Abstract
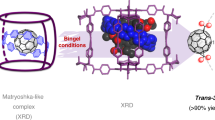
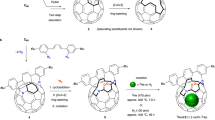
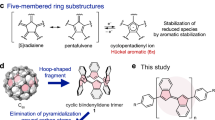
One abiding surprise in fullerene science is that Ih-symmetric buckminsterfullerene C60 (ref. 1) (Ih–C60 or #1,812C60, the nomenclature specified by symmetry or by Fowler’s spiral algorithm2) remains the sole C60 species experimentally available. Setting it apart from the other 1,811 topological isomers (isobuckminsterfullerenes) is its exclusive conformity with the isolated-pentagon rule3, which states that stable fullerenes have isolated pentagons. Although gas-phase existence of isobuckminsterfullerenes has long been suspected4,5,6,7, synthetic efforts have yet to yield successful results. Here, we report the realization of two isobuckminsterfullerenes by means of chlorination of the respective C2v- and Cs-symmetric C60 cages. These chlorinated species, #1,809C60Cl8(1) and #1,804C60Cl12(2), were isolated in experimentally useful yields. Structural characterization by crystallography unambiguously established the unique pentagon–pentagon ring fusions. These distinct structural features are directly responsible for the regioselectivity observed in subsequent substitution of chlorines, and also render these unprecedented derivatives of C60 isomers important for resolving the long-standing puzzle of fullerene formation by the Stone–Wales transformation scheme8,9,10,11.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kroto, H. W., Heath, J. R., O’Brien, S. C., Curl, R. F. & Smalley, R. E. C60: Buckminsterfullerene. Nature 318, 162–163 (1985).
Fowler, P. W. & Manolopoulos, D. E. An Atlas of Fullerenes (Oxford Univ. Press, Oxford, 1995).
Kroto, H. W. The stability of the fullerenes Cn, with n=24, 28, 32, 36, 50, 60 and 70. Nature 329, 529–531 (1987).
Yang, S. H., Pettiette, C. L., Conceicao, J., Cheshnovsky, O. & Smalley, R. E. UPS of buckminsterfullerene and other large clusters of carbon. Chem. Phys. Lett. 139, 233–238 (1987).
Hunter, J., Fye, J. & Jarrold, M. F. Annealing C60+: Synthesis of fullerenes and large carbon rings. Science 260, 784–786 (1993).
von Helden, G., Gotts, N. G. & Bowers, M. T. Experimental evidence for the formation of fullerenes by collisional heating of carbon rings in the gas phase. Nature 363, 60–63 (1993).
Xie, S. Y., Deng, S. L., Huang, R. B., Yu, L. J. & Zheng, L. S. Five isomers of C60 generated in microwave plasma of chloroform. Chem. Phys. Lett. 343, 458–464 (2001).
Stone, A. J. & Wales, D. J. Theoretical studies of icosahedral C60 and some related species. Chem. Phys. Lett. 128, 501–503 (1986).
Hawkins, J. M., Nambu, M. & Meyer, A. Resolution and configurational stability of the chiral fullerenes C76, C78, and C84: A limit for the activation energy of the Stone–Wales transformation. J. Am. Chem. Soc. 116, 7642–7645 (1994).
Austin, S.J., Fowler, P. W., Manolopoulos, D. E. & Zerbetto, F. The Stone–Wales map for C60 . Chem. Phys. Lett. 235, 146–151 (1995).
Bettinger, H. F., Yakobson, B. I. & Scuseria, G. E. Scratching the surface of buckminsterfullerene: The barriers for Stone–Wales transformation through symmetric and asymmetric transition states. J. Am. Chem. Soc. 125, 5572–5580 (2003).
Krätschmer, W., Lamb, L. D., Fostiropoulos, K. & Huffman, D. R. Solid C60: A new form of carbon. Nature 347, 354–358 (1990).
Xie, S. Y. et al. Capturing the labile fullerene[50] as C50Cl10 . Science 304, 699–699 (2004).
Chen, Z. F. The smaller fullerene C50, isolated as C50Cl10 . Angew. Chem. Int. Ed. 43, 4690–4691 (2004).
Lu, X. & Chen, Z. F. Curved π-conjugation, aromaticity, and the related chemistry of small fullerenes (<C60) and single-walled carbon nanotubes. Chem. Rev. 105, 3643–3696 (2005).
Dunsch, L. & Yang, S. F. Metal nitride cluster fullerenes: Their current state and future prospects. Small 3, 1298–1320 (2007).
Taylor, R. Surprises, serendipity, and symmetry in fullerene chemistry. Synlett 6, 776–793 (2000).
Taylor, R. Why fluorinate fullerenes? J. Fluor. Chem. 125, 359–368 (2004).
Han, X. et al. Crystal structures of Saturn-like C50Cl10 and pineapple-shaped C64Cl4: Geometric implications of double- and triple-pentagon-fused chlorofullerenes. Angew. Chem. Int. Ed. 47, 5340–5343 (2008).
Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 113, 7756–7764 (2000).
Troshin, P. A. et al. Isolation of two seven-membered ring C58 fullerene derivatives: C58F17CF3 and C58F18 . Science 309, 278–281 (2005).
Jia, J. F., Wu, H. S., Xu, X. H., Zhang, X. M. & Jiao, H. J. Fused five-membered rings determine the stability of C60F60 . J. Am. Chem. Soc. 130, 3985–3988 (2008).
Chen, Z. F. & King, R. B. Spherical aromaticity: Recent work on fullerenes, polyhedral boranes, and related structures. Chem. Rev. 105, 3613–3642 (2005).
Solomons, T. W. G. & Fryhle, C. B. Organic Chemistry 8th edn 672–673 (Wiley, New York, 2004).
Avent, A. G. et al. The structure of C60Ph5Cl and C60Cl5H, formed via electrophilic aromatic substitution. J. Chem. Soc. Chem. Commun. 1994, 1463–1464 (1994).
Birkett, P. R. et al. Arylation of [60]fullerene via electrophilic aromatic substitution involving the electrophile C60Cl6: Frontside nucleophilic substitution of fullerenes. J. Chem. Soc. Perkin Trans. 2. 1997, 1121–1125 (1997).
Hirsch, A. & Brettreich, M. Fullerenes: Chemistry and Reactions 279–282 (Wiley–VCH, Weinheim, 2005).
Haddon, R. C. π-electrons in three dimensions. Acc. Chem. Res. 21, 243–249 (1988).
Goroff, N. S. Mechanism of fullerene formation. Acc. Chem. Res. 29, 77–83 (1996).
Suenaga, K. et al. Imaging active topological defects in carbon nanotubes. Nature Nanotechnol. 2, 358–360 (2007).
Acknowledgements
We gratefully acknowledge helpful discussions with R. F. Curl, Y. D. Li, L. B. Gan, Y. L. Li, G. M. Blackburn and N. F. Zheng. We thank Y. Q. Feng for HPLC support; H. Y. Huang, J. L. Ye, Q. He, L. Zhang, J. M. Li, W. Z. Wen and Y. S. Zhou for experimental support; and G. M. Blackburn for revising the English of the manuscript. This work was supported by the NNSF of China (grant nos 20525103, 20531050, 20721001, 20571062, 20425312) and the 973 Program (grant no. 2007CB815301).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Information
Supplementary Information (PDF 1807 kb)
Rights and permissions
About this article
Cite this article
Tan, YZ., Liao, ZJ., Qian, ZZ. et al. Two Ih-symmetry-breaking C60 isomers stabilized by chlorination. Nature Mater 7, 790–794 (2008). https://doi.org/10.1038/nmat2275
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nmat2275
This article is cited by
-
Diels–Alder cycloaddition of the silicon–silicon bonds at pentagon junctions of Si-doped non-IPR and SW defective fullerenes
Monatshefte für Chemie - Chemical Monthly (2021)
-
Electroactive Polymerization Behaviors of Fused-Pentagon Chlorofullerenes: #1809C60Cl8 and #271C50Cl10
Journal of Cluster Science (2017)
-
Theoretical study of the structures and first hyperpolarizabilities of C60Cl n and Li@C60Cl n (n = 4, 6, 8, 10)
Journal of Molecular Modeling (2016)
-
Cage connectivity and frontier π orbitals govern the relative stability of charged fullerene isomers
Nature Chemistry (2015)
-
Progress of nanoscience in China
Frontiers of Physics (2014)