Abstract
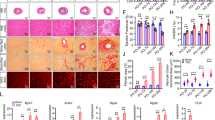
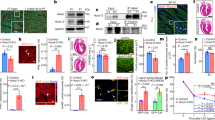
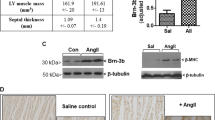
Hypertrophy represents the major physiological response of the heart to adapt to chronically enhanced workload, but is also crucial in the development of heart failure. Although we know of numerous inducers of cardiac hypertrophy, little is known about mechanisms that limit cardiac hypertrophy. Here, we describe the transcriptional repressor NAB1 as an endogenous regulator of cardiac growth. We identified NAB1 as being upregulated in both mouse and human heart failure. Nab1 is highly expressed in mammalian cardiac myocytes and it inhibited cardiomyocyte hypertrophy through repression of its targets, transcription factor Egr. Transgenic mice with cardiac-specific overexpression of Nab1 showed that Nab1 is a potent inhibitor of cardiac growth in response to pathological stimuli in vivo. Nab1 overexpression suppressed adrenergically induced and pressure overload–induced hypertrophy, whereas physiological growth during development and in response to exercise was not affected. These findings implicate the Nab1-Egr1 axis as a crucial regulator of pathological cardiac growth.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Towbin, J.A. & Bowles, N.E. The failing heart. Nature 415, 227–233 (2002).
Dorn, G.W., II, Robbins, J. & Sugden, P.H. Phenotyping hypertrophy: eschew obfuscation. Circ. Res. 92, 1171–1175 (2003).
Lohse, M.J., Engelhardt, S. & Eschenhagen, T. What is the role of b-adrenergic signaling in heart failure? Circ. Res. 93, 896–906 (2003).
Molkentin, J.D. & Dorn, I.G., II Cytoplasmic signaling pathways that regulate cardiac hypertrophy. Annu. Rev. Physiol. 63, 391–426 (2001).
Sussman, M.A. et al. Prevention of cardiac hypertrophy in mice by calcineurin inhibition. Science 281, 1690–1693 (1998).
Molkentin, J.D. et al. A calcineurin-dependent transcriptional pathway for cardiac hypertrophy. Cell 93, 215–228 (1998).
Zhang, C.L. et al. Class II histone deacetylases act as signal-responsive repressors of cardiac hypertrophy. Cell 110, 479–488 (2002).
Chien, K.R. & Olson, E.N. Converging pathways and principles in heart development and disease: CV@CSH. Cell 110, 153–162 (2002).
Russo, M.W., Sevetson, B.R. & Milbrandt, J. Identification of NAB1, a repressor of NGFI-A- and Krox20-mediated transcription. Proc. Natl Acad. Sci. USA 92, 6873–6877 (1995).
Swirnoff, A.H. et al. Nab1, a corepressor of NGFI-A (Egr-1), contains an active transcriptional repression domain. Mol. Cell. Biol. 18, 512–524 (1998).
Thiel, G. & Cibelli, G. Regulation of life and death by the zinc finger transcription factor Egr-1. J. Cell. Physiol. 193, 287–292 (2002).
Warner, L.E. et al. Mutations in the early growth response 2 (EGR2) gene are associated with hereditary myelinopathies. Nat. Genet. 18, 382–384 (1998).
Engelhardt, S., Hein, L., Wiesmann, F. & Lohse, M.J. Progressive hypertrophy and heart failure in β1-adrenergic receptor transgenic mice. Proc. Natl Acad. Sci. USA 96, 7059–7064 (1999).
Topilko, P. et al. Krox-20 controls myelination in the peripheral nervous system. Nature 371, 796–799 (1994).
Svaren, J. et al. Novel mutants of NAB corepressors enhance activation by Egr transactivators. EMBO J. 17, 6010–6019 (1998).
Allen, D.L. et al. Cardiac and skeletal muscle adaptations to voluntary wheel running in the mouse. J. Appl. Physiol. 90, 1900–1908 (2001).
Frey, N., Katus, H.A., Olson, E.N. & Hill, J.A. Hypertrophy of the heart: a new therapeutic target? Circulation 109, 1580–1589 (2004).
Esposito, G. et al. Genetic alterations that inhibit in vivo pressure-overload hypertrophy prevent cardiac dysfunction despite increased wall stress. Circulation 105, 85–92 (2002).
Hill, J.A. et al. Targeted inhibition of calcineurin in pressure-overload cardiac hypertrophy. Preservation of systolic function. J. Biol. Chem. 277, 10251–10255 (2002).
Rapacciuolo, A. et al. Important role of endogenous norepinephrine and epinephrine in the development of in vivo pressure-overload cardiac hypertrophy. J. Am. Coll. Cardiol. 38, 876–882 (2001).
Levy, D., Garrison, R.J., Savage, D.D., Kannel, W.B. & Castelli, W.P. Prognostic implications of echocardiographically determined left ventricular mass in the Framingham Heart Study. N. Engl. J. Med. 322, 1561–1566 (1990).
Yasukawa, H. et al. The suppressor of cytokine signaling-1 (SOCS1) is a novel therapeutic target for enterovirus-induced cardiac injury. J. Clin. Invest. 111, 469–478 (2003).
Rothermel, B.A. et al. Myocyte-enriched calcineurin-interacting protein, MCIP1, inhibits cardiac hypertrophy in vivo . Proc. Natl Acad. Sci. USA 98, 3328–3333 (2001).
Bueno, O.F. et al. The dual-specificity phosphatase MKP-1 limits the cardiac hypertrophic response in vitro and in vivo . Circ. Res. 88, 88–96 (2001).
Crackower, M.A. et al. Regulation of myocardial contractility and cell size by distinct PI3K-PTEN signaling pathways. Cell 110, 737–749 (2002).
Milbrandt, J. A nerve growth factor-induced gene encodes a possible transcriptional regulatory factor. Science 238, 797–799 (1987).
Svaren, J. et al. NAB2, a corepressor of NGFI-A (Egr-1) and Krox20, is induced by proliferative and differentiative stimuli. Mol. Cell. Biol. 16, 3545–3553 (1996).
Gashler, A. & Sukhatme, V.P. Early growth response protein 1 (Egr-1): prototype of a zinc-finger family of transcription factors. Prog. Nucleic Acid Res. Mol. Biol. 50, 191–224 (1995).
Lee, S.L. et al. Luteinizing hormone deficiency and female infertility in mice lacking the transcription factor NGFI-A (Egr-1). Science 273, 1219–1221 (1996).
Neyses, L. et al. Induction of immediate-early genes by angiotensin II and endothelin-1 in adult rat cardiomyocytes. J. Hypertens. 11, 927–934 (1993).
Neyses, L., Nouskas, J. & Vetter, H. Inhibition of endothelin-1 induced myocardial protein synthesis by an antisense oligonucleotide against the early growth response gene-1. Biochem. Biophys. Res. Commun. 181, 22–27 (1991).
Sadoshima, J., Jahn, L., Takahashi, T., Kulik, T.J. & Izumo, S. Molecular characterization of the stretch-induced adaptation of cultured cardiac cells. An in vitro model of load-induced cardiac hypertrophy. J. Biol. Chem. 267, 10551–10560 (1992).
Saadane, N., Alpert, L. & Chalifour, L.E. Altered molecular response to adrenoreceptor-induced cardiac hypertrophy in Egr-1-deficient mice. Am. J. Physiol. Heart Circ. Physiol. 278, H796–H805 (2000).
Livak, K.J. & Schmittgen, T.D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408 (2001).
Buvoli, M. & Leinwand, L.A. Direct gene transfer into mouse heart. Methods Enzymol. 346, 134–142 (2002).
Acknowledgements
These studies were supported by grants from the Deutsche Forschungsgemeinschaft (Leibniz award), the Rudolf-Virchow-Center/DFG-Research Center for Experimental Biomedicine, the Bavarian Ministry of Technology, ProCorde and Sanofi-Aventis. We thank A. Simm (University of Halle) for advice on cardiomyocyte culture, C. Dienesch for help with aortic banding experiments and G. Thiel (University of Homburg) for the Egr-reporter gene construct and suggestions. We would like to thank P. Charnay for providing Egr1-deficient mice and L. Hein (University of Freiburg) for discussion of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Stegan Engelhardt, Monika Buitrago and Martin J. Lohse have filed a patent regarding the use of NAB/Egr in heart disease.
Supplementary information
Supplementary Table 1
Nab1 transgenic mice show normal cardiac function in vivo (PDF 16 kb)
Rights and permissions
About this article
Cite this article
Buitrago, M., Lorenz, K., Maass, A. et al. The transcriptional repressor Nab1 is a specific regulator of pathological cardiac hypertrophy. Nat Med 11, 837–844 (2005). https://doi.org/10.1038/nm1272
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1272
This article is cited by
-
Variable outcomes of human heart attack recapitulated in genetically diverse mice
npj Regenerative Medicine (2019)
-
Distinct submembrane localisation compartmentalises cardiac NPR1 and NPR2 signalling to cGMP
Nature Communications (2018)
-
Three TF Co-expression Modules Regulate Pressure-Overload Cardiac Hypertrophy in Male Mice
Scientific Reports (2017)
-
In vivo model with targeted cAMP biosensor reveals changes in receptor–microdomain communication in cardiac disease
Nature Communications (2015)
-
Nfat and miR-25 cooperate to reactivate the transcription factor Hand2 in heart failure
Nature Cell Biology (2013)