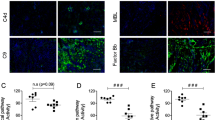
Abstract
The susceptibility of xenografts to hyperacute rejection is postulated to reflect in part failure of complement regulatory proteins (CRPs) to control activation of heterologous complement on graft endothelium. To test this concept, transgenic swine expressing the human CRP decay accelerating factor and CD59 were developed using a novel expression system involving transfer of the proteins from erythrocytes to endothelial cells. Hearts from transgenic swine transplanted into baboons had markedly less vascular injury and functioned for prolonged periods compared to hearts from nontransgenic swine. These results indicate that expression of human CRPs in xenogeneic organs may contribute to successful xenografting and suggest that intercellular protein transfer might be a useful approach for expression of heterologous proteins in endothelial cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Platt, J.L. et al. Transplantation of discordant xenografts: a review of progress. Immun. Today 11, 450–456 (1990).
Cooper, D.K.C., Preface. In Xenotransplantation: The Transplantation of Organs and Tissues Between Species, (eds Cooper, D.K.C., Kemp, E., Reemtsma, K. & White, D.J.G.) xi–xiv (Springer, New York, 1991).
White, D. & Wallwork, J. Xenografting: Probability, possibility, or pipe dream? Lancet 342, 879–880 (1993).
Platt, J.L. et al. Immunopathology of hyperacute xenograft rejection in a swine-to-primate model. Transplantation 52, 214–220 (1991).
Platt, J.L. et al. Release of heparan sulfate from endothelial cells: Implications for pathogenesis of hyperacute rejection. J. exp. Med. 171, 1363–1368 (1990).
Dalmasso, A.P. et al. Mechanism of complement activation in the hyperacute rejection of porcine organs transplanted into primate recipients. Am. J. Pathol. 140, 1157–1166 (1992).
Miyagawa, S. et al. The mechanism of discordant xenograft rejection. Transplantation 46, 825–830 (1988).
Leventhal, J.R. et al. Prolongation of cardiac xenograft survival by depletion of complement. Transplantation 55, 857–866 (1993).
Pruitt, S.K. et al. The effect of soluble complement receptor type 1 on hyperacute rejection of porcine xenografts. Transplantation 57, 363–370 (1994).
Shin, M.L., Hänsch, G., Hu, V.W. & Nicholson-Weller, A. Membrane factors responsible for homologous species restriction of complement-mediated lysis: Evidence for a factor other than DAF operating at the stage of C8 and C9. J. Immun. 136, 1777–1782 (1986).
Hourcade, D., Holers, V.M. & Atkinson, J.P. The regulators of complement activation (RCA) gene cluster. Adv. Immun. 45, 381–416 (1989).
Davies, A. et al. CD59, an LY-6-like protein expressed in human lymphoid cells, regulates the action of the complement membrane attack complex on homologous cells. J. exp. Med. 170, 637–654 (1989).
Rollins, S., Zhao, J., Ninomiya, H. & Sims, P. Inhibition of homologous complement by CD59 is mediated by a species-selective recognition conferred through binding to C8 within C5b-8 or C9 within C5b-9. J. Immun. 146, 2345–2351 (1991).
Kooyman, D. et al. Erythroid-specific expression of human CD59 and transfer to vascular endothelial cells. Transplants Proc. 26, 1241 (1994).
Kaplon, R.J. et al. Absence of hyperacute rejection in newborn pig-to-baboon cardiac xenografts. Transplantation 59, 1–6 (1994).
Platt, J.L. et al. An ELISA assay for xenoreactive natural antibodies. Transplantation 49, 1000–1001 (1990).
McCurry, K.R. et al. Transgenic expression of human complement regulatory proteins in mice results in diminished complement deposition during organ xenoperfusion. Transplantation (in the press).
Charreau, B. et al. Protection of rat endothelial cells from primate complement-mediated lysis by expression of human CD59 and/or decay-accelerating factor. Transplantation 58, 1222–1229 (1994).
Falk, R.J. et al. Neoantigen of the polymerized ninth component of complement. Characterization of a monoclonal antibody and immunohistochemical localization in renal disease. J. clin. Invest. 72, 560 (1983).
Platt, J.L. Hyperacute Xenograft Rejection (R.G. Landes Co., Austin 1995).
Townes, T.M., Lingrel, J.B., Chen, H.Y., Brinster, R.L. & Palmiter, R.D. Erythroid-specific expression of human b-globin genes in transgenic mice. EMBO J. 4, 1715–1723 (1985).
Logan, J.S. & Martin, M.J. Transgenic swine as a recombinant production system for human hemoglobin. Meth. Enzym. 231, 435–445 (1994).
Alexandre, G.P.J. et al. Splenectomy as a prerequisite for successful human ABO-incompatible renal transplantation. Transplants Proc. 27, 138–143 (1985).
Michler, R.E., McManus, R.P., Smith, C.R., Sadeghi, A.N. & Rose, E.A. Technique for primate heterotopic cardiac Xenotransplantation. J. med. Primatol. 14, 357–362 (1985).
Platt, J.L., LeBien, T.W. & Michael, A.F. Interstitial mononuclear cell populations in renal graft rejection: Identification by monoclonal antibodies in tissue sections. J. exp. Med. 155, 17–30 (1982).
Platt, J.L. & Michael, A.F. Retardation of fading and enhancement of lntensity of immunofluorescence by p-phenylenediamine. J. Histochem. Cy-
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
McCurry, K., Kooyman, D., Alvarado, C. et al. Human complement regulatory proteins protect swine-to-primate cardiac xenografts from humoral injury. Nat Med 1, 423–427 (1995). https://doi.org/10.1038/nm0595-423
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm0595-423
This article is cited by
-
Versatile cell ablation tools and their applications to study loss of cell functions
Cellular and Molecular Life Sciences (2019)
-
The current state of xenotransplantation
Journal of Applied Genetics (2015)
-
Kidney xenotransplantation
Kidney International (2014)
-
Generation of GGTA1 biallelic knockout pigs via zinc-finger nucleases and somatic cell nuclear transfer
Science China Life Sciences (2014)