Abstract
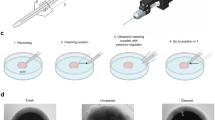
To obtain electrophysiological recordings in brain slices, sophisticated and expensive pieces of equipment can be used. However, costly microscope equipment with infrared differential interference contrast optics is not always necessary or even desirable. For instance, obtaining a randomized unbiased sample in a given preparation would be better accomplished if cells were not directly visualized before recording. In addition, some preparations require thick slices, and direct visualization is not possible. Here we describe a protocol for the 'blind patch clamp method' that we developed several years ago to perform electrophysiological recordings in mammalian brain slices using a standard patch clamp amplifier, dissecting microscope and recording chamber. Overall, it takes approximately 3–4 h to set up this procedure.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Cole, K.S. & Curtis, H.J. Electric impedance of the squid giant axon during activity. J. Gen. Physiol. 22, 649–670 (1939).
Neher, E. & Sakmann, B. Single-channel currents recorded from membrane of denervated frog muscle fibres. Nature 260, 799–802 (1976).
Gray, R. & Johnston, D. Rectification of single GABA-gated chloride channels in adult hippocampal neurons. J. Neurophysiol. 54, 134–142 (1985).
Edwards, F.A., Konnerth, A., Sakmann, B. & Takahashi, T. A thin slice preparation for patch clamp recordings from neurones of the mammalian central nervous system. Pflugers Arch. 414, 600–612 (1989).
Blanton, M.G., LoTurco, J.J. & Kriegstein, A.R. Whole cell recording from neurons in slices of reptilian and mammalian cerebral cortex. J. Neurosci. Methods 30, 203–210 (1989).
Heyward, P., Ennis, M., Keller, A. & Shipley, M.T. Membrane bistability in olfactory bulb mitral cells. J. Neurosci. 21, 5311–5320 (2001).
Margrie, T.W., Brecht, M. & Sakmann, B. In vivo, low-resistance, whole-cell recordings from neurons in the anaesthetized and awake mammalian brain. Pflugers Arch. 444, 491–498 (2002).
Schneider, S.P. Mechanosensory afferent input and neuronal firing properties in rodent spinal laminae III–V: re-examination of relationships with analysis of responses to static and time-varying stimuli. Brain Res. 1034, 71–89 (2005).
Sawatari, A. & Callaway, E.M. Diversity and cell type specificity of local excitatory connections to neurons in layer 3B of monkey primary visual cortex. Neuron 25, 459–471 (2000).
Dalva, M.B., Weliky, M. & Katz, L.C. Relationships between local synaptic connections and orientation domains in primary visual cortex. Neuron 19, 871–880 (1997).
Flint, A.C., Liu, X. & Kriegstein, A.R. Nonsynaptic glycine receptor activation during early neocortical development. Neuron 20, 43–53 (1998).
Pelkey, K.A. et al. Tyrosine Phosphatase STEP is a tonic brake on induction of long-term potentiation. Neuron 34, 127–138 (2002).
LoTurco, J.J. & Kriegstein, A.R. Clusters of coupled neuroblasts in embryonic neocortex. Science 252, 563–566 (1991).
Shin, D. S-h., Ghai, H., Cain, S.W. & Buck, L.T. Gap junctions do not underlie changes in whole-cell conductance in anoxic turtle brain. Comp. Biochem. Physiol. A Mol. Integr. Physiol. 134, 181–194 (2003).
Hamill, O.P., Marty, A., Neher, E., Sakmann, B. & Sigworth, F.J. Improved patch-clamp techniques for high-resolution current recording from cells and cell-free membrane patches. Pflugers Arch. 391, 85–100 (1981).
Spruston, N. & Johnston, D. Perforated patch-clamp analysis of the passive membrane properties of three classes of hippocampal neurons. J. Neurophysiol. 67, 508–529 (1992).
Owens, D.F., Boyce, L.H., Davis, M.B.E. & Kriegstein, A.R. Excitatory GABA responses in embryonic and neonatal cortical slices demonstrated by gramicidin perforated patch recordings and calcium imaging. J. Neurosci. 16, 6414–6423 (1996).
Puche, A.C., Heyward, P. & Shipley, M.T. Transmembrane dye labeling and immunohistochemical staining of electrophysiologically characterized single neurons. J. Neurosci. Methods 137, 235–240 (2004).
Hamam, B.N. & Kennedy, T.E. Visualization of the dendritic arbor of neurons in intact 500 μm thick brain slices. J. Neurosci. Methods 123, 61–67 (2003).
Neher, E. Correction of liquid junction potentials in patch clamp experiments. Methods Enzymol. 207, 123–131 (1992).
Sakmann, B. & Neher, E. Single-Channel Recording (Plenum, New York, 1995).
Flint, A.C., Maisch, U.S., Weishaupt, J., Kriegstein, A.R. & Monyer, H. NR2A subunit expression shortens NMDA receptor synaptic currents in developing neocortex. J. Neurosci. 17, 2469–2476 (1997).
Eberwine, J. et al. Analysis of gene expression in single live neurons. Proc. Natl. Acad. Sci. USA 89, 3010–3014 (1992).
Geiger, J.R. et al. Relative abundance of subunit mRNAs determines gating and Ca2+ permeability of AMPA receptors in principal neurons and interneurons in rat CNS. Neuron 15, 193–204 (1995).
Lambolez, B., Audinat, E., Bochet, P., Crepel, F. & Rossier, J. AMPA receptor subunits expressed by single Purkinje cells. Neuron 9, 247–258 (1992).
Brown, K.T. & Flaming, D.G. Advanced Micropipette Techniques for Cell Physiology (Wiley, New York, 1986).
Committee on Guidelines for the Use of Animals in Neuroscience and Behavioral Research, Institute for Laboratory Animal Research, Division on Earth and Life Studies, National Research Council (U.S.). Guidelines for the Care and Use of Mammals in Neuroscience and Behavioral Research (National Academies Press, Washington, DC, 2003).
Acknowledgements
The electrophysiological traces shown in Fig. 2 were kindly provided by Guillermo Muñoz Elias.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Castañeda-Castellanos, D., Flint, A. & Kriegstein, A. Blind patch clamp recordings in embryonic and adult mammalian brain slices. Nat Protoc 1, 532–542 (2006). https://doi.org/10.1038/nprot.2006.75
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.75
This article is cited by
-
Mechanism behind the neuronal ephaptic coupling during synchronizing by specific brain-triggered wave as neuronal motor toolkit
Scientific Reports (2021)
-
Using pancreas tissue slices for in situ studies of islet of Langerhans and acinar cell biology
Nature Protocols (2014)
-
In vitro functional imaging in brain slices using fast voltage-sensitive dye imaging combined with whole-cell patch recording
Nature Protocols (2008)
-
Paired-recordings from synaptically coupled cortical and hippocampal neurons in acute and cultured brain slices
Nature Protocols (2008)
-
Studying properties of neurotransmitter receptors by non-stationary noise analysis of spontaneous postsynaptic currents and agonist-evoked responses in outside-out patches
Nature Protocols (2007)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.