Abstract
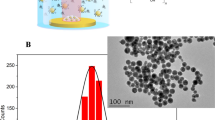
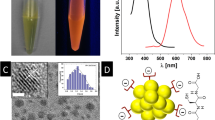
Inorganic nanoparticles are frequently engineered with an organic surface coating to improve their physicochemical properties, and it is well known that their colloidal properties1 may change upon internalization by cells2,3. While the stability of such nanoparticles is typically assayed in simple in vitro tests, their stability in a mammalian organism remains unknown. Here, we show that firmly grafted polymer shells around gold nanoparticles may degrade when injected into rats. We synthesized monodisperse radioactively labelled gold nanoparticles (198Au)4 and engineered an 111In-labelled polymer shell around them5. Upon intravenous injection into rats, quantitative biodistribution analyses performed independently for 198Au and 111In showed partial removal of the polymer shell in vivo. While 198Au accumulates mostly in the liver, part of the 111In shows a non-particulate biodistribution similar to intravenous injection of chelated 111In. Further in vitro studies suggest that degradation of the polymer shell is caused by proteolytic enzymes in the liver. Our results show that even nanoparticles with high colloidal stability can change their physicochemical properties in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Rivera Gil, P. et al. The challenge to relate the physicochemical properties of colloidal nanoparticles to their cytotoxicity. Acc. Chem. Res. 46, 743–749 (2013).
Chanana, M., Rivera Gil, P., Correa-Duarte, M. A., Parak, W. J. & Liz-Marzán, L. M. Physicochemical properties of protein-coated gold nanoparticles in biological fluids and cells before and after proteolytic digestion. Angew. Chem. Int. Ed. 52, 4179–4183 (2013).
Chen, H. W., Zou, P., Connarn, J., Paholak, H. & Sun, D. X. Intracellular dissociation of a polymer coating from nanoparticles. Nano Res. 5, 815–825 (2012).
Semmler-Behnke, M. et al. Biodistribution of 1.4- and 18-nm gold particles in rats. Small 4, 2108–2111 (2008).
Ali, Z. et al. Multifunctional nanoparticles for dual imaging. Anal. Chem. 83, 2877–2882 (2011).
Kreyling, W. G. et al. Interspecies comparison of phagolysosomal pH in alveolar acrophages. Inhal. Toxicol. 3, 91–100 (1991).
Soenen, S. J. H. et al. Intracellular nanoparticle coating stability determines nanoparticle diagnostics efficacy and cell functionality. Small 6, 2136–2145 (2010).
Levy, M. et al. Long term in vivo biotransformation of iron oxide nanoparticles. Biomaterials 32, 3988–3999 (2011).
Derfus, A. M., Chan, W. C. W. & Bhatia, S. N. Probing the cytotoxicity of semiconductor quantum dots. Nano Lett. 4, 11–18 (2004).
Kirchner, C. et al. Cytotoxicity of colloidal CdSe and CdSe/ZnS nanoparticles. Nano Lett. 5, 331–338 (2005).
Kittler, S., Greulich, C., Diendorf, J., Koller, M. & Epple, M. Toxicity of silver nanoparticles increases during storage because of slow dissolution under release of silver ions. Chem. Mater. 22, 4548–4554 (2010).
Caballero-Díaz, E. et al. The toxicity of silver nanoparticles depends on their uptake by cells and thus on their surface chemistry. Part. Part. Syst. Charact. 30, 1079–1085 (2013).
Bose, K., Koch, M., Cavelius, C., Kiemer, A. K. & Kraegeloh, A. A correlative analysis of gold nanoparticles internalized by A549 cells. Part. Part. Syst. Charact. 31, 439–448 (2014).
Lunov, O. et al. Lysosomal degradation of the carboxydextran shell of coated superparamagnetic iron oxide nanoparticles and the fate of professional phagocytes. Biomaterials 31, 9015–9022 (2010).
Tietze, R. et al. Efficient drug-delivery using magnetic nanoparticles—biodistribution and therapeutic effects in tumour bearing rabbits. Nanomed. Nanotechnol. 9, 961–971 (2013).
Ducongé, F. et al. Fluorine-18-labeled phospholipid quantum dot micelles for in vivo multimodal imaging from whole body to cellular scales. Bioconj. Chem. 19, 1921–1926 (2008).
Sée, V. et al. Cathepsin l digestion of nanobioconjugates upon endocytosis. ACS Nano 3, 2461–2468 (2009).
Wang, F. et al. The biomolecular corona is retained during nanoparticle uptake and protects the cells from the damage induced by cationic nanoparticles until degraded in the lysosomes. Nanomed. Nanotechnol. 9, 1159–1168 (2013).
Schleh, C. et al. Size and surface charge of gold nanoparticles determine absorption across intestinal barriers and accumulation in secondary target organs after oral administration. Nanotoxicology 6, 36–46 (2012).
Hirn, S. et al. Particle size-dependent and surface charge-dependent biodistribution of gold nanoparticles after intravenous administration. Eur. J. Pharm. Biopharm. 77, 407–416 (2011).
Oeff, K., Krentz, K. & Kessel, M. J-131-Clearance der normalen und pathologischen Magenschleimhaut. Klin. Wochenschr. 33, 59–63 (1955).
Kreyling, W. G. et al. Air–blood barrier translocation of tracheally instilled gold nanoparticles inversely depends on particle size. ACS Nano 8, 222–233 (2014).
Lipka, M. et al. Biodistribution of PEG-modified gold nanoparticles following intratracheal instillation and intravenous injection. Biomaterials 31, 6574–6581 (2010).
Pan, Y. et al. Gold nanoparticles of diameter 1.4 nm trigger necrosis by oxidative stress and mitochondrial damage. Small 5, 2067–2076 (2009).
Acknowledgements
This work was supported by the Deutsche Forschungsgemeinschaft (DFG grant ‘PA794/11-1’ to W.J.P., grant ‘SPP1313’ to W.G.K.) and the European Commission Seventh Framework Programme (EC-FP7 grant ‘ENPRA NMP4-SL-2009-228789’ and ‘Neuronano NMP4-SL-2008-214547’ to W.G.K.). The authors would like to thank N. Senger, S. Kaidel, B. Kupferschmid and B. Krieger for technical assistance and to D. Hühn, R. Gill, M. Klapper and K. Dawson for scientific discussions.
Author information
Authors and Affiliations
Contributions
W.G.K. and W.J.P. conceived the idea, designed and analysed the experiments, and mainly wrote the manuscript. All authors discussed the results, contributed to data analysis, and commented on the manuscript. A.M.A., Z.A., J.M.M., T.R. and I.R.d.L. prepared and characterized the nanoparticles. R.H., K.K., D.J.d.A. and J.R. performed the in vitro experiments. A.W., N.H., S.H. and G.K.S. carried out in vivo experiments with radionucleotides. F.A. and R.U. carried out in vivo experiments with fluorescent nanoparticles. M.G. performed transmission electron microscopy measurements.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary information (PDF 17955 kb)
Rights and permissions
About this article
Cite this article
Kreyling, W., Abdelmonem, A., Ali, Z. et al. In vivo integrity of polymer-coated gold nanoparticles. Nature Nanotech 10, 619–623 (2015). https://doi.org/10.1038/nnano.2015.111
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nnano.2015.111
This article is cited by
-
Biological Interaction and Imaging of Ultrasmall Gold Nanoparticles
Nano-Micro Letters (2024)
-
Fabrication of surface-functionalizable amphiphilic curcumin nanogels for biosensing and biomedical applications
Chemical Papers (2024)
-
Digital micelles of encoded polymeric amphiphiles for direct sequence reading and ex vivo label-free quantification
Nature Chemistry (2023)
-
Multimodal radiolabeled gold nanoparticle molecular probes: synthesis, imaging, and applications
Journal of Radioanalytical and Nuclear Chemistry (2023)
-
Development of a multi-route physiologically based pharmacokinetic (PBPK) model for nanomaterials: a comparison between a traditional versus a new route-specific approach using gold nanoparticles in rats
Particle and Fibre Toxicology (2022)