Abstract
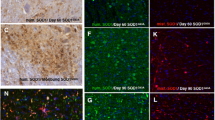
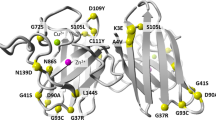
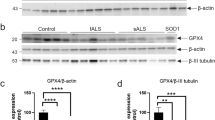
Many mutations confer one or more toxic function(s) on copper/zinc superoxide dismutase 1 (SOD1) that impair motor neuron viability and cause familial amyotrophic lateral sclerosis (FALS). Using a conformation-specific antibody that detects misfolded SOD1 (C4F6), we found that oxidized wild-type SOD1 and mutant SOD1 share a conformational epitope that is not present in normal wild-type SOD1. In a subset of human sporadic ALS (SALS) cases, motor neurons in the lumbosacral spinal cord were markedly C4F6 immunoreactive, indicating that an aberrant wild-type SOD1 species was present. Recombinant, oxidized wild-type SOD1 and wild-type SOD1 immunopurified from SALS tissues inhibited kinesin-based fast axonal transport in a manner similar to that of FALS-linked mutant SOD1. Our findings suggest that wild-type SOD1 can be pathogenic in SALS and identify an SOD1-dependent pathogenic mechanism common to FALS and SALS.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
Accession codes
References
Tandan, R. & Bradley, W.G. Amyotrophic lateral sclerosis. Part 1. Clinical features, pathology, and ethical issues in management. Ann. Neurol. 18, 271–280 (1985).
Valdmanis, P.N., Daoud, H., Dion, P.A. & Rouleau, G.A. Recent advances in the genetics of amyotrophic lateral sclerosis. Curr. Neurol. Neurosci. Rep. 9, 198–205 (2009).
Valentine, J.S., Doucette, P.A. & Zittin Potter, S. Copper-zinc superoxide dismutase and amyotrophic lateral sclerosis. Annu. Rev. Biochem. 74, 563–593 (2005).
Bruijn, L.I. et al. Aggregation and motor neuron toxicity of an ALS-linked SOD1 mutant independent from wild-type SOD1. Science 281, 1851–1854 (1998).
Chattopadhyay, M. & Valentine, J.S. Aggregation of copper-zinc superoxide dismutase in familial and sporadic ALS. Antioxid. Redox. Signal 11, 1603–1614 (2009).
Furukawa, Y., Fu, R., Deng, H.X., Siddique, T. & O'Halloran, T.V. Disulfide cross-linked protein represents a significant fraction of ALS-associated Cu, Zn-superoxide dismutase aggregates in spinal cords of model mice. Proc. Natl. Acad. Sci. USA 103, 7148–7153 (2006).
Molnar, K.S. et al. A common property of amyotrophic lateral sclerosis-associated variants: destabilization of the Cu/Zn superoxide dismutase electrostatic loop. J. Biol. Chem. 284, 30965–30973 (2009).
Prudencio, M., Hart, P.J., Borchelt, D.R. & Andersen, P.M. Variation in aggregation propensities among ALS-associated variants of SOD1: correlation to human disease. Hum. Mol. Genet. 18, 3217–3226 (2009).
Wang, Q., Johnson, J.L., Agar, N.Y. & Agar, J.N. Protein aggregation and protein instability govern familial amyotrophic lateral sclerosis patient survival. PLoS Biol. 6, e170 (2008).
Morfini, G.A. et al. Axonal transport defects in neurodegenerative diseases. J. Neurosci. 29, 12776–12786 (2009).
Ezzi, S.A., Urushitani, M. & Julien, J.P. Wild-type superoxide dismutase acquires binding and toxic properties of ALS-linked mutant forms through oxidation. J. Neurochem. 102, 170–178 (2007).
Gruzman, A. et al. Common molecular signature in SOD1 for both sporadic and familial amyotrophic lateral sclerosis. Proc. Natl. Acad. Sci. USA 104, 12524–12529 (2007).
Beckman, J.S., Estevez, A.G., Crow, J.P. & Barbeito, L. Superoxide dismutase and the death of motoneurons in ALS. Trends Neurosci. 24, S15–S20 (2001).
Bredesen, D.E., Ellerby, L.M., Hart, P.J., Wiedau-Pazos, M. & Valentine, J.S. Do posttranslational modifications of CuZnSOD lead to sporadic amyotrophic lateral sclerosis? Ann. Neurol. 42, 135–137 (1997).
Kabashi, E., Valdmanis, P.N., Dion, P. & Rouleau, G.A. Oxidized/misfolded superoxide dismutase-1: the cause of all amyotrophic lateral sclerosis? Ann. Neurol. 62, 553–559 (2007).
Durazo, A. et al. Metal-free superoxide dismutase-1 and three different ALS variants share a similar partially unfolded {beta}-barrel at physiological temperature. J. Biol. Chem. 277, 15923–15931 (2009).
Estévez, A.G. et al. Induction of nitric oxide–dependent apoptosis in motor neurons by zinc-deficient superoxide dismutase. Science 286, 2498–2500 (1999).
Rakhit, R. et al. Oxidation-induced misfolding and aggregation of superoxide dismutase and its implications for amyotrophic lateral sclerosis. J. Biol. Chem. 277, 47551–47556 (2002).
Banci, L. et al. Metal-free superoxide dismutase forms soluble oligomers under physiological conditions: a possible general mechanism for familial ALS. Proc. Natl. Acad. Sci. USA 104, 11263–11267 (2007).
Urushitani, M., Ezzi, S.A. & Julien, J.P. Therapeutic effects of immunization with mutant superoxide dismutase in mice models of amyotrophic lateral sclerosis. Proc. Natl. Acad. Sci. USA 104, 2495–2500 (2007).
Brady, S.T., Lasek, R.J. & Allen, R.D. Fast axonal transport in extruded axoplasm from squid giant axon. Science 218, 1129–1131 (1982).
Gros-Louis, F., Soucy, G., Lariviere, R. & Julien, J.P. Intracerebroventricular infusion of monoclonal antibody or its derived Fab fragment against misfolded forms of SOD1 mutant delays mortality in a mouse model of ALS. J. Neurochem. 113, 1188–1199 (2010).
Fujiwara, N. et al. Oxidative modification to cysteine sulfonic acid of Cys111 in human copper-zinc superoxide dismutase. J. Biol. Chem. 282, 35933–35944 (2007).
Tiwari, A. et al. Metal deficiency increases aberrant hydrophobicity of mutant superoxide dismutases that cause amyotrophic lateral sclerosis. J. Biol. Chem. 284, 27746–27758 (2009).
Rakhit, R. et al. Monomeric Cu,Zn-superoxide dismutase is a common misfolding intermediate in the oxidation models of sporadic and familial amyotrophic lateral sclerosis. J. Biol. Chem. 279, 15499–15504 (2004).
Svensson, A.K., Bilsel, O., Kondrashkina, E., Zitzewitz, J.A. & Matthews, C.R. Mapping the folding free energy surface for metal-free human Cu,Zn superoxide dismutase. J. Mol. Biol. 364, 1084–1102 (2006).
Brady, S.T., Lasek, R.J. & Allen, R.D. Video microscopy of fast axonal transport in extruded axoplasm: a new model for study of molecular mechanisms. Cell Motil. 5, 81–101 (1985).
Morfini, G., Szebenyi, G., Elluru, R., Ratner, N. & Brady, S.T. Glycogen synthase kinase 3 phosphorylates kinesin light chains and negatively regulates kinesin-based motility. EMBO J. 21, 281–293 (2002).
Morfini, G. et al. JNK mediates pathogenic effects of polyglutamine-expanded androgen receptor on fast axonal transport. Nat. Neurosci. 9, 907–916 (2006).
Morfini, G.A. et al. Pathogenic huntingtin inhibits fast axonal transport by activating JNK3 and phosphorylating kinesin. Nat. Neurosci. 12, 864–871 (2009).
Fabian, M.A. et al. A small molecule-kinase interaction map for clinical kinase inhibitors. Nat. Biotechnol. 23, 329–336 (2005).
Munoz, L. et al. A novel p38 alpha MAPK inhibitor suppresses brain proinflammatory cytokine up-regulation and attenuates synaptic dysfunction and behavioral deficits in an Alzheimer's disease mouse model. J. Neuroinflammation 4, 21 (2007).
Kerman, A. et al. Amyotrophic lateral sclerosis is a non-amyloid disease in which extensive misfolding of SOD1 is unique to the familial form. Acta Neuropathol. 119, 335–344 (2010).
Liu, H.N. et al. Lack of evidence of monomer/misfolded superoxide dismutase-1 in sporadic amyotrophic lateral sclerosis. Ann. Neurol. 66, 75–80 (2009).
Shibata, N., Asayama, K., Hirano, A. & Kobayashi, M. Immunohistochemical study on superoxide dismutases in spinal cords from autopsied patients with amyotrophic lateral sclerosis. Dev. Neurosci. 18, 492–498 (1996).
Shibata, N. et al. Cu/Zn superoxide dismutase–like immunoreactivity in Lewy body–like inclusions of sporadic amyotrophic lateral sclerosis. Neurosci. Lett. 179, 149–152 (1994).
Watanabe, M. et al. Histological evidence of protein aggregation in mutant SOD1 transgenic mice and in amyotrophic lateral sclerosis neural tissues. Neurobiol. Dis. 8, 933–941 (2001).
Rakhit, R. et al. An immunological epitope selective for pathological monomer-misfolded SOD1 in ALS. Nat. Med. 13, 754–759 (2007).
Pasinelli, P. et al. Amyotrophic lateral sclerosis-associated SOD1 mutant proteins bind and aggregate with Bcl-2 in spinal cord mitochondria. Neuron 43, 19–30 (2004).
Urushitani, M. et al. Chromogranin-mediated secretion of mutant superoxide dismutase proteins linked to amyotrophic lateral sclerosis. Nat. Neurosci. 9, 108–118 (2006).
Vande Velde, C., Miller, T.M., Cashman, N.R. & Cleveland, D.W. Selective association of misfolded ALS-linked mutant SOD1 with the cytoplasmic face of mitochondria. Proc. Natl. Acad. Sci. USA 105, 4022–4027 (2008).
Lindberg, M.J., Normark, J., Holmgren, A. & Oliveberg, M. Folding of human superoxide dismutase: disulfide reduction prevents dimerization and produces marginally stable monomers. Proc. Natl. Acad. Sci. USA 101, 15893–15898 (2004).
De Vos, K.J., Grierson, A.J., Ackerley, S. & Miller, C.C. Role of axonal transport in neurodegenerative diseases. Annu. Rev. Neurosci. 31, 151–173 (2008).
Ström, A.L. et al. Retrograde axonal transport and motor neuron disease. J. Neurochem. 106, 495–505 (2008).
Collard, J.F., Cote, F. & Julien, J.P. Defective axonal transport in a transgenic mouse model of amyotrophic lateral sclerosis. Nature 375, 61–64 (1995).
Williamson, T.L. & Cleveland, D.W. Slowing of axonal transport is a very early event in the toxicity of ALS-linked SOD1 mutants to motor neurons. Nat. Neurosci. 2, 50–56 (1999).
Fischer, L.R. & Glass, J.D. Axonal degeneration in motor neuron disease. Neurodegener. Dis. 4, 431–442 (2007).
Strange, R.W. et al. Variable metallation of human superoxide dismutase: atomic resolution crystal structures of Cu-Zn, Zn-Zn and as-isolated wild-type enzymes. J. Mol. Biol. 356, 1152–1162 (2006).
Hayward, L.J. et al. Decreased metallation and activity in subsets of mutant superoxide dismutases associated with familial amyotrophic lateral sclerosis. J. Biol. Chem. 277, 15923–15931 (2002).
Wang, L. et al. Wild-type SOD1 overexpression accelerates disease onset of a G85R SOD1 mouse. Hum. Mol. Genet. 18, 1642–1651 (2009).
Acknowledgements
We are grateful to J. Landers and P. Sapp for DNA sequencing analysis of the SALS cases employed in this study, L. Hayward, A. Tiwari and R.-J. Chain for help with expression of recombinant wild-type SOD1, S. Berth, A. Leitman and M. Saparauskaite for help with axoplasm vesicle transport assays, A. Kaminska and L. Molla for help with biochemical experiments in squid axoplasm, M. Prudencio and D. Borchelt for cell lysates containing SOD1 G93 mutants, C. Vanderburg, E. Tamrazian, A. Bialik and the Diabetes and Endocrinology Research Center (University of Massachusetts Medical Center) for assistance with immunohistochemistry, K. Fitch and the Massachusetts Alzheimer Disease Research Center (P50AG005134) for assistance with human tissue samples, J. Zitzewitz for C6A/C111S-SOD1 protein, A. Weiss for assistance with mice, K. Green at the University of Massachusetts Medical Center Proteomics and Mass Spectrometry Facility for analysis of C6A/C111S-SOD1, and G. Petsko for insightful dialogue and support. We acknowledge financial support from the ALS Therapy Alliance-CVS Pharmacy (D.A.B. and G.M.), 2007/2008 Marine Biological Laboratory research fellowships (G.M.), the ALS Association (D.A.B., R.H.B. Jr, G.M. and S.T.B.), the US National Institutes of Health (D.A.B. (National Institute on Neurological Disorders and Stroke, RO1NS067206-01), R.H.B. Jr (National Institute on Neurological Disorders and Stroke, U01NS05225-03, R01NS050557-05, 1RC1NS068391-01 and 1RC2NS070342-01), S.T.B. and J.N.A.), Canadian Institutes of Health Research (J.-P.J.), the Angel Fund (R.H.B. Jr) and Project ALS (R.H.B. Jr).
Author information
Authors and Affiliations
Contributions
D.A.B., G.M., S.T.B. and R.H.B. Jr wrote the manuscript. D.A.B. prepared recombinant and immunopurified SOD1 proteins. G.M., Y.S. and S.T.B. performed vesicle motility assays and biochemical experiments in isolated squid axoplasm. N.M.K. and J.N.A. performed the mass spectrometry. F.G.-L. and J.-P.J. prepared the mutant-specific antibodies. P.P. made the SOD1 exon deleted constructs. H.G., D.M.-Y. and M.P.F. provided human tissues for staining. D.A.B., B.A.F. and N.L. performed western analyses. All of the authors reviewed and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–3 and Supplementary Tables 1–3 (PDF 426 kb)
Rights and permissions
About this article
Cite this article
Bosco, D., Morfini, G., Karabacak, N. et al. Wild-type and mutant SOD1 share an aberrant conformation and a common pathogenic pathway in ALS. Nat Neurosci 13, 1396–1403 (2010). https://doi.org/10.1038/nn.2660
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.2660
This article is cited by
-
Translating the ALS Genetic Revolution into Therapies: A Review
Current Treatment Options in Neurology (2024)
-
Unveiling the dark side of glucose-regulated protein 78 (GRP78) in cancers and other human pathology: a systematic review
Molecular Medicine (2023)
-
Amyotrophic lateral sclerosis: translating genetic discoveries into therapies
Nature Reviews Genetics (2023)
-
Selective removal of misfolded SOD1 delays disease onset in a mouse model of amyotrophic lateral sclerosis
Cellular and Molecular Life Sciences (2023)
-
Persistent NRG1 Type III Overexpression in Spinal Motor Neurons Has No Therapeutic Effect on ALS-Related Pathology in SOD1G93A Mice
Neurotherapeutics (2023)