Abstract
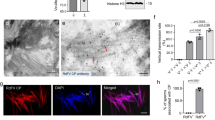
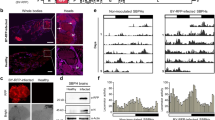
Many insects, including mosquitoes, planthoppers, aphids and leafhoppers, are the hosts of bacterial symbionts and the vectors for transmitting viral pathogens1–3. In general, symbiotic bacteria can indirectly affect viral transmission by enhancing immunity and resistance to viruses in insects3–5. Whether symbiotic bacteria can directly interact with the virus and mediate its transmission has been unknown. Here, we show that an insect symbiotic bacterium directly harbours a viral pathogen and mediates its transovarial transmission to offspring. We observe rice dwarf virus (a plant reovirus) binding to the envelopes of the bacterium Sulcia, a common obligate symbiont of leafhoppers6–8, allowing the virus to exploit the ancient oocyte entry path of Sulcia in rice leafhopper vectors. Such virus–bacterium binding is mediated by the specific interaction of the viral capsid protein and the Sulcia outer membrane protein. Treatment with antibiotics or antibodies against Sulcia outer membrane protein interferes with this interaction and strongly prevents viral transmission to insect offspring. This newly discovered virus–bacterium interaction represents the first evidence that a viral pathogen can directly exploit a symbiotic bacterium for its transmission. We believe that such a model of virus–bacterium communication is a common phenomenon in nature.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Gray, S. M. & Banerjee, N. Mechanisms of arthropod transmission of plant and animal viruses. Microbiol. Mol. Biol. Rev. 63, 128–148 (1999).
Baumann, P. Biology bacteriocyte-associated endosymbionts of plant sap-sucking insects. Annu. Rev. Microbiol. 59, 155–189 (2005).
Feldhaar, H. Bacterial symbionts as mediators of ecologically important traits of insect host. Ecol. Entomol. 36, 533–543 (2011).
Martinez, J. et al. Symbionts commonly provide broad spectrum resistance to viruses in insects: a comparative analysis of Wolbachia strains. PLoS Pathog. 10, e1004369 (2014).
Pinheiro, P. V., Kliot, A., Ghanim, M. & Cilia, M. Is there a role for symbiotic bacteria in plant virus transmission by insects? Curr. Opin. Insect Sci. 8, 69–78 (2015).
Koga, R., Bennett, G. M., Cryan, J. R. & Moran, N. A. Evolutionary replacement of obligate symbionts in an ancient and diverse insect lineage. Environ. Microbiol. 15, 2073–2081 (2013).
Noda, H. et al. Bacteriome-associated endosymbionts of the green rice leafhopper Nephotettix cincticeps (Hemiptera: cicadellidae). Appl. Entomol. Zool. 47, 217–225 (2012).
Hibino, H. Biology and epidemiology of rice viruses. Annu. Rev. Phytopathol. 34, 249–274 (1996).
Koga, R., Meng, X. Y., Tsuchida, T. & Fukatsu, T. Cellular mechanism for selective vertical transmission of an obligate insect symbiont at the bacteriocyte–embryo interface. Proc. Natl Acad. Sci. USA. 109, E1230–E1237 (2012).
Huo, Y. et al. Transovarial transmission of a plant virus is mediated by vitellogenin of its insect vector. PLoS Pathog. 10, e1003949 (2014).
Hogenhout, S. A., Ammar, E. D., Whitfield, A. E. & Redinbaugh, M. G. Insect vector interactions with persistently transmitted viruses. Annu. Rev. Phytopathol. 46, 327–359 (2008).
Fukushi, T. Transmission of the virus through the eggs of an insect vector. Proc. Inme. Acad. 9, 457–460 (1933).
Nasu, S. Electron microscopic studies on transovarial passage of rice dwarf virus. Jpn J. Appl. Entomol. Zool. 9, 225–237 (1965).
Chen, Q. et al. Tubular structure induced by a plant virus facilitates viral spread in its vector insect. PLoS Pathog. 8, e1003032 (2012).
Moran, N., Tran, P. & Gerardo, N. Symbiosis and insect diversification: an ancient symbiont of sap-feeding insects from the bacterial phylum Bacteroidetes. Appl. Environ. Microbiol. 71, 8802–8810 (2005).
Miyazaki, N. et al. Electron microscopic imaging revealed the flexible filamentous structure of the cell attachment protein P2 of rice dwarf virus located around the icosahedral 5-fold axes. J. Biochem. 159, 181–190 (2016).
Orlova, E. V. How viruses infect bacteria? EMBO J. 28, 797–798 (2009).
Honda, K. et al. Retention of rice dwarf virus by descendants of pairs of viruliferous vector insects after rearing for 6 years. Phytopathology 97, 712–716 (2007).
Bennett, G. M. & Moran, N. A. Heritable symbiosis: the advantages and perils of an evolutionary rabbit hole. Proc. Natl Acad. Sci. USA. 112, 10169–10176 (2015).
Chang, H. H. et al. Complete genome sequence of ‘Candidatus Sulcia muelleri’ ML, an obligate nutritional symbiont of maize leafhopper (Dalbulus maidis). Genome Announc. 3, e01483–14 (2015).
Brasset, E. et al. Viral particles of the endogenous retrovirus ZAM from Drosophila melanogaster use a pre-existing endosome/exosome pathway for transfer to the oocyte. Retrovirology 3, 25 (2006).
Márquez, L. M., Redman, R. S., Rodriguez, R. J. & Roossinck, M. J. A virus in a fungus in a plant: three-way symbiosis required for thermal tolerance. Science 315, 513–515 (2007).
Vautrin, E. & Vavre, F. Interactions between vertically transmitted symbionts: cooperation or conflict? Trends Microbiol. 17, 95–99 (2009).
Wei, T., Chen, H., Ichiki-Uehara, T., Hibino, H. & Omura, T. Entry of rice dwarf virus into cultured cells of its insect vector involves clathrin-mediated endocytosis. J. Virol. 81, 7811–7815 (2007).
Jia, D. et al. Development of an insect vector cell culture and RNA interference system to investigate the functional role of Fijivirus replication protein. J. Virol. 86, 5800–5807 (2012).
Omura, T., Morinaka, T., Inoue, H. & Saito, Y. Purification and some properties of rice gall dwarf virus, a new phytoreovirus. Phytopathology 72, 1246–1249 (1982).
Liu, S., Ding, Z., Zhang, C., Yang, B. & Liu, Z. Gene knockdown by intro-thoracic injection of double-stranded RNA in the brown planthopper, Nilaparvata lugens. Insect Biochem. Mol. Biol. 40, 666–671 (2010).
Lane, H. A. & Nigg, E. A. Antibody microinjection reveals an essential role for human polo-like kinase 1 (Plk1) in the functional maturation of mitotic centrosomes. J. Cell Biol. 135, 1701–1713 (1996).
Noda, H., Koizumi, Y., Zhang, Q. & Deng, K. J. Infection density of Wolbachia and incompatibility level in two planthopper species, Laodelphax striatellus and Sogatella furcifera. Insect Biochem. Mol. Biol. 31, 727–737 (2001).
Lan, H. et al. Small interfering RNA pathway modulates initial viral infection in midgut epithelium of insect after ingestion of virus. J. Virol. 90, 917–929 (2016).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2-ΔΔCT method. Methods 25, 402–408 (2001).
Acknowledgements
This work was supported by the National Science Foundation for Outstanding Youth (grant no. 31325023), the National Basic Research Program of China (973 Program, no. 2014CB138400), the National Natural Science Foundation of China (grant no. 31571979) and the FAFU Foundation for Outstanding Youth (grant no. XJQ201507).
Author information
Authors and Affiliations
Contributions
All authors read and approved the manuscript. D.J., Q.M., Y.C., Y.Li and T.W. conceived and designed the study, and wrote the paper. D.J., Q.M. and Y.C. contributed equally to this work, performed most experiments and helped with data analysis. Y.Liu and H.C. performed the transmission electron microscopy. W.W. and X.Z. performed gene cloning and immunoprecipitation. Q.C. performed RT–qPCR experiments. Y.Li and T.W. discussed the data and revised the manuscript. T.W. and Y.Li organized and directed the project.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–5 (PDF 968 kb)
Rights and permissions
About this article
Cite this article
Jia, D., Mao, Q., Chen, Y. et al. Insect symbiotic bacteria harbour viral pathogens for transovarial transmission. Nat Microbiol 2, 17025 (2017). https://doi.org/10.1038/nmicrobiol.2017.25
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2017.25
This article is cited by
-
Diversity and functional characteristics of culturable bacterial endosymbionts from cassava whitefly biotype Asia II-5, Bemisia tabaci
3 Biotech (2024)
-
Obligate mutualistic heritable symbiosis in sap-feeding insects: an intricate relationship in nature
Symbiosis (2024)
-
Arboviruses and symbiotic viruses cooperatively hijack insect sperm-specific proteins for paternal transmission
Nature Communications (2023)
-
Autophagy mediates a direct synergistic interaction during co-transmission of two distinct arboviruses by insect vectors
Science China Life Sciences (2023)
-
Microorganisms in the reproductive tissues of arthropods
Nature Reviews Microbiology (2020)