Abstract
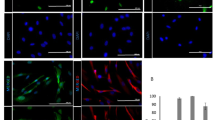
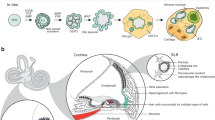
This protocol describes a growth medium–based approach for obtaining cochlear endothelial cells (ECs), pericytes (PCs) and perivascular resident macrophage-like melanocytes (PVM/Ms) from the stria vascularis of mice aged between P10 and P15 (P, postnatal day). The procedure does not involve mechanical or enzymatic digestion of the sample tissue. Explants of stria vascularis, 'mini-chips', are selectively cultured in growth medium, and primary cell lines are obtained in 7–10 d. The method is simple and reliable, and it provides high-quality ECs, PVM/Ms and PCs with a purity >90% after two passages. This protocol is suitable for producing primary culture cells from organs and tissues of small volume and high anatomical complexity, such as the inner ear capillaries. The highly purified primary cell lines enable cell culture–based in vitro modeling of cell-cell interactions, barrier control function and drug action.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Cohen-Salmon, M. et al. Connexin30 deficiency causes instrastrial fluid-blood barrier disruption within the cochlear stria vascularis. Proc. Natl. Acad. Sci. USA 104, 6229–6234 (2007).
Juhn, S.K., Hunter, B.A. & Odland, R.M. Blood-labyrinth barrier and fluid dynamics of the inner ear. Int. Tinnitus J. 7, 72–83 (2001).
Juhn, S.K. & Rybak, L.P. Labyrinthine barriers and cochlear homeostasis. Acta Otolaryngol. 91, 529–534 (1981).
Juhn, S.K., Rybak, L.P. & Fowlks, W.L. Transport characteristics of the blood–perilymph barrier. Am. J. Otolaryngol. 3, 392–396 (1982).
Zhang, W. et al. Perivascular-resident macrophage-like melanocytes in the inner ear are essential for the integrity of the intrastrial fluid-blood barrier. Proc. Natl. Acad. Sci. USA 109, 10388–10393 (2012).
Cucullo, L. et al. A new dynamic in vitro model for the multidimensional study of astrocyte-endothelial cell interactions at the blood-brain barrier. Brain Res. 951, 243–254 (2002).
Duport, S. et al. An in vitro blood-brain barrier model: cocultures between endothelial cells and organotypic brain slice cultures. Proc. Natl. Acad. Sci. USA 95, 1840–1845 (1998).
Lai, C.H. & Kuo, K.H. The critical component to establish in vitro BBB model: pericyte. Brain Res. Brain Res. Rev. 50, 258–265 (2005).
Folkman, J., Haudenschild, C.C. & Zetter, B.R. Long-term culture of capillary endothelial cells. Proc. Natl. Acad. Sci. USA 76, 5217–5221 (1979).
Baudin, B., Bruneel, A., Bosselut, N. & Vaubourdolle, M. A protocol for isolation and culture of human umbilical vein endothelial cells. Nat. Protoc. 2, 481–485 (2007).
Jaffe, E.A., Nachman, R.L., Becker, C.G. & Minick, C.R. Culture of human endothelial cells derived from umbilical veins. Identification by morphologic and immunologic criteria. J. Clin. Invest. 52, 2745–2756 (1973).
Sobczak, M., Dargatz, J. & Chrzanowska-Wodnicka, M. Isolation and culture of pulmonary endothelial cells from neonatal mice. J. Vis. Exp. 46, 2316 (2010).
Bernas, M.J. et al. Establishment of primary cultures of human brain microvascular endothelial cells to provide an in vitro cellular model of the blood-brain barrier. Nat. Protoc. 5, 1265–1272 (2010).
Maier, C.L., Shepherd, B.R., Yi, T. & Pober, J.S. Explant outgrowth, propagation and characterization of human pericytes. Microcirculation 17, 367–380 (2010).
Mogensen, C. et al. Isolation and functional characterization of pericytes derived from hamster skeletal muscle. Acta Physiol. 201, 413–426 (2011).
Weber, S.C. et al. Isolation and culture of fibroblasts, vascular smooth muscle, and endothelial cells from the fetal rat ductus arteriosus. Pediatr. Res. 70, 236–241 (2011).
Bobilya, D.J. A model for transport studies of the blood-brain barrier. Methods Mol. Biol. 637, 149–163 (2010).
Bryan, B.A. & D′Amore, P.A. Pericyte isolation and use in endothelial/pericyte coculture models. Methods Enzymol. 443, 315–331 (2008).
Rops, A.L. et al. Isolation and characterization of conditionally immortalized mouse glomerular endothelial cell lines. Kidney Int. 66, 2193–2201 (2004).
Ribatti, D., Nico, B., Vacca, A., Roncali, L. & Dammacco, F. Endothelial cell heterogeneity and organ specificity. J. Hematother. Stem Cell Res. 11, 81–90 (2002).
Seaman, S. et al. Genes that distinguish physiological and pathological angiogenesis. Cancer cell 11, 539–554 (2007).
Neng, L., Zhang, F., Kachelmeier, A. & Shi, X. Endothelial cell, pericyte, and perivascular resident macrophage-type melanocyte interactions regulate cochlear intrastrial fluid-blood barrier permeability. J. Assoc. Res. Otolaryngol. http://dx.doi.org/10.1007/s10162-012-0365-9 (2012).
Iwagaki, T., Suzuki, T. & Nakashima, T. Development and regression of cochlear blood vessels in fetal and newborn mice. Hear. Res. 145, 75–81 (2000).
Shi, X. Resident macrophages in the cochlear blood-labyrinth barrier and their renewal via migration of bone-marrow-derived cells. Cell Tissue Res. 342, 21–30 (2010).
Tigges, U., Welser-Alves, J.V., Boroujerdi, A. & Milner, R. A novel and simple method for culturing pericytes from mouse brain. Microvasc. Res. 84, 74–80 (2012).
Wisniewska-Kruk, J. et al. A novel co-culture model of the blood-retinal barrier based on primary retinal endothelial cells, pericytes and astrocytes. Exp. Eye Res. 96, 181–190 (2012).
Yamagishi, S. et al. Pigment epithelium-derived factor protects cultured retinal pericytes from advanced glycation end product-induced injury through its antioxidative properties. Biochem. Biophys. Res. Commun. 296, 877–882 (2002).
Dawson, D.W. et al. Pigment epithelium-derived factor: a potent inhibitor of angiogenesis. Science 285, 245–248 (1999).
Shepro, D. & Morel, N.M. Pericyte physiology. FASEB J. 7, 1031–1038 (1993).
Vandenhaute, E. et al. Brain pericytes from stress-susceptible pigs increase blood-brain barrier permeability. Fluids Barriers CNS 9, 11 (2012).
Bouchard, B.A., Shatos, M.A. & Tracy, P.B. Human brain pericytes differentially regulate expression of procoagulant enzyme complexes comprising the extrinsic pathway of blood coagulation. Arterioscler. Thromb. Vasc. Biol. 17, 1–9 (1997).
Provance, D.W. Jr., Wei, M., Ipe, V. & Mercer, J.A. Cultured melanocytes from dilute mutant mice exhibit dendritic morphology and altered melanosome distribution. Proc. Natl. Acad. Sci. USA 93, 14554–14558 (1996).
Gerrity, R.G., Richardson, M., Somer, J.B., Bell, F.P. & Schwartz, C.J. Endothelial cell morphology in areas of in vivo Evans blue uptake in the aorta of young pigs. II. Ultrastructure of the intima in areas of differing permeability to proteins. Am. J. Pathol. 89, 313–334 (1977).
Acknowledgements
This work was supported by US National Institutes of Health (NIH) National Institute on Deafness and Other Communication Disorders (NIDCD) grant no. DC008888-02A1 (X.S.), NIH NIDCD grant no. DC008888-02S1 (X.S.), NIH NIDCD grant no. R01-DC010844 (X.S.), NIH NIDCD grant no. R21-DC12398-01 (X.S.), NIH grant no. P30-DC005983 and NIH National Institute of General Medical Services (NIGMS) grant no. P01-051487-15 (M.A.).
Author information
Authors and Affiliations
Contributions
W.Z. and L.N. created the initial protocol. L.N. refined the protocol. A.H. and M.Z. performed the SEM imaging. A.K. supervised the flow cytometry. X.S., A.F. and M.A. supervised the project. X.S. wrote the manuscript. All the authors discussed the results, procedures and commented on the manuscript at different stages.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Neng, L., Zhang, W., Hassan, A. et al. Isolation and culture of endothelial cells, pericytes and perivascular resident macrophage-like melanocytes from the young mouse ear. Nat Protoc 8, 709–720 (2013). https://doi.org/10.1038/nprot.2013.033
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2013.033
This article is cited by
-
Serum/glucocorticoid-inducible kinase 1 deficiency induces NLRP3 inflammasome activation and autoinflammation of macrophages in a murine endolymphatic hydrops model
Nature Communications (2023)
-
Latrophilin-2 is a novel receptor of LRG1 that rescues vascular and neurological abnormalities and restores diabetic erectile function
Experimental & Molecular Medicine (2022)
-
RNA-sequencing profiling analysis of pericyte-derived extracellular vesicle–mimetic nanovesicles-regulated genes in primary cultured fibroblasts from normal and Peyronie’s disease penile tunica albuginea
BMC Urology (2021)
-
Long non-coding RNA Rian promotes the expression of tight junction proteins in endothelial cells by regulating perivascular-resident macrophage-like melanocytes and PEDF secretion
Human Cell (2021)
-
Pericytes in Vascular Development
Current Tissue Microenvironment Reports (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.