Abstract
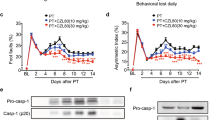
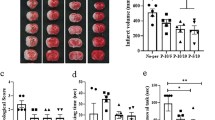
We show here that transient forebrain ischemia selectively elevates levels of neuronal apoptosis inhibitory protein (NAIP) in rat neurons that are resistant to the injurious effects of this treatment. This observation suggests that increasing NAIP levels may confer protection against ischemic cell death. Consistent with this proposal, we demonstrate that two other treatments that increase neuronal NAIP levels, systemic administration of the bacterial alkaloid K2S2a and intracerebral injection of an adenovirus vector capable of overexpressing NAIP in vivo, reduce ischemic damage in the rat hippocampus. Taken together, these findings suggest that NAIP may play a key role in conferring resistance to ischemic damage and that treatments that elevate neuronal levels of this antiapoptotic protein may have utility in the treatment of stroke.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Pulsinelli, W.A., Brierly, J.B. & Plum, F. Temporal profile of neuronal damage in a model of transient forebrain ischemia. Ann. Neural. 11, 491–498 (1982).
Smith, M.L., Auer, R.N. & Siesjo, B.K. The density and distribution of ischemic brain injury in the rat following 2–10 min of forebrain ischemia. Acta Neuropathol. (Berl) 64, 319–332 (1984).
MacManus, J.P., Buchan, A.M., Hill, I.E., Rasquinha, I. & Preston, E. Differences in DNA fragmentation following transient cerebral or decapitation ischemia in rats. J. Cerebral. Blood Flow Metab. 15, 728–737 (1995).
Nitatori, T. et al. Delayed neuronal death in the CA1 pyramidal cell layer of the gerbil hippocampus following transient ischemia is apoptosis. J. Neurosci. 15, 1001–1011 (1995).
Martinou, J.-C. et al. Overexpression of bcl-2 in transgenic mice protects neurons from naturally occurring cell death and experimental ischemia. Neuron 13, 1017–1030 (1994).
Lawrence, M.S., Ho, D.Y., Sun, G.H., Steinberg, C.K. & Sapolsky, R.M. Overexpression of Bcl-2 with herpes simplex virus vectors protects CNS neurons against neurological insults in vitro and in vivo. J. Neurosci 16, 486–496 (1996).
Krajewski, S. et al. Up regulation of bax levels in neurons following cerebral ischemia. J. Neurosd. 15, 6364–6376 (1995).
Roy, N. et al. The gene for neuronal apoptosis inhibitory protein is partially deleted in individuals with spinal muscular atrophy. Cell 80, 167–178 (1995).
Listen, P. et al. Suppression of apoptosis in mammalian cells by NAIP and a related family of IAP genes. Nature 379, 349–353 (1996).
Xu, D.G. et al. Distribution of NAIP-LI in the rat central nervous system. J. Comp. Neurol. 382, 247–259 (1997).
Smith-Swintosky, V.L. et. al. Bacterial alkaloids mitigate seizure-induced hippocampal damage and spatial memory deficits. Exp. Neurol. 141, 287–296 (1996).
Thilman, R., Xie, Y., Kleihues, P. & Kiessling, M. Persistent inhibition of protein synthesis precedes delayed neuronal death in postischemic gerbil hippocampus. Acta Neuropathal. 71, 88–93 (1986).
Maruno, M. & Yanagihara, T. Progressive loss of messenger RNA and delayed neuronal death following transient cerebral ischemia in gerbils. Neurosci. Lett. 115, 155–160 (1990).
Widmann, R. et al. [14C]Leucine incorporation into brain proteins in gerbils after transient ischemia: Relationship to selective vulnerability of hippocampus. J. Neurochem. 56, 789–796 (1991).
Glicksman, M.A. et al. K252a and staurosporine promote choline acetyltrans-ferase activity in rat spinal cord cultures. J. Neurochem. 61, 210–221 (1993).
Glicksman, M.A., Forbes, M.E., Prantner, J.E. & Neff, N.T. K-252a promotes survival and choline acetyltransferase activity in striatal and basal forebrain neuronal cultures. J. Neurochem. 64, 1502–1512 (1995).
Knusel, B. & Hefti, F. K252 compounds: Modulators of neurotrophin signal transduction. J. Neurochem. 59, 1987–1996 (1992).
Maroney, A.C. et al. K-252a induces tyrosine phosphorylatIon of the focal adhesion kinase and neurite outgrowth in human neuroblastoma SH-SYS cells. J. Neurochem. 64, 540–549 (1995).
Cheng, B., Barger, S.W. & Mattson, M.P. Staurosporine, K-252a and K-252b stabilize calcium homeostasis and promote survival of CNS neurons in the absence of glucose. J. Neurochem. 62, 1319–1329 (1994).
Paxinos, G. & Watson, C. The Rat Brain in Stereotaxic Coordinates, 2nd edn. (Academic Press, New York, 1986).
Fliss, H. & Gattinger, D. Apoptosis in ischemic and reperfused rat myocardium. Circ. Res. 79, 949–956 (1996).
Council on Animal Care. Guide to the Care and Use of Experimental Animals. (eds., Olfert, E. D., Cross, B. M. & McWilliam, A. A.) (Canadian Council on Animal Care, Ottawa, 1993).
Pulsinelli, W.A. & Buchan, A.M. The four-vessel occlusion rat model: Method for complete occlusion of vertebral arteries and control of collateral circulation. Stroke 19, 913–914 (1988).
Mullen, R.J., Buck, C.R. & Smith, A.M., NeuN, a neuronal specific nuclear protein in vertebrates. Development 116, 201–211 (1992).
Evan, G.I., Lewis, G.K., Ramsy, G. & Bishop, J.M. Isolation of monoclonal antibodies specific for human c-myc proto-oncogene product. Mol. Cell Biol. 5, 3610–3616 (1985).
Shu, S., Ju, G. & Fan, L. The glucose oxidase-DAB-nickel method in peroxidase histochemistry of the nervous system. Neurosci. Lett. 85, 169–171 (1988).
Doucet, J.-P., Murphy, B.J. & Tuana, B.S. Modification of a discontinuous and highly porous sodium dodecyl sulfate-polyacrylamide gel system for minigel electrophoresis. Anal. Biochem. 190, 209–211 (1990).
Towbin, H., Staehelin, T. & Gordon, T. Electrophoretic transfer of proteins from polyacrylamide gels to nitrocellulose sheets: procedures and some applications. Proc. Natl. Acad. Sci. USA 76, 435–4354 (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Xu, D., Crocker, S., Doucet, JP. et al. Elevation of neuronal expression of NAIP reduces ischemic damage in the rat hippocampus. Nat Med 3, 997–1004 (1997). https://doi.org/10.1038/nm0997-997
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0997-997
This article is cited by
-
Roles for NF-κB and Gene Targets of NF-κB in Synaptic Plasticity, Memory, and Navigation
Molecular Neurobiology (2014)
-
NLR functions beyond pathogen recognition
Nature Immunology (2011)
-
Lead Affects Apoptosis and Related Gene XIAP and Smac Expression in the Hippocampus of Developing Rats
Neurochemical Research (2010)
-
Mitochondrial membrane permeabilization in neuronal injury
Nature Reviews Neuroscience (2009)
-
Molecular chaperones as regulators of cell death
Oncogene (2008)