Abstract
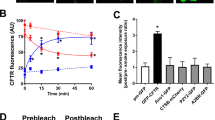
The most common mutation in cystic fibrosis, ΔF508, results in a cystic fibrosis transmembrane conductance regulator (CFTR) protein that is retained in the endoplasmic reticulum (ER). Retention is dependent upon chaperone proteins, many of which require Ca++ for optimal activity. Interfering with chaperone activity by depleting ER Ca++ stores might allow functional ΔF508-CFTR to reach the cell surface. We exposed several cystic fibrosis cell lines to the ER Ca++ pump inhibitor thapsigargin and evaluated surface expression of ΔF508-CFTR. Treatment released ER-retained ΔF508-CFTR to the plasma membrane, where it functioned effectively as a Cl− channel. Treatment with aerosolized calcium-pump inhibitors reversed the nasal epithelial potential defect observed in a mouse model of ΔF508-CFTR expression. Thus, ER calcium-pump inhibitors represent a potential target for correcting the cystic fibrosis defect.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Davis, P.B., Drumm, M. & Konstan, M.W. Cystic fibrosis. Am. J. Respir. Crit. Care Med. 154, 1229–1256 (1996).
Sheppard, D.N. & Welsh, M.J. Structure and function of the CFTR chloride channel. Physiol. Rev. 79, Suppl. S23–S45 (1999).
Thomas, P.J., Shenbagamurthi, P., Sondek, J., Hullihen, J.M. & Pedersen, P.L. The cystic fibrosis transmembrane conductance regulator: Effects of the most common cystic fibrosis-causing mutation on the secondary structure and the stability of a synthetic peptide. J. Biol. Chem. 267, 5727–5730 (1992).
Cheng, S.H. et al. Defective intracellular transport and processing of CFTR is the molecular basis of most cystic fibrosis. Cell 63, 827–834 (1990).
Ward, C.L., Omura, S. & Kopito, R.R. Degradation of CFTR by the ubiquitin-proteasome pathway. Cell 83, 122–127 (1995).
Egan, M.E., Schwiebert, E.M. & Guggino, W.B. Differential expression of outwardly rectifying chloride channels and CFTR induced by low temperature in CF airway epithelial cells. Am. J. Physiol. 268, C243–C251 (1995).
Rubenstein, R.C., Egan, M.E. & Zeitlin, P.L. In vitro pharmacologic restoration of CFTR-mediated chloride transport with sodium 4-phenylbutyrate in cystic fibrosis epithelial cells containing ΔF508-CFTR. J. Clin. Invest. 100, 2457–2465 (1997).
Hwang, T.C., Wang, F., Yang, I.C. & Reenstra, W.W. Genistein potentiates wild-type and ΔF508-CFTR channel activity. Am. J. Physiol. 273, C988–C998 (1997).
Dalemans, W. et al. Altered chloride ion channel kinetics associated with the ΔF508 cystic fibrosis mutation. Nature 354, 526–528 (1991).
Maitra, R., Shaw, C.M., Stanton, B.A. & Hamilton, J.W. Increased functional cell surface expression of CFTR and ΔF508-CFTR by the anthracycline doxorubicin. Am. J. Physiol. 280, C1031C1037 (2001).
Nigam, S.K. et al. A set of endoplasmic reticulum proteins possessing properties of molecular chaperones includes Ca(2+)-binding proteins and members of the thioredoxin superfamily. J. Biol. Chem. 269, 1744–1749 (1994).
Ellgaard, L., Molinari, M. & Helenius, A. Setting the standards: Quality control in the secretory pathway. Science 286, 1882–1888 (1999).
Montero, M. et al. Ca homeostasis in the endoplasmic reticulum: Coexistence of high and low [Ca] subcompartments in intact HeLa cells. J. Cell Biol. 139, 601–611 (1997).
Zeitlin, P.L. et al. A cystic fibrosis bronchial epithelial cell line: Immortalization by adeno-12-SV40 infection. Am. J. Resp. Cell. Mol. Biol. 4, 313–319 (1991).
Kunzelman, K. et al. An immortalized cystic fibrosis tracheal epithelial cell line homozygous for the DF508-CFTR mutation. Am. J. Resp. Cell. Mol. Biol. 8, 522–529 (1993).
Haws, C.M. et al. ΔF508-CFTR channels: Kinetics, activation by forskolin, and potentiation by xanthines. Am. J. Physiol. 270, C1544–C1555 (1996).
Schultz, B.D. et al. Glibenclamide blockade of CFTR chloride channels. Am. J. Physiol. 271, L192–L200 (1996).
Verkman, A.S. Development and biological applications of chloride-sensitive fluorescent indicators. Am. J. Physiol. 259, C375–C388 (1990).
Zhang, Z.R., Zeltwanger, S. & McCarty, N.A. Direct comparison of NPPB and DPC as probes of CFTR expressed in Xenopus oocytes. J. Membr. Biol. 175, 35–52 (2000).
Schoumacher, R.A. et al. A cystic fibrosis pancreatic adenocarcinoma cell line. Proc. Natl. Acad. Sci. USA 87, 4012–4016 (1990).
Grubb, B., Lazarowski, E., Knowles, M. & Boucher, R.C. Isobutylmethylxanthine fails to stimulate chloride secretion in cystic fibrosis airway epithelia. Am. J. Resp. Cell. Mol. Biol. 8, 454–460 (1993).
Choudhury, P., Liu, Y., Bick, R.J. & Sifers, R.N. Intracellular association between UDP-glucose:glycoprotein glucosyltransferase and an incompletely folded variant of α1-antitrypsin. J. Biol. Chem. 272, 13446–13451 (1997).
Shachar, I., Rabinovich, E., Kerem, A. & Bar-Nun, S. Thiol-reducing agents and calcium perturbants alter intracellular sorting of immunoglobulin M. J. Biol. Chem. 269, 27344–27350 (1994).
Tsien, R.Y. New calcium indicators and buffers with high selectivity against magnesium and protons: Design, synthesis and properties of prototype structures. Biochem. J. 19, 2396–2404 (1980).
Mason, M.J., Garcia-Rodriguez, C. & Grinstein, S. Coupling between intracellular Ca2+ stores and the Ca2+ permeability of the plasma membrane. Comparison of the effects of thapsigargin, 2, 5-di-(tert-butyl)-1, 4-hydroquinone, and cyclopiazonic acid in rat thymic lymphocytes. J. Biol. Chem. 266, 20856–20862 (1991).
Booth, C. & Koch, G.L.E. Perturbation of cellular calcium induces secretion of luminal ER proteins. Cell 59, 729–737 (1989).
Lee, A.S. The glucose-regulated proteins: Stress induction and clinical applications. TIBS 26, 504–10 (2001).
Llewellyn, D.H., Kendall, J.M., Sheikh, F.N. & Campbell, A.K. Induction of calreticulin expression in HeLa cells by depletion of the endoplasmic reticulum Ca++ store and inhibition of N-linked glycosylation. Biochem. J. 318, 555–560 (1996).
Zeiher, B.G. et al. A mouse model for the ΔF508 allele of cystic fibrosis. J. Clin. Invest. 96, 2051–2064 (1995).
Grubb, B.R., Vick, R.N. & Boucher, R.C. Hyperabsorption of Na+ and raised Ca(2+)-mediated Cl− secretion in nasal epithelia of CF mice. Am. J. Physiol. 266, C1478–C1483 (1994).
Zeitlin, P.L. Novel pharmacologic therapies for cystic fibrosis. J. Clin. Invest. 103, 447–452 (1999).
Rosenfeld, M.A. & Collins, F.S. Gene therapy for cystic fibrosis. Chest. 109, 241–52 (1996).
Sato, S., Ward, C.L., Krouse, M.E., Wine, J.J. & Kopito, R.R. Glycerol reverses the misfolding phenotype of the most common cystic fibrosis mutation. J. Biol. Chem. 271, 635–638 (1996).
Brown, C.R., Hong-Brown, L.Q. & Welch, W.J. Correcting temperature-sensitive protein folding defects. J. Clin. Invest. 99, 1432–1444 (1997).
Pind, S., Riordan, J.R. & Williams, D.B. Participation of the endoplasmic reticulum chaperone calnexin (p88, IP90) in the biogenesis of the cystic fibrosis transmembrane conductance regulator. J. Biol. Chem. 269, 12784–12788 (1994).
Meacham, G.C. et al. The Hdj-2/HSC70 chaperone pair facilitates early steps in CFTR biogenesis. EMBO J. 18, 1492–1505 (1999).
Lodish, H.F. & Kong, N. Perturbation of cellular calcium blocks exit of secretory proteins from the rough endoplasmic reticulum. J. Biol. Chem. 265, 10893–10899 (1990).
Wong, W.L., Brostrom, M.A., Kuznetsov, G., Gmitter-Yellen, D. & Brostrom, C.O. Inhibition of protein synthesis and early protein processing by thapsigargin in cultured cells. Biochem. J. 289, 71–79 (1993).
Hofer, A.M. & Machen, T.E. Technique for in situ measurement of calcium in intracellular inositol 1,4,5-triphosphate-sensitive stores using the fluorescent indicator mag-fura-2. Proc. Nat. Acad. Sci. USA 90, 2598–2602 (1993).
Christensen, S.B., Andersen, A., Poulsen, J.C.J. & Treiman, M. Derivatives of thapsigargin as probes of its binding site on endoplasmic reticulum Ca-ATPase: Stereoselectivity and important functional groups. FEBS Lett. 335, 345–348 (1993).
Tsien, R.Y., Pozzan, T. & Rink, T.J. Calcium homeostasis in intact lymphocytes: Cytoplasmic free calcium monitored with a new, intracellularly trapped fluorescent indicator. J. Cell Biol. 94, 325–334 (1982).
Chao, A., Kouyama, K., Heist, E., Dong, Y. & Gardner, P. Calcium- and CaMKII-dependent chloride secretion induced by the microsomal Ca-ATPase inhibitor 2,5-di-(tert-butyl)-1,4-hydroquinone in cystic fibrosis pancreatic epithelial cells. J. Clin. Invest. 96, 1794–1801 (1995).
Crawford, I. et al. Immunocytochemical localization of the cystic fibrosis gene product CFTR. Proc. Nat. Acad. Sci. USA 88, 9262–9266 (1991).
Gottardi, C.J. & Caplan, M.J. An ion transporting ATPase encodes multiple apical localization signals. J. Cell Biol. 121, 283–293 (1993).
Biemesderfer, D., Dekan, G., Aronson, P.S. & Farquhar, M.G. Assembly of distinctive coated pit and microvillar microdomains in the renal brush border. Am. J. Physiol. 262, F55–F67 (1992).
Acknowledgements
We thank M. Drumm for ΔF508-CFTR mice; W. Guggino, M. Blaustein, P. Aronson, J. Burger, G. Fyfe, G. Giebisch, G. Haddad, P. De Camilli, K. Bottomly, R. Lifton and members of the Caplan lab group for suggestions and readings of the manuscript; S.A. Mentone for help with electron microscopy; V. Rajendran and M.W. Nason for technical support; and M. Kashgarian for assistance in evaluating histopathologic specimens. This work was supported by the Alyward Family/Pitney Bowes Gift Fund, Panacea Pharmaceuticals (M.E.E. and M.J.C.) and by NIH grants DK53428 (to M.E.E.), DK50230 (to J.G.), HD32573 (to J.G.), GM42136 (to M.J.C.) and DK17433 (to J.G. and M.J.C.).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
These studies were supported to a small extent by a sponsored research agreement between Yale University and Panacea Pharmaceuticals, a small biotech firm that has licensed this technology from Yale University.
Rights and permissions
About this article
Cite this article
Egan, M., Glöckner-Pagel, J., Ambrose, C. et al. Calcium-pump inhibitors induce functional surface expression of ΔF508-CFTR protein in cystic fibrosis epithelial cells. Nat Med 8, 485–492 (2002). https://doi.org/10.1038/nm0502-485
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0502-485
This article is cited by
-
S-nitrosothiols signaling in cystic fibrosis airways
Journal of Biosciences (2021)
-
Disorders of FZ-CRD; insights towards FZ-CRD folding and therapeutic landscape
Molecular Medicine (2020)
-
Targeting DNAJB9, a novel ER luminal co-chaperone, to rescue ΔF508-CFTR
Scientific Reports (2019)
-
Small molecules as therapeutic agents for inborn errors of metabolism
Journal of Inherited Metabolic Disease (2017)
-
Nanoparticles that deliver triplex-forming peptide nucleic acid molecules correct F508del CFTR in airway epithelium
Nature Communications (2015)