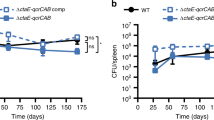
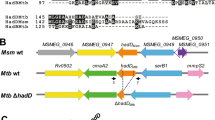
Abstract
Genes involved in fatty acid catabolism have undergone extensive duplication in the genus Mycobacterium, which includes the etiologic agents of leprosy and tuberculosis. Here, we show that prokaryotic- and eukaryotic-like isoforms of the glyoxylate cycle enzyme isocitrate lyase (ICL) are jointly required for fatty acid catabolism and virulence in Mycobacterium tuberculosis. Although deletion of icl1 or icl2, the genes that encode ICL1 and ICL2, respectively, had little effect on bacterial growth in macrophages and mice, deletion of both genes resulted in complete impairment of intracellular replication and rapid elimination from the lungs. The feasibility of targeting ICL1 and ICL2 for chemical inhibition was shown using a dual-specific ICL inhibitor, which blocked growth of M. tuberculosis on fatty acids and in macrophages. The absence of ICL orthologs in mammals should facilitate the development of glyoxylate cycle inhibitors as new drugs for the treatment of tuberculosis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Smith, H. Questions about the behaviour of bacterial pathogens in vivo . Phil. Trans. R. Soc. Lond. B 355, 551–564 (2000).
McDermott, W. Microbial persistence. Yale J. Biol. Med. 30, 257–291 (1958).
Charpentier, E. & Tuomanen, E. Mechanisms of antibiotic resistance and tolerance in Streptococcus pneumoniae . Microbes Infect. 2, 1855–1864 (2000).
Gomez, J.E. & McKinney, J.D. Mycobacterium tuberculosis persistence, latency, and drug tolerance. Tuberculosis (Edinb.) 84, 29–44 (2004).
Boshoff, H.I. & Barry, C.E., Tuberculosis - metabolism and respiration in the absence of growth. Nat. Rev. Microbiol. 3, 70–80 (2005).
Segal, W. & Bloch, H. Biochemical differentiation of Mycobacterium tuberculosis grown in vivo and in vitro . J. Bacteriol. 72, 132–141 (1956).
Sassetti, C.M. & Rubin, E.J. Genetic requirements for mycobacterial survival during infection. Proc. Natl Acad. Sci. USA 100, 12989–12994 (2003).
Keating, L.A. et al. The pyruvate requirement of some members of the Mycobacterium tuberculosis complex is due to an inactive pyruvate kinase: implications for in vivo growth. Mol. Microbiol. 56, 163–174 (2005).
Collins, D.M. et al. Production of avirulent mutants of Mycobacterium bovis with vaccine properties by the use of illegitimate recombination and screening of stationary-phase cultures. Microbiology 148, 3019–3027 (2002).
Liu, K. et al. pckA-deficient Mycobacterium bovis BCG shows attenuated virulence in mice and in macrophages. Microbiology 149, 1829–1835 (2003).
Kornberg, H.L. & Krebs, H.A. Synthesis of cell constituents from C2-units by a modified tricarboxylic acid cycle. Nature 179, 988–991 (1957).
Pertierra, A.G. & Cooper, R.A. Pyruvate formation during the catabolism of simple hexose sugars by Escherichia coli: studies with pyruvate kinase-negative mutants. J. Bacteriol. 129, 1208–1214 (1977).
Cole, S.T. et al. Deciphering the biology of Mycobacterium tuberculosis from the complete genome sequence. Nature 393, 537–544 (1998).
Fleischmann, R.D. et al. Whole-genome comparison of Mycobacterium tuberculosis clinical and laboratory strains. J. Bacteriol. 184, 5479–5490 (2002).
Höner zu Bentrup, K. et al. Characterization of activity and expression of isocitrate lyase in Mycobacterium avium and Mycobacterium tuberculosis . J. Bacteriol. 181, 7161–7167 (1999).
Timm, J. et al. Differential expression of iron-, carbon-, and oxygen-responsive mycobacterial genes in the lungs of chronically infected mice and tuberculosis patients. Proc. Natl Acad. Sci. USA 100, 14321–14326 (2003).
McKinney, J.D. et al. Persistence of Mycobacterium tuberculosis in macrophages and mice requires the glyoxylate shunt enzyme isocitrate lyase. Nature 406, 735–738 (2000).
Idnurm, A. & Howlett, B.J. Isocitrate lyase is essential for pathogenicity of the fungus Leptosphaeria maculans to canola (Brassica napus). Eukaryot. Cell 1, 719–724 (2002).
Lorenz, M.C. & Fink, G.R. The glyoxylate cycle is required for fungal virulence. Nature 412, 83–86 (2001).
Solomon, P.S. et al. Pathogenicity of Stagonospora nodorum requires malate synthase. Mol. Microbiol. 53, 1065–1073 (2004).
Vereecke, D. et al. Chromosomal locus that affects pathogenicity of Rhodococcus fascians . J. Bacteriol. 184, 1112–1120 (2002).
Wang, Z-Y. et al. The glyoxylate cycle is required for temporal regulation of virulence by the plant pathogenic fungus Magnaporthe grisea . Mol. Microbiol. 47, 1601–1612 (2003).
Vanni, P. et al. Comparative structure, function and regulation of isocitrate lyase, an important assimilatory enzyme. Comp. Biochem. Physiol. 95B, 431–458 (1970).
Sharma, V. et al. Structure of isocitrate lyase, a persistence factor of Mycobacterium tuberculosis . Nat. Struct. Biol. 7, 663–668 (2000).
Britton, K. et al. The crystal structure and active site location of isocitrate lyase from the fungus Aspergillus nidulans . Structure 8, 349–362 (2000).
Wendisch, V.F. et al. Quantitative determination of metabolic fluxes during coutilization of two carbon sources: comparative analyses with Corynebacterium glutamicum during growth on acetate and/or glucose. J. Bacteriol. 182, 3088–3096 (2000).
El-Sadr, W.M. et al. A review of efficacy studies of 6-month short-course therapy for tuberculosis among patients infected with human immunodeficiency virus: differences in study outcomes. Clin. Infect. Dis. 32, 623–632 (2001).
Flynn, J.L. & Chan, J. Immunology of tuberculosis. Annu. Rev. Immunol. 19, 93–129 (2001).
Russell, D.G. Mycobacterium tuberculosis: here today, and here tomorrow. Nat. Rev. Mol. Cell Biol. 2, 569–577 (2001).
Nishino, H. et al. Acute 3-nitropropionic acid intoxication induces striatal astrocytic cell death and dysfunction of the blood-brain barrier: involvement of dopamine toxicity. Neurosci. Res. 27, 343–355 (1997).
Chang, D-E. et al. Carbon nutrition of Escherichia coli in the mouse intestine. Proc. Natl Acad. Sci. USA 101, 7427–7432 (2004).
Chico-Calero, I. et al. Hpt, a bacterial homolog of the microsomal glucose- 6-phosphate translocase, mediates rapid intracellular proliferation in Listeria . Proc. Natl Acad. Sci. USA 99, 431–436 (2002).
Eriksson, S. et al. Unravelling the biology of macrophage infection by gene expression profiling of intracellular Salmonella enterica . Mol. Microbiol. 47, 103–118 (2003).
Schnappinger, D. et al. Transcriptional adaptation of Mycobacterium tuberculosis within macrophages: insights into the phagosomal environment. J. Exp. Med. 198, 693–704 (2003).
Ehrt, S. et al. Controlling gene expression in mycobacteria with anhydrotetracycline and Tet repressor. Nucleic Acids Res. 33, e21 (2005).
Blokpoel, M.C. et al. Tetracycline-inducible gene regulation in mycobacteria. Nucleic Acids Res. 33, e22 (2005).
Fischer, E. & Sauer, U. A novel metabolic cycle catalyzes glucose oxidation and anaplerosis in hungry Escherichia coli . J. Biol. Chem. 278, 46446–46451 (2003).
Peters-Wendisch, P. et al. Pyruvate carboxylase from Corynebacterium glutamicum: characterization, expression and inactivation of the pyc gene. Microbiology 144, 915–927 (1998).
Kondo, E. & Kanai, K. An attempt to cultivate mycobacteria in simple synthetic liquid medium containing lecithin-cholesterol liposomes. Jpn. J. Med. Sci. Biol. 29, 109–121 (1976).
Raynaud, C. et al. Phospholipases C are involved in the virulence of Mycobacterium tuberculosis . Mol. Microbiol. 45, 203–217 (2002).
Daniel, J. et al. Induction of a novel class of diacylglycerol acyltransferases and triacylglycerol accumulation in Mycobacterium tuberculosis as it goes into a dormancy-like state in culture. J. Bacteriol. 186, 5017–5030 (2004).
Claes, W.A. et al. Identification of two prpDBC gene clusters in Corynebacterium glutamicum and their involvement in propionate degradation via the 2-methylcitrate cycle. J. Bacteriol. 184, 2728–2739 (2002).
Tabuchi, T. & Uchiyama, H. Methylcitrate condensing and methylisocitrate cleaving enzymes; evidence for the pathway of oxidation of propionyl-CoA to pyruvate via C7-tricarboxylic acids. Agr. Biol. Chem. 39, 2035–2042 (1975).
Luttik, M.A. et al. The Saccharomyces cerevisiae ICL2 gene encodes a mitochondrial 2-methylisocitrate lyase involved in propionyl-coenzyme A metabolism. J. Bacteriol. 182, 7007–7013 (2000).
Ranes, M. et al. Functional analysis of pAL5000, a plasmid from Mycobacterium fortuitum: construction of a “Mini” Mycobacterium-Escherichia coli shuttle vector. J. Bacteriol. 172, 2793–2797 (1990).
Prentki, P. & Krisch, H.M. In vitro insertional mutagenesis with a selectable DNA fragment. Gene 29, 303–313 (1984).
Pavelka, M.S., Jr & Jacobs, W.R., Jr. Comparison of the construction of unmarked deletion mutations in Mycobacterium smegmatis, Mycobacterium bovis bacillus Calmette-Guerin, and Mycobacterium tuberculosis H37Rv by allelic exchange. J. Bacteriol. 181, 4780–4789 (1999).
Stover, C.K. et al. New use of BCG for recombinant vaccines. Nature 351, 456–460 (1991).
Acknowledgements
We thank N. Mirkovic and A. Sâli for construction of the ICL2 in silico model, B. Hanna for use of the BACTEC apparatus, J. Timm for assistance with real-time RT-PCR assays, M. Glickman for providing the cosmid containing icl2, S. Ehrt for providing the L-cell line and W.T. Chan and P. Giannakas for technical assistance. E.J.M.-E was supported by a Robert D. Watkins Graduate Fellowship from the American Society for Microbiology. J.D.M. acknowledges support from the Sequella Global Tuberculosis Foundation, the Ellison Medical Foundation, the Sinsheimer Fund and the Irma T. Hirschl Trust. This work was funded by grants (to J.D.M.) from GlaxoSmithKline and the US National Institutes of Health (AI46392).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Table 1
Enzymes, genes and genome designations. (PDF 207 kb)
Rights and permissions
About this article
Cite this article
Muñoz-Elías, E., McKinney, J. Mycobacterium tuberculosis isocitrate lyases 1 and 2 are jointly required for in vivo growth and virulence. Nat Med 11, 638–644 (2005). https://doi.org/10.1038/nm1252
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm1252
This article is cited by
-
Dimethyl itaconate is effective in host-directed antimicrobial responses against mycobacterial infections through multifaceted innate immune pathways
Cell & Bioscience (2023)
-
From immunology to artificial intelligence: revolutionizing latent tuberculosis infection diagnosis with machine learning
Military Medical Research (2023)
-
Identification of natural potent inhibitors against Mycobacterium tuberculosis isocitrate lyase: an in silico study
Molecular Diversity (2023)
-
The pathogenic mechanism of Mycobacterium tuberculosis: implication for new drug development
Molecular Biomedicine (2022)
-
Immunometabolic crosstalk during bacterial infection
Nature Microbiology (2022)