strands. Upon recognition by these sets of three antibodies, pairs of DNA strands brought in proximity are joined by ligation. The ligated reporter DNA strands are then detected via methods such as real-time PCR or next-generation sequencing (NGS). We describe how to construct assays that can offer improved detection specificity by virtue of recognition by three antibodies, as well as enhanced sensitivity owing to reduced background and amplified detection. Finally, we also illustrate how the assays can be applied for parallel detection of proteins, taking advantage of the oligonucleotide ligation step to avoid background problems that might arise with multiplexing. The protocol for the singleplex solid-phase proximity ligation assay takes ∼5 h. The multiplex version of the assay takes 7–8 h depending on whether quantitative PCR (qPCR) or sequencing is used as the readout. The time for the sequencing-based protocol includes the library preparation but not the actual sequencing, as times may vary based on the choice of sequencing platform.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
16 January 2015
In the version of this article initially published, the PCR table at Step 34C(iii) was incorrect and the sliding splint sequence in Supplementary Table 3 was also incorrect. The errors have been corrected in the HTML and PDF versions of the article and in the revised Supplementary Table 3.
References
Nong, R.Y., Gu, J., Darmanis, S., Kamali-Moghaddam, M. & Landegren, U. DNA-assisted protein detection technologies. Expert Rev. Proteomics 9, 21–32 (2012).
Fredriksson, S. et al. Protein detection using proximity-dependent DNA ligation assays. Nat. Biotechnol. 20, 473–477 (2002).
Lundberg, M., Eriksson, A., Tran, B., Assarsson, E. & Fredriksson, S. Homogeneous antibody-based proximity extension assays provide sensitive and specific detection of low-abundant proteins in human blood. Nucleic Acids Res. 39, e102 (2011).
Sano, T., Smith, C.L. & Cantor, C.R. Immuno-PCR: very sensitive antigen detection by means of specific antibody-DNA conjugates. Science 258, 120–122 (1992).
Schweitzer, B. et al. Immunoassays with rolling circle DNA amplification: a versatile platform for ultrasensitive antigen detection. Proc. Natl. Acad. Sci. USA 97, 10113–10119 (2000).
Soderberg, O. et al. Direct observation of individual endogenous protein complexes in situ by proximity ligation. Nat. Meth. 3, 995–1000 (2006).
Gullberg, M. et al. Cytokine detection by antibody-based proximity ligation. Proc. Natl. Acad. Sci. U.S.A. 101, 8420–8424 (2004).
Schallmeiner, E. et al. Sensitive protein detection via triple-binder proximity ligation assays. Nat. Meth. 4, 135–137 (2007).
Lundberg, M. et al. Multiplexed homogeneous proximity ligation assays for high-throughput protein biomarker research in serological material. Mol. Cell Proteomics 10, M110 004978 (2011).
McGregor, L.M., Gorin, D.J., Dumelin, C.E. & Liu, D.R. Interaction-dependent PCR: identification of ligand-target pairs from libraries of ligands and libraries of targets in a single solution-phase experiment. J. Am. Chem. Soc. 132, 15522–15524 (2010).
Darmanis, S. et al. Sensitive plasma protein analysis by microparticle-based proximity ligation assays. Mol. Cell Proteomics 9, 327–335 (2010).
Darmanis, S. et al. ProteinSeq: high-performance proteomic analyses by proximity ligation and next generation sequencing. PLoS ONE 6, e25583 (2011).
Wallin, U. et al. Growth differentiation factor 15: a prognostic marker for recurrence in colorectal cancer. Br. J. Cancer 104, 1619–1627 (2011).
Kamali-Moghaddam, M. et al. Sensitive detection of Aβ protofibrils by proximity ligation—relevance for Alzheimer′s disease. BMC Neurosci. 11, 124 (2010).
Tavoosidana, G. et al. Multiple recognition assay reveals prostasomes as promising plasma biomarkers for prostate cancer. Proc. Natl. Acad. Sci. U.S.A. 108, 8809–8814 (2011).
Goransson, J. et al. Rapid identification of bio-molecules applied for detection of biosecurity agents using rolling circle amplification. PLoS ONE 7, e31068 (2012).
Hammond, M., Nong, R.Y., Ericsson, O., Pardali, K. & Landegren, U. Profiling cellular protein complexes by proximity ligation with dual tag microarray readout. PLoS ONE 7, e40405 (2012).
Cheng, S. et al. Sensitive detection of small molecules by competitive immunomagnetic-proximity ligation assay. Anal. Chem. 84, 2129–2132 (2012).
Anderson, N.L. & Anderson, N.G. The human plasma proteome: history, character, and diagnostic prospects. Mol. Cell Proteomics 1, 845–867 (2002).
Anderson, N.L. et al. Mass spectrometric quantitation of peptides and proteins using Stable Isotope Standards and Capture by Anti-Peptide Antibodies (SISCAPA). J. Proteome Res. 3, 235–244 (2004).
Lange, V., Picotti, P., Domon, B. & Aebersold, R. Selected reaction monitoring for quantitative proteomics: a tutorial. Mol. Syst. Biol. 4, 222 (2008).
Nicol, G.R. et al. Use of an immunoaffinity-mass spectrometry-based approach for the quantification of protein biomarkers from serum samples of lung cancer patients. Mol. Cell Proteomics 7, 1974–1982 (2008).
Nam, J.M., Thaxton, C.S. & Mirkin, C.A. Nanoparticle-based bio-bar codes for the ultrasensitive detection of proteins. Science 301, 1884–1886 (2003).
Hyndman, D.L. & Mitsuhashi, M. PCR primer design. Methods Mol. Biol. 226, 81–88 (2003).
Acknowledgements
Our work has been funded by the Knut and Alice Wallenberg Foundation, the Swedish Research Council for Medicine (2007-2720, UL); the Swedish Foundation for Strategic Research; the Innovative Medicines Initiative Joint Undertaking under grant agreement no. 115234 (OncoTrack), resources of which are composed of financial contributions from the European Union's Seventh Framework Programme (FP7/2007-2013) and EFPIA companies' in-kind contribution; and by the European Community's 6th and 7th Framework Programs, including a European Research Council (ERC) senior investigator grant for high-throughput protein biomarker analysis (the European Research Council under the European Union's Seventh Framework Programme (FP7/2007-2013) / ERC grant agreement no. 294409), as well as for coordination of the EU project DiaTools focused on minimally invasive diagnostics (the European Community's 7th Framework Program (FP7/2007-2013) under grant agreement no. 259796).
Author information
Authors and Affiliations
Contributions
All authors contributed to writing the protocol and accepted the final version. Questions regarding the protocol should be addressed to U.L.
Corresponding author
Ethics declarations
Competing interests
U.L. is a founder of Olink Bioscience, which is commercializing reagents for PLA and PEA.
Supplementary information
Supplementary Table 1
List of validated antibodies. (PDF 568 kb)
Supplementary Table 2
List of different oligonucleotides used in multiplex spPLA. (PDF 497 kb)
Supplementary Table 3
List of the connector oligonucleotide and universal primers used in multiplex spPLA. (PDF 485 kb)
Supplementary Table 4
List of oligonucleotides used for sequencing library preparation in multiplex spPLA. (PDF 498 kb)
Supplementary Table 5
List of oligonucleotides used in singleplex spPLA. (PDF 489 kb)
Rights and permissions
About this article
Cite this article
Nong, R., Wu, D., Yan, J. et al. Solid-phase proximity ligation assays for individual or parallel protein analyses with readout via real-time PCR or sequencing. Nat Protoc 8, 1234–1248 (2013). https://doi.org/10.1038/nprot.2013.070
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.070
This article is cited by
-
Extending the dynamic range of biomarker quantification through molecular equalization
Nature Communications (2023)
-
Surface protein profiling of prostate-derived extracellular vesicles by mass spectrometry and proximity assays
Communications Biology (2022)
-
Proximity ligation assay: an ultrasensitive method for protein quantification and its applications in pathogen detection
Applied Microbiology and Biotechnology (2021)
-
Ultrasensitive digital quantification of cytokines and bacteria predicts septic shock outcomes
Nature Communications (2020)
-
Ratiometric electrochemical proximity assay for sensitive one-step protein detection
Scientific Reports (2014)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.