Abstract
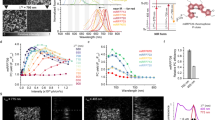
All biological reactions depend on the diffusion and re-localization of biomolecules. Our understanding of biological processes requires accurate measurement of biomolecule mobility in living cells. Currently, approaches for investigating the mobility of biomolecules are generally restricted to measuring either fast or slow diffusion kinetics. We describe the development and application of a photoconvertible fluorescent protein, Phamret, that can be highlighted by UV light stimulation inducing a change in fluorescence emission from cyan fluorescent protein (CFP) to photoactivated GFP (PA-GFP). Phamret can be monitored by single excitation-dual emission mode for visualization of molecular dynamics for a broad range of kinetics. We also devised a microscopy-based method to measure the diffusion coefficient from the fluorescence decay after photostimulation of Phamret, enabling analysis of diffusion kinetics ranging from less than 0.1 μm2/s up to ∼100 μm2/s, and found significant changes in free protein movement during cell-cycle progression.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Lippincott-Schwartz, J., Snapp, E. & Kenworthy, A. Studying protein dynamics in living cells. Nat. Rev. Mol. Cell Biol. 2, 444–456 (2001).
Zhang, J., Campbell, R.E., Ting, A.Y. & Tsien, R.Y. Creating new fluorescent probes for cell biology. Nat. Rev. Mol. Cell Biol. 3, 906–918 (2002).
Miyawaki, A. Visualization of the spatial and temporal dynamics of intracellular signaling. Dev. Cell 4, 295–305 (2003).
Medina, M.A. & Schwille, P. Fluorescence correlation spectroscopy for the detection and study of single molecules in biology. Bioessays 24, 758–764 (2002).
Reits, E.A. & Neefjes, J.J. From fixed to FRAP: measuring protein mobility and activity in living cells. Nat. Cell Biol. 3, E145–E147 (2001).
White, J. & Stelzer, E. Photobleaching GFP reveals protein dynamics inside live cells. Trends Cell Biol. 9, 61–65 (1999).
Lukyanov, K.A., Chudakov, D.M., Lukyanov, S. & Verkhusha, V.V. Innovation: Photoactivatable fluorescent proteins. Nat. Rev. Mol. Cell Biol. 6, 885–891 (2005).
Patterson, G.H. & Lippincott-Schwartz, J. A photoactivatable GFP for selective photolabeling of proteins and cells. Science 297, 1873–1877 (2002).
Verkhusha, V.V. & Sorkin, A. Conversion of the monomeric red fluorescent protein into a photoactivatable probe. Chem. Biol. 12, 279–285 (2005).
Chudakov, D.M. et al. Kindling fluorescent proteins for precise in vivo photolabeling. Nat. Biotechnol. 21, 191–194 (2003).
Ando, R., Mizuno, H. & Miyawaki, A. Regulated fast nucleocytoplasmic shuttling observed by reversible protein highlighting. Science 306, 1370–1373 (2004).
Ando, R., Hama, H., Yamamoto-Hino, M., Mizuno, H. & Miyawaki, A. An optical marker based on the UV-induced green-to-red photoconversion of a fluorescent protein. Proc. Natl. Acad. Sci. USA 99, 12651–12656 (2002).
Wiedenmann, J. et al. EosFP, a fluorescent marker protein with UV-inducible green-to-red fluorescence conversion. Proc. Natl. Acad. Sci. USA 101, 15905–15910 (2004).
Chudakov, D.M. et al. Photoswitchable cyan fluorescent protein for protein tracking. Nat. Biotechnol. 22, 1435–1439 (2004).
Tsutsui, H., Karasawa, S., Shimizu, H., Nukina, N. & Miyawaki, A. Semi-rational engineering of a coral fluorescent protein into an efficient highlighter. EMBO Rep. 6, 233–238 (2005).
Gurskaya, N.G. et al. Engineering of a monomeric green-to-red photoactivatable fluorescent protein induced by blue light. Nat. Biotechnol. 24, 461–465 (2006).
Ballestrem, C., Wehrle-Haller, B. & Imhof, B.A. Actin dynamics in living mammalian cells. J. Cell Sci. 111, 1649–1658 (1998).
Phair, R.D. & Misteli, T. High mobility of proteins in the mammalian cell nucleus. Nature 404, 604–609 (2000).
Llopis, J., McCaffery, J.M., Miyawaki, A., Farquhar, M.G. & Tsien, R.Y. Measurement of cytosolic, mitochondrial, and Golgi pH in single living cells with green fluorescent proteins. Proc. Natl. Acad. Sci. USA 95, 6803–6808 (1998).
Recalcati, S., Menotti, E. & Kühn, L.C. Peroxisomal targeting of mammalian hydroxyacid oxidase 1 requires the C-terminal tripeptide SKI. J. Cell Sci. 114, 1625–1629 (2001).
Kimura, H. & Cook, P.R. Kinetics of core histones in living human cells: little exchange of H3 and H4 and some rapid exchange of H2B. J. Cell Biol. 153, 1341–1353 (2001).
Gerlich, D. et al. Global chromosome positions are transmitted through mitosis in mammalian cells. Cell 112, 751–764 (2003).
Pack, C., Saito, K., Tamura, M. & Kinjo, M. Microenvironment and effect of energy depletion in the nucleus analyzed by mobility of multiple oligomeric EGFPs. Biophys. J. 91, 3921–3936 (2006).
Braga, J., Desterro, J.M.P. & Carmo-Fonseca, M. Intracellular macromolecular mobility measured by fluorescence recovery after photobleaching with confocal laser scanning microscopes. Mol. Biol. Cell 15, 4749–4760 (2004).
Meyvis, T.K., De Smedt, S.C., Van Oostveldt, P. & Demeester, J. Fluorescence recovery after photobleaching: a versatile tool for mobility and interaction measurements in pharmaceutical research. Pharm. Res. 16, 1153–1162 (1999).
Axelrod, D., Koppel, D.E., Schlessinger, J., Elson, E. & Webb, W.W. Mobility measurement by analysis of fluorescence photobleaching recovery kinetics. Biophys. J. 16, 1055–1069 (1976).
Calapez, A. et al. The intranuclear mobility of messenger RNA binding proteins is ATP dependent and temperature sensitive. J. Cell Biol. 59, 795–805 (2002).
Shimozono, S. et al. Concatenation of cyan and yellow fluorescent proteins for efficient resonance energy transfer. Biochemistry 45, 6267–6271 (2006).
Acknowledgements
We thank H. Kimura and Y. Yoneda (Osaka University) for providing the cDNA encoding H2B-GFP and PP2Cγ, and fibrillalin, respectively. We also thank D. Goto and I. Kotera for helpful comments. We also thank F. Inagaki and N. Noda for help with gel filtration analysis. This work was partially supported by grants from Scientific Research on Advanced Medical Technology of the Ministry of Labor, Health and Welfare of Japan, Precursory Research for Embryonic Science and Technology of the Japan Science and Technology Agency, and the Japanese Ministry of Education, Science and Technology.
Author information
Authors and Affiliations
Contributions
T.M. performed experiments, analyzed data and prepared the manuscript; A.M. contributed to data analysis; T.N. contributed to the conceptual development and experimental design and performed experiments, analyzed data and prepared the manuscript.
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–4, Supplementary Methods (PDF 496 kb)
Supplementary Video 1
Tracking of mitochondria fusion with mitochondria-Phamret in HeLa cells. (MOV 3037 kb)
Supplementary Video 2
Tracking distribution of activated H2B-Phamret throughout mitosis in HeLa cells. (MOV 2172 kb)
Supplementary Video 3
Tracking rapid diffusion of PP2Cγ-Phamret within nucleus in HeLa Cells. (MOV 20357 kb)
Rights and permissions
About this article
Cite this article
Matsuda, T., Miyawaki, A. & Nagai, T. Direct measurement of protein dynamics inside cells using a rationally designed photoconvertible protein. Nat Methods 5, 339–345 (2008). https://doi.org/10.1038/nmeth.1193
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmeth.1193
This article is cited by
-
Switchable stimulated Raman scattering microscopy with photochromic vibrational probes
Nature Communications (2021)
-
Hybrid cell reactor system from Escherichia coli protoplast cells and arrayed lipid bilayer chamber device
Scientific Reports (2018)
-
TGFβ pathway limits dedifferentiation following WNT and MAPK pathway activation to suppress intestinal tumourigenesis
Cell Death & Differentiation (2017)
-
Fusion protein analysis reveals the precise regulation between Hsp70 and Hsp100 during protein disaggregation
Scientific Reports (2017)
-
A possible pathogenetic factor of sickle-cell disease based on fluorescent analysis via an optofluidic resonator
Scientific Reports (2017)