Abstract
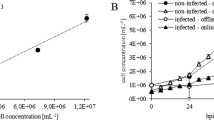
In this paper, a simple and rapid protocol for determination of baculovirus titers based on increasing viable insect cell size/diameter following virus infection is presented. There are different methods available for determining virus titers such as plaque assays end-point dilution, quantitative real-time polymerase chain reaction and flow cytometry. However, most of these methods are time consuming and labor intensive. The titer estimation method presented here can be completed in ∼28 h from start to finish. In this method, the Vi-CELL (Beckman Coulter) was used to measure cell diameter change over a range of virus dilutions, following infection. The cell diameter change data were used to compute the virus titer using a statistical method called the method of moments that we have described previously.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
King, L.A. & Possee, R.D. The Baculovirus Expression System: A Laboratory Guide (Chapman & Hall, London, 1992).
Lynn, D.E. Improved efficiency in determining the titer of the Autographa californica baculovirus nonoccluded virus. Biotechniques 13, 282–285 (1992).
Sussman, D.J. 24-hour assay for estimating the titer of beta-galactosidase-expressing baculovirus. Biotechniques 18, 50–51 (1995).
Yahata, T., Andriole, S., Isselbacher, K.J. & Shioda, T. Estimation of baculovirus titer by beta-galactosidase activity assay of virus preparations. Biotechniques 29, 214–215 (2000).
Cha, H.J., Gotoh, T. & Bentley, W.E. Simplification of titer determination for recombinant baculovirus by green fluorescent protein marker. Biotechniques 23, 782–784 (1997).
Pouliquen, Y., Kolbinger, F., Geisse, S. & Mahnke, M. Automated baculovirus titration assay based on viable cell growth monitoring using a colorimetric indicator. Biotechniques 40, 282–286 (2006).
Lo, H.R. & Chao, Y.C. Rapid titer determination of baculovirus by quantitative real-time polymerase chain reaction. Biotechnol. Prog. 20, 354–360 (2004).
Shen, C.F., Meghrous, J. & Kamen, A. Quantitation of baculovirus particles by flow cytometry. J. Virol. Methods 105, 321–330 (2002).
Mulvania, T., Hayes, B. & Hedin, D.A. Flow cytometric assay for rapid, accurate determination of baculovirus titers. BioProcessing J. 3, 47–53 (2004).
Kitts, P.A. & Possee, R.D. A method for producing recombinant baculovirus expression vectors at high frequency. Biotechniques 14, 810–817 (1993).
Kwon, M.S., Dojima, T., Toriyama, M. & Park, E.Y. Development of an antibody-based assay for determination of baculovirus titers in 10 hours. Biotechnol. Prog. 18, 647–651 (2002).
Mena, J.A., Ramirez, O.T. & Palomares, L.A. Titration of non-occluded baculovirus using a cell viability assay. Biotechniques 34, 260–262 (2003).
Philipps, B., Forstner, M. & Mayr, L.M. Baculovirus expression system for magnetic sorting of infected cells and enhanced titer determination. Biotechniques 36, 80–83 (2004).
Malde, V. & Hunt, I. Calculation of baculovirus titer using a microfluidic-based bioanalyzer. Biotechniques 36, 942–946 (2004).
Chico, E. & Jager, V. New developments and new applications in animal cell technology. in Measurements of Changes in Cell Size Distribution to Monitor Baculovirus Infection of Insect Cells (eds. Merten, O.W. et al.) 329–333 (Kluwer, The Netherlands, 1998).
Palomares, L.A., Pedroza, J.C. & Ramirez, O.T. Cell size as a tool to predict the production of recombinant protein by the insect-cell baculovirus expression system. Biotechnol. Lett. 23, 359–364 (2001).
O'Reilly, D.R., Miller, L.K. & Luckow, V.A. Baculovirus Expression Vectors: A Laboratory Manual (WH Freeman Company, New York, USA, 1992).
Kamen, A.A., Bédard, C., Tom, R., Perret, S. & Jardin, B. Online monitoring of respiration in recombinant-baculovirus infected and uninfected insect cell bioreactor cultures. Biotechnol. Bioeng. 50, 36–48 (1996).
Taticek, R.A. & Shuler, M.L. Effect of elevated oxygen and glutamine levels on foreign protein production at high cell densities using the insect cell–baculovirus expression system. Biotechnol. Bioeng. 54, 142–152 (1997).
Janakiraman, V., Forrest, W.F., Chow, B. & Seshagiri, S. A rapid method for estimation of baculovirus titer based on viable cell size. J. Virol. Methods 132, 48–58 (2006).
Rosinski, M., Reid, S. & Nielsen, L.K. Kinetics of baculovirus replication and release using real-time quantitative polymerase chain reaction. Biotechnol. Bioeng. 77, 476–480 (2002).
Dee, K.U. & Shuler, M.L. Optimization of an assay for baculovirus titer and design of regimens for the synchronous infection of insect cells. Biotechnol. Prog. 13, 14–24 (1997).
Vaughn, J.L., Goodwin, R.H., Tompkins, G.J. & McCawley, P. The establishment of two cell lines from the insect Spodoptera frugiperda (Lepidoptera; Noctuidae). In Vitro 13, 213–217 (1977).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
All the authors hold Genentech Stocks.
Rights and permissions
About this article
Cite this article
Janakiraman, V., Forrest, W. & Seshagiri, S. Estimation of baculovirus titer based on viable cell size. Nat Protoc 1, 2271–2276 (2006). https://doi.org/10.1038/nprot.2006.387
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2006.387
This article is cited by
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.