Abstract
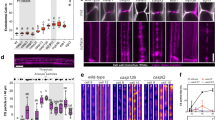
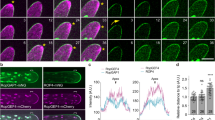
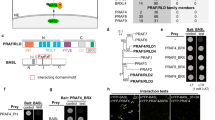
In a striking case of evolutionary convergence, polarized cell layers with ring-like diffusion barriers have evolved in both plant and animal lineages independently. In plants, ring-like Casparian strips become localized by the CASPARIAN STRIP MEMBRANE DOMAIN PROTEINS (CASPs). The mechanism of this striking localization, however, has remained enigmatic. Here we present a genetic screen aimed at isolating determinants of CASP localization. One of the mutants, lord of the rings 2 (lotr2)/exo70a1, displays dramatic de-localization of CASPs into randomly localized microdomains. EXO70A1 is a subunit of the exocyst complex, a central component of secretion in eukaryotes. Irradiation of EXO70 subunit genes in plants has suggested specialization of this conserved complex. Intriguingly, lotr2/exo70a1 does neither affect secretion of the CASPs, nor that of other membrane proteins in the endodermis, thus separating exocyst activity in localization from a general defect in secretion. Our results establish EXO70A1 as a central player in Casparian strip formation, generating a transient positional information that will be translated into a precisely localized cell wall modification.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Priestley, J. & North, E. Physiological studies in plant anatomy – the structure of the endodermis in relation to its function. New Phytol. 21, 113–139 (1922).
Hosmani, P. S. et al. Dirigent domain-containing protein is part of the machinery required for formation of the lignin-based Casparian strip in the root. Proc. Natl Acad. Sci. USA 110, 14498–14503 (2013).
Pfister, A. et al. A receptor-like kinase mutant with absent endodermal diffusion barrier displays selective nutrient homeostasis defects. eLife 3, e03115 (2014).
Kamiya, T. et al. The MYB36 transcription factor orchestrates Casparian strip formation. Proc. Natl Acad. Sci. USA 112, 10533–10538 (2015).
Barberon, M. et al. Adaptation of root function by nutrient-induced plasticity of endodermal differentiation. Cell 164, 447–459 (2016).
Geldner, N. The endodermis. Annu. Rev. Plant Biol. 64, 531–558 (2013).
Barberon, M. & Geldner, N. Radial transport of nutrients: the plant root as a polarized epithelium. Plant Physiol. 166, 528–537 (2014).
Lee, Y., Rubio, M. C., Alassimone, J. & Geldner, N. A mechanism for localized lignin deposition in the endodermis. Cell 153, 402–412 (2013).
Naseer, S. et al. Casparian strip diffusion barrier in Arabidopsis is made of a lignin polymer without suberin. Proc. Natl Acad. Sci. USA 109, 10101–10106 (2012).
Roppolo, D. et al. A novel protein family mediates Casparian strip formation in the endodermis. Nature 473, 380–383 (2011).
Alassimone, J., Naseer, S. & Geldner, N. A developmental framework for endodermal differentiation and polarity. Proc. Natl Acad. Sci. USA 107, 5214–5219 (2010).
Alassimone, J. et al. Polarly localized kinase SGN1 is required for Casparian strip integrity and positioning. Nat. Plants 2, 16113 (2016).
Liberman, L. M., Sparks, E. E., Moreno-Risueno, M. A., Petricka, J. J. & Benfey, P. N. MYB36 regulates the transition from proliferation to differentiation in the Arabidopsis root. Proc. Natl Acad. Sci. USA 112, 12099–12104 (2015).
He, B. & Guo, W. The exocyst complex in polarized exocytosis. 21, 537–542 (2009).
Heider, M. R. & Munson, M. Exorcising the exocyst complex. Traffic 13, 898–907 (2012).
Cvrcková, F. et al. Evolution of the land plant exocyst complexes. Front. Plant Sci. 3, 159 (2012).
Li, S. et al. Expression and functional analyses of EXO70 genes in Arabidopsis implicate their roles in regulating cell type-specific exocytosis. Plant Physiol. 154, 1819–1830 (2010).
Synek, L. et al. AtEXO70A1, a member of a family of putative exocyst subunits specifically expanded in land plants, is important for polar growth and plant development. Plant J. 48, 54–72 (2006).
He, B. et al. Exo70p mediates the secretion of specific exocytic vesicles at early stages of the cell cycle for polarized cell growth. J. Cell Biol. 176, 771–777 (2007).
Novick, P., Field, C. & Schekman, R. Identification of 23 complementation groups required for post-translational events in the yeast secretory pathway. Cell 21, 205–215 (1980).
Novick, P. & Schekman, R. Secretion and cell-surface growth are blocked in a temperature-sensitive mutant of saccharomyces cerevisiae. Proc. Natl Acad. Sci. USA 76, 1858–1862 (1979).
Takano, J. et al. Polar localization and degradation of Arabidopsis boron transporters through distinct trafficking pathways. Proc. Natl Acad. Sci. USA 107, 5220–5225 (2010).
Fendrych, M. et al. The Arabidopsis exocyst complex is involved in cytokinesis and cell plate maturation. Plant Cell 22, 3053–3065 (2010).
Cole, R. A., Synek, L., Zárský, V. & Fowler, J. E. SEC8, a subunit of the putative Arabidopsis exocyst complex, facilitates pollen germination and competitive pollen tube growth. Plant Physiol. 138, 2005–2018 (2005).
Wu, J. et al. Regulation of cytokinesis by exocyst subunit SEC6 and KEULE in Arabidopsis thaliana. Mol. Plant 6, 1863–1876 (2013).
He, B., Xi, F., Zhang, X., Zhang, J. & Guo, W. Exo70 interacts with phospholipids and mediates the targeting of the exocyst to the plasma membrane. EMBO J. 26, 4053–4065 (2007).
Liu, J., Zuo, X., Yue, P. & Guo, W. Phosphatidylinositol 4,5-bisphosphate mediates the targeting of the exocyst to the plasma membrane for exocytosis in mammalian cells. Mol. Biol. Cell 18, 4483–4492 (2007).
Santagata, S. et al. G-protein signaling through tubby proteins. Science 292, 2041–2050 (2001).
Simon, M. L. A. et al. A multi-colour/multi-affinity marker set to visualize phosphoinositide dynamics in Arabidopsis. Plant J. 77, 322–337 (2014).
Cereijido, M., Contreras, R. G. & Shoshani, L. Cell adhesion, polarity, and epithelia in the dawn of metazoans. Physiol. Rev. 84, 1229–1262 (2004).
Bonnett, H. T. The root endodermis: fine structure and function. J. Cell Biol. 37, 199–205 (1968).
Zárský, V. & Potocký, M. Recycling domains in plant cell morphogenesis: small GTPase effectors, plasma membrane signalling and the exocyst. Biochem. Soc. Trans. 38, 723–728 (2010).
Vukašinović, N. et al. Microtubule-dependent targeting of the exocyst complex is necessary for xylem development in Arabidopsis. New Phytol. 213, 1052–1067 (2016).
Mao, H., Nakamura, M., Viotti, C. & Grebe, M. A framework for lateral membrane trafficking and polar tethering of the PEN3 ATP-binding cassette transporter. Plant Physiol. 172, 2245–2260 (2016).
Kulich, I. et al. Cell wall maturation of Arabidopsis trichomes is dependent on exocyst subunit EXO70H4 and involves callose deposition. Plant Physiol. 168, 120–131 (2015).
Drdová, E. J. et al. The exocyst complex contributes to PIN auxin efflux carrier recycling and polar auxin transport in Arabidopsis. Plant J. 73, 709–719 (2013).
Kulich, I. et al. Arabidopsis exocyst subunits SEC8 and EXO70A1 and exocyst interactor ROH1 are involved in the localized deposition of seed coat pectin. New Phytol. 188, 615–625 (2010).
Fendrych, M. et al. Visualization of the exocyst complex dynamics at the plasma membrane of Arabidopsis thaliana. Mol. Biol. Cell 24, 510–520 (2013).
Kulich, I. et al. Arabidopsis exocyst subcomplex containing subunit EXO70B1 is involved in autophagy-related transport to the vacuole. Traffic 14, 1155–1165 (2013).
Coego, A. et al. The TRANSPLANTA collection of Arabidopsis lines: a resource for functional analysis of transcription factors based on their conditional overexpression. Plant J. 77, 944–953 (2014).
Clough, S. J. & Bent, A. F. Floral dip: a simplified method for Agrobacterium-mediated transformation of Arabidopsis thaliana. Plant J. 16, 735–743 (1998).
Mähönen, A. P. et al. A novel two-component hybrid molecule regulates vascular morphogenesis of the Arabidopsis root. Genes Dev. 14, 2938–2943 (2000).
Kurihara, D., Mizuta, Y., Sato, Y. & Higashiyama, T. Clearsee: a rapid optical clearing reagent for whole-plant fluorescence imaging. Development 142, 4168–4179 (2015).
R Core Team. R: A Language and Environment for Statistical Computing (R Foundation for Statistical Computing, 2015).
Acknowledgements
We thank the Central Imaging Facility (CIF) and the Genomic Technologies Facility (GTF) of the University of Lausanne for technical support. B.M. Humbel of the Electron Microscopy Facility (EMF) for expert technical assistance and input, V. Žárský and Y. Jaillais for sharing published material. T. Fitzpatrick of the University of Geneva for kindly providing green house facilities. The Nottingham Arabidopsis Stock Centre (NASC) is acknowledged for providing seeds. J.E.M. Vermeer, J. Alassimone, S. Ammar, C. Schwab, P. Marhavy, J. Weber, I. Acosta, M. Sankar and E. Schmid are thanked for technical expertise, assistance and helpful discussions. This work was funded by grants from the Swiss National Science Foundation (SNF) and the European Research Council (ERC) to N.G., by a short term fellowship of the Société Académique Vaudoise to L. K., by the AgreenSkills+ fellowship programme which has received funding from the EU's Seventh Framework Programme under grant agreement no. FP7-609398 and the LabEx Saclay Plant Sciences-SPS (ANR-10-LABX-0040-SPS) to K.H. (IJPB), by a Japanese Society for the Promotion of Science (JSPS) postdoctoral fellowship for research abroad to S.F., and by an EMBO long-term postdoctoral fellowship to M. B.
Author information
Authors and Affiliations
Contributions
L.K., K.H. and N.G. designed experiments, L.K., K.H., D.D.B., M.B., S.F., J.D. performed experiments L.K., K.H., D.D.B., J.D. and N.G. interpreted the results. R.U. provided unpublished protocols. Manuscript written by L.K., K.H. and N.G. All authors edited and commented on the manuscript.
Corresponding author
Ethics declarations
Competing interests
The corresponding author declares on behalf of all co-authors that there are no competing financial interests
Supplementary information
Supplementary Information
Supplementary Figures 1–8, legend for Supplementary Video 1, Supplementary Methods and Material, Supplementary Tables 1–3, Supplementary References. (PDF 10121 kb)
Supplementary Video 1
CASP1-GFP gradually accumulates at ectopic patches in exo70a1. (AVI 6142 kb)
Rights and permissions
About this article
Cite this article
Kalmbach, L., Hématy, K., De Bellis, D. et al. Transient cell-specific EXO70A1 activity in the CASP domain and Casparian strip localization. Nature Plants 3, 17058 (2017). https://doi.org/10.1038/nplants.2017.58
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nplants.2017.58
This article is cited by
-
Directed growth and fusion of membrane-wall microdomains requires CASP-mediated inhibition and displacement of secretory foci
Nature Communications (2023)
-
Extracellular vesiculo-tubular structures associated with suberin deposition in plant cell walls
Nature Communications (2022)
-
A dirigent family protein confers variation of Casparian strip thickness and salt tolerance in maize
Nature Communications (2022)
-
Transcriptome analysis of different rice cultivars provides novel insights into the rice response to bacterial leaf streak infection
Functional & Integrative Genomics (2020)
-
Surveillance of cell wall diffusion barrier integrity modulates water and solute transport in plants
Scientific Reports (2019)