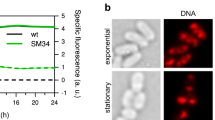
Abstract
Bacteria growing under different conditions experience a broad range of demand on the rate of protein synthesis, which profoundly affects cellular resource allocation. During fast growth, protein synthesis has long been known to be modulated by adjusting the ribosome content, with the vast majority of ribosomes engaged at a near-maximal rate of elongation. Here, we systematically characterize protein synthesis by Escherichia coli, focusing on slow-growth conditions. We establish that the translational elongation rate decreases as growth slows, exhibiting a Michaelis–Menten dependence on the abundance of the cellular translational apparatus. However, an appreciable elongation rate is maintained even towards zero growth, including the stationary phase. This maintenance, critical for timely protein synthesis in harsh environments, is accompanied by a drastic reduction in the fraction of active ribosomes. Interestingly, well-known antibiotics such as chloramphenicol also cause a substantial reduction in the pool of active ribosomes, instead of slowing down translational elongation as commonly thought.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Change history
14 July 2017
In the PDF version of this article previously published, the year of publication provided in the footer of each page and in the 'How to cite' section was erroneously given as 2017, it should have been 2016. This error has now been corrected. The HTML version of the article was not affected.
References
Scott, M., Gunderson, C. W., Mateescu, E. M., Zhang, Z. & Hwa, T. Interdependence of cell growth and gene expression: origins and consequences. Science 330, 1099–1102 (2010).
Gerosa, L., Kochanowski, K., Heinemann, M. & Sauer, U. Dissecting specific and global transcriptional regulation of bacterial gene expression. Mol. Syst. Biol. 9, 658 (2013).
Li, G.-W., Burkhardt, D., Gross, C. & Weissman, J. S. Quantifying absolute protein synthesis rates reveals principles underlying allocation of cellular resources. Cell 157, 624–635 (2014).
Hui, S. et al. Quantitative proteomic analysis reveals a simple strategy of global resource allocation in bacteria. Mol. Syst. Biol. 11, 784 (2015).
You, C. et al. Coordination of bacterial proteome with metabolism by cyclic AMP signalling. Nature 500, 301–306 (2013).
Basan, M. et al. Overflow metabolism in Escherichia coli results from efficient proteome allocation. Nature 528, 99–104 (2015).
Maaløe, O. in Biological Regulation and Development (ed. Goldberger, R. F. ) 487–542 (Plenum, 1979).
Neidhardt, F. C. & Magasanik, B. Studies on the role of ribonucleic acid in the growth of bacteria. Biochim. Biophys. Acta 42, 99–116 (1960).
Dalbow, D. G. & Young, R. Synthesis time of β-galactosidase in Escherichia coli B/r as a function of growth rate. Biochem. J. 150, 13–20 (1975).
Young, R. & Bremer, H. Polypeptide-chain-elongation rate in Escherichia coli B/r as a function of growth rate. Biochem. J. 160, 185–194 (1976).
Pedersen, S. Escherichia coli ribosomes translate in vivo with variable rate. EMBO J. 3, 2895–2898 (1984).
Klumpp, S., Scott, M., Pedersen, S. & Hwa, T. Molecular crowding limits translation and cell growth. Proc. Natl Acad. Sci. USA 110, 16754–16759 (2013).
Ryals, J., Little, R. & Bremer, H. Control of rRNA and tRNA syntheses in Escherichia coli by guanosine tetraphosphate. J. Bacteriol. 151, 1261–1268 (1982).
Kolter, R., Siegele, D. A. & Tormo, A. The stationary phase of the bacterial life cycle. Annu. Rev. Microbiol. 47, 855–874 (1993).
Koch, A. L. & Deppe, C. S. In vivo assay of protein synthesizing capacity of Escherichia coli from slowly growing chemostat cultures. J. Mol. Biol. 55, 549–562 (1971).
Schleif, R., Hess, W., Finkelstein, S. & Ellis, D. Induction kinetics of the l-arabinose operon of Escherichia coli. J. Bacteriol. 115, 9–14 (1973).
Link, H., Fuhrer, T., Gerosa, L., Zamboni, N. & Sauer, U. Real-time metabolome profiling of the metabolic switch between starvation and growth. Nat. Methods 12, 1091–1097 (2015).
Zhu, M., Dai, X. & Wang, Y. P. Real time determination of bacterial in vivo ribosome translation elongation speed based on LacZα complementation system. Nucleic Acids Res. http://dx.doi.org/10.1093/nar/gkw698 (2016).
Basan, M. et al. Inflating bacterial cells by increased protein synthesis. Mol. Syst. Biol. 11, 836 (2015).
Bremer, H. & Dennis, P. P. in Escherichia coli and Salmonella 2nd edn (ed. Neidhardt, F. C. ) 1553–1569 (American Society of Microbiology, 1996).
Neidhardt, F. C., Ingraham, J. L. & Schaechter, M. Physiology of the Bacterial Cell—A Molecular Approach (Sinauer Associates, 1990).
Harvey, R. J. & Koch, A. L. How partially inhibitory concentrations of chloramphenicol affect the growth of Escherichia coli. Antimicrob. Agents Chemother. 18, 323–337 (1980).
Chopra, I. & Howe, T. G. Bacterial resistance to the tetracyclines. Microbiol. Rev. 42, 707–724 (1978).
Day, L. E. Tetracycline inhibition of cell-free protein synthesis. II. Effect of the binding of tetracycline to the components of the system. J. Bacteriol. 92, 197–203 (1966).
Pestka, S. Binding of [14C]erythromycin to Escherichia coli ribosomes. Antimicrob. Agents Chemother. 6, 474–478 (1974).
Seo, H.-S. et al. EF-G-dependent GTPase on the ribosome. conformational change and fusidic acid inhibition. Biochemistry 45, 2504–2514 (2006).
Okura, A., Kinoshita, T. & Tanaka, N. Formation of fusidic acid-G factor-GDP-ribosome complex and the relationship to the inhibition of GTP hydrolysis. J. Antibiot. 24, 655–661 (1971).
Uemura, S. et al. Real-time tRNA transit on single translating ribosomes at codon resolution. Nature 464, 1012–1017 (2010).
Hughes, J. & Mellows, G. Inhibition of isoleucyl-transfer ribonucleic acid synthetase in Escherichia coli by pseudomonic acid. Biochem. J. 176, 305–318 (1978).
Dennis, P. P. Effects of chloramphenicol on the transcriptional activities of ribosomal RNA and ribosomal protein genes in Escherichia coli. J. Mol. Biol. 108, 535–546 (1976).
Ehrenberg, M. & Kurland, C. G. Costs of accuracy determined by a maximal growth rate constraint. Q. Rev. Biophys. 17, 45–82 (1984).
Zhang, G. et al. Global and local depletion of ternary complex limits translational elongation. Nucleic Acids Res. 38, 4778–4787 (2010).
Dong, H., Nilsson, L. & Kurland, C. G. Co-variation of tRNA abundance and codon usage in Escherichia coli at different growth rates. J. Mol. Biol. 260, 649–663 (1996).
Johansson, M., Bouakaz, E., Lovmar, M. & Ehrenberg, M. The kinetics of ribosomal peptidyl transfer revisited. Mol. Cell 30, 589–598 (2008).
Siibak, T. et al. Erythromycin- and chloramphenicol-induced ribosomal assembly defects are secondary effects of protein synthesis inhibition. Antimicrob. Agents Chemother. 53, 563–571 (2009).
Polikanov, Y. S., Blaha, G. M. & Steitz, T. A. How hibernation factors RMF, HPF, and YfiA turn off protein synthesis. Science 336, 915–918 (2012).
Sin, C., Chiarugi, D. & Valleriani, A. Quantitative assessment of ribosome drop-off in E. coli. Nucleic Acids Res. 44, 2528–2537 (2016).
Forchhammer, J. & Lindahl, L. Growth rate of polypeptide chains as a function of the cell growth rate in a mutant of Escherichia coli 15. J. Mol. Biol. 55, 563–568 (1971).
Harvey, R. J. Fraction of ribosomes synthesizing protein as a function of specific growth rate. J. Bacteriol. 114, 287–293 (1973).
Godson, G. N. & Sinsheimer, R. L. Use of Brij lysis as a general method to prepare polyribosomes from Escherichia coli. Biochim. Biophys. Acta 149, 489–495 (1967).
Nath, K. & Koch, A. L. Protein degradation in Escherichia coli. I. Measurement of rapidly and slowly decaying components. J. Biol. Chem. 245, 2889–2900 (1970).
Mandelstam, J. Turnover of protein in starved bacteria and its relationship to the induced synthesis of enzyme. Nature 179, 1179–1181 (1957).
Milon, P. et al. The nucleotide-binding site of bacterial translation initiation factor 2 (IF2) as a metabolic sensor. Proc. Natl Acad. Sci. USA 103, 13962–13967 (2006).
Hauser, R. et al. Rsfa (YbeB) proteins are conserved ribosomal silencing factors. PLoS Genet. 8, e1002815 (2012).
Izutsu, K., Wada, A. & Wada, C. Expression of ribosome modulation factor (RMF) in Escherichia coli requires ppGpp. Genes Cells 6, 665–676 (2001).
Shimizu, Y. Biochemical aspects of bacterial strategies for handling the incomplete translation processes. Front. Microbiol. 5, 170 (2014).
Subramaniam, A. R., Zid, B. M. & O'Shea, E. K. An integrated approach reveals regulatory controls on bacterial translation elongation. Cell 159, 1200–1211 (2014).
Dresden, M. H. & Hoagland, M. B. Polyribosomes of Escherichia coli. Re-formation during recovery from glucose starvation. J. Biol. Chem. 242, 1069–1073 (1967).
Koch, A. L. The adaptive responses of Escherichia coli to a feast and famine existence. Adv. Microb. Physiol. 6, 147–217 (1971).
Moore, S. D. & Sauer, R. T. Ribosome rescue: tmRNA tagging activity and capacity in Escherichia coli. Mol. Microbiol. 58, 456–466 (2005).
Soupene, E. et al. Physiological studies of Escherichia coli strain MG1655: growth defects and apparent cross-regulation of gene expression. J. Bacteriol. 185, 5611–5626 (2003).
Lyons, E., Freeling, M., Kustu, S. & Inwood, W. Using genomic sequencing for classical genetics in E. coli K12. PLoS ONE 6, e16717 (2011).
Steinsiek, S. & Bettenbrock, K. Glucose transport in Escherichia coli mutant strains with defects in sugar transport systems. J. Bacteriol. 194, 5897–5908 (2012).
Langley, K. E., Villarejo, M. R., Fowler, A. V., Zamenhof, P. J. & Zabin, I. Molecular basis of beta-galactosidase alpha-complementation. Proc. Natl Acad. Sci. USA 72, 1254–1257 (1975).
Datsenko, K. A. & Wanner, B. L. One-step inactivation of chromosomal genes in Escherichia coli K-12 using PCR products. Proc. Natl Acad. Sci. USA 97, 6640–6645 (2000).
Lutz, R. & Bujard, H. Independent and tight regulation of transcriptional units in Escherichia coli via the LacR/O, the TetR/O and AraC/I1-I2 regulatory elements. Nucleic Acids Res. 25, 1203–1210 (1997).
Morris, D. R. & Hansen, M. T. Influence of polyamine limitation on the chain growth rate of beta-galactosidase and of its messenger ribonucleic acid. J. Bacteriol. 116, 588–592 (1973).
Cayley, S., Lewis, B. A., Guttman, H. J. & Record, M. T. Jr. Characterization of the cytoplasm of Escherichia coli K-12 as a function of external osmolarity. Implications for protein–DNA interactions in vivo. J. Mol. Biol. 222, 281–300 (1991).
Neidhardt, F. C., Bloch, P. L. & Smith, D. F. Culture medium for enterobacteria. J. Bacteriol. 119, 736–747 (1974).
Andersson, D. I., Bohman, K., Isaksson, L. A. & Kurland, C. G. Translation rates and misreading characteristics of rpsD mutants in Escherichia coli. Mol. Gen. Genet. 187, 467–472 (1982).
Miller, J. H. Experiments in Molecular Genetics (Cold Spring Harbor Laboratory, 1972).
Vidal-Aroca, F. et al. One-step high-throughput assay for quantitative detection of β-galactosidase activity in intact Gram-negative bacteria, yeast, and mammalian cells. Biotechniques 40, 433–440, 436, 438 passim (2006).
Martin, L., Che, A. & Endy, D. Gemini, a bifunctional enzymatic and fluorescent reporter of gene expression. PLoS ONE 4, e7569 (2009).
Janssen, B. D., Diner, E. J. & Hayes, C. S. Analysis of aminoacyl- and peptidyl-tRNAs by gel electrophoresis. Methods Mol. Biol. 905, 291–309 (2012).
Balakrishnan, R., Oman, K., Shoji, S., Bundschuh, R. & Fredrick, K. The conserved GTPase LepA contributes mainly to translation initiation in Escherichia coli. Nucleic Acids Res. 42, 13370–13383 (2014).
Qin, D. & Fredrick, K. Analysis of polysomes from bacteria. Methods Enzymol. 530, 159–172 (2013).
Deutsh, E. W. et al. A guided tour of the trans-proteomic pipeline. Proteomics 10, 1150–1159 (2010).
Craig, R. & Beavis, R. C. TANDEM matching proteins with tandem mass spectra. Bioinformatics 20, 1466–1467 (2004).
Lam, H. et al. Building consensus spectral libraries for peptide identification in proteomics. Nat. Methods 5, 873–875 (2008).
Sperling, E., Bunner, A. E., Sykes, M. T. & Williamson, J. R. Quantitative analysis of isotope distributions in proteomic mass spectrometry using least-square Fourier transform convolution. Anal. Chem. 80, 4906–4917 (2008).
Sykes, M. T., Sperling, E., Chen, S. S. & Williamson, J. R. Quantitation of the ribosomal protein autoregulatory network using mass spectrometry. Anal. Chem. 82, 5038–5045 (2010).
Acknowledgements
The authors are grateful for discussions with numerous colleagues including Z. Ignatova, S. Klumpp, S. Pedersen, S. Schink, J. Silverman, M. Scott, R. Young and members of the Hwa laboratory at various stages of this work. This research is supported by NIH grant R01GM109069 (to T.H.) and grant 31530081 of the National Natural Science Fund of the People's Republic of China (NSFC; to Y.P.W.). K.F. acknowledges support from NIH grant R01GM072528. J.R.W. acknowledges NIH grant GM118850. M.Z. acknowledges financial support from the China Scholarship Council (CSC; 201306010039).
Author information
Authors and Affiliations
Contributions
X.D., M.Z., M.W. and T.H. designed the study. X.D., M.Z., R.B. and V.P. performed experiments. X.D., M.Z., M.W., H.O., V.P., J.R.W., K.F., Y.-P.W. and T.H. analysed the data. X.D., M.Z., M.W., Y.-P.W. and T.H. wrote the paper and the Supplementary Information.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information
Supplementary Notes 1–5, Supplementary Tables 1–12, Supplementary Figures 1–20, Supplementary References (PDF 4670 kb)
Supplementary Table 9
Proteome abundances of individual r-proteins under nutrient limitation and Cm inhibition. (XLSX 48 kb)
Supplementary Table 10
Proteome abundance of individual r-proteins of the reference condition. (XLSX 64 kb)
Rights and permissions
About this article
Cite this article
Dai, X., Zhu, M., Warren, M. et al. Reduction of translating ribosomes enables Escherichia coli to maintain elongation rates during slow growth. Nat Microbiol 2, 16231 (2017). https://doi.org/10.1038/nmicrobiol.2016.231
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nmicrobiol.2016.231
This article is cited by
-
A coarse-grained bacterial cell model for resource-aware analysis and design of synthetic gene circuits
Nature Communications (2024)
-
Costs of ribosomal RNA stabilization affect ribosome composition at maximum growth rate
Communications Biology (2024)
-
Inherited chitinases enable sustained growth and rapid dispersal of bacteria from chitin particles
Nature Microbiology (2023)
-
Shaping bacterial gene expression by physiological and proteome allocation constraints
Nature Reviews Microbiology (2023)
-
Growth-rate dependency of ribosome abundance and translation elongation rate in Corynebacterium glutamicum differs from that in Escherichia coli
Nature Communications (2023)