Abstract
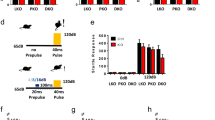
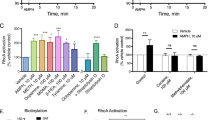
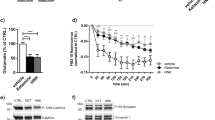
We demonstrated previously that ethanol inhibition of NMDA receptor (NMDAR) function is accompanied by a reduction in tyrosine phosphorylation of Tyr^1472^ on the NR2B subunit, and this action of ethanol is attenuated by a broad spectrum tyrosine phosphatase inhibitor. Here we examined whether this ethanol inhibition of NMDAR activity was due to the actions of STriatal Enriched protein tyrosine Phosphatase (STEP) which has been shown to regulate NMDAR internalization by dephosphorylating Tyr^1472^ on the NR2B subunit. Using whole-cell recordings of pharmacologically isolated NMDAR-mediated excitatory post-synaptic currents (NMDA EPSCs) from hippocampal CA1 pyramidal neurons, we show that intracellular infusion of a substrate-trapping inactive form of STEP (TAT-STEP C/S) significantly blocks ethanol inhibition of NMDA EPSCs. Ethanol does not inhibit NMDA EPSCs or LTP in neurons from STEP knockout mice, but its effect is restored after acute intracellular delivery of wild type TAT-STEP, suggesting that STEP mediates ethanol inhibition of NMDAR function.
Similar content being viewed by others
Article PDF
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Wu, P., Freund, R., Procotor, W. et al. Striatal Enriched Protein Tyrosine Phosphatase mediates Ethanol inhibition of NMDA Receptor Activity. Nat Prec (2008). https://doi.org/10.1038/npre.2008.2473.1
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/npre.2008.2473.1