Abstract
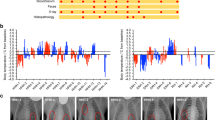
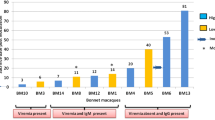
A nonhuman primate model of tuberculosis that closely resembles human disease is urgently needed. We have evaluated the Philippine cynomolgus monkey, Macaca fasicularis, as a model of TB. Cynomolgus monkeys challenged intratracheally with extremely high doses of Mycobacterium tuberculosis (105 or 104 CFU) developed an acute, rapidly progressive, highly fatal multilobar pneumonia. However, monkeys challenged with moderate or low doses of M. tuberculosis (≤103 CFU) developed a chronic, slowly progressive, localized form of pulmonary TB, akin to the disease in humans, that was frequently accompanied by such clinical syndromes as ocular tuberculosis, meningitis and tuberculous spondylitis. A significant proportion of monkeys challenged with 102 or 101 CFU contained the infection in a subclinical state. The Philippine cynomolgus monkey model is an excellent model of chronic TB and provides an opportunity to study subclinical and potentially latent disease in an animal model.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Good, R.C. Diseases in nonhuman primates, in. The Mycobacteria: A Sourcebook. (eds. Kubica, G.P. & Wayne, L.G.) 903—921 (Dekker, New York, 1984).
Schmidt, L.H. The problem of the rapid inactivator of isoniazid. Bull. Int. Union Tuberc. 32, 487–495 (1962).
Good, R.C. Simian tuberculosis: Immunologic aspects. Ann. NYAcad. Sci. 154, 200 (1968).
Schmidt, L.H. Improving existing methods of control of tuberculosis — A prime challenge to the experimentalist. The John Barnwell Lecture. Am. Rev. Respir. Dis. 105, 183–205 (1972).
Corcoran, K.D. & Jaax, G.P. An attempt to predict anergy in tuberculosis suspect cynomolgus monkeys. Lab. Anim. Sci. 41, 57–62 (1991).
Ward, G.S., Elwell, M.R., Tingpalapong, M. & Pomsdhit, J. Use of streptomycin and isoniazid during a tuberculosis epizootic in a rhesus and cynomolgus breeding colony. Lab. Anim. Sci. 35, 395–399 (1985).
Good, R.C. Tuberculosis and bacterial infection. in Nonhuman Primates and Medical Research, (ed. Bourne, G.H.). 39–60 (Academic Press, New York, 1978).
Tribe, G.W. & Welburn, A.E. Value of combining the erythrocyte sedimentation rate test with tuberculin testing in the control of tuberculosis in baboons. Lab. Anim. 10, 39–46 (1976).
Chaparas, S.D., Hodrick, S.R., Clark, R.G. & Garman, R. Comparison of the lymphocyte transformation test with the tuberculin test in rhesus monkeys and chimpanzees. Am. J. Vet. Res. 31, 1437–1441 (1970).
Chaparas, S.D., Good, R.C. & Janicki, B.W. Tuberculin-induced lymphocyte transformation and skin reactivity in monkeys vaccinated or not vaccinated with bacille Calmette-Guérin, then challenged with virulent Mycobacterium tuberculosis. Am. Rev. Respir. Dis. 112, 43–47 (1975).
Muscoplat, C.C. et al. Comparison of lymphocyte stimulation and tuberculin skin reactivity in Mycobacterium bovfs-infected Macaca mulatta. Am. J. Vet. Res. 36, 699–701 (1975).
Donahue, H.C. Ophthalmic experience in a tuberculosis sanitarium. Am. J. Ophthalmol. 64, 742–748 (1967).
Barrett-Connor, E. Tuberculous meningitis in adults. South. Med. J. 60, 1061–1067 (1967).
Farer, L.S., Lowell, L.M. & Meador, M.P. Extrapulmonary tuberculosis in the United States. Am. J. Epidemiol. 109, 205–217 (1979).
Auerbach, O. Tuberculous meningitis: Correlation of therapeutic results with the pathogenesis and pathologic changes: Pathologic changes in untreated and treated cases. Am. Rev. Tuberc. 64, 419–429 (1951).
Wurtz, R.B., Quadar, Z., Simon, D. & Langer, B. Cervical tuberculous vertebral osteomyelitis: Case report and discussion of the literature. Clin. Infect. Dis. 16, 806–808 (1993).
Schmidt, L.H. Some observations on the utility of simian tuberculosis in defining the therapeutic potential of isoniazid. Am. Rev. Tuberc. 74, 138–146 (1956).
Pal, P.G. & Horwitz, M.A. Immunization with extracellular proteins of Mycobacterium tuberculosis induces cell-mediated immune responses and substantial protective immunity in a guinea pig model of pulmonary tuberculosis. Infect. Immun. 60, 4781–4792 (1992).
Horwitz, M.A., Lee, B.W.E., Dillon, B.J. & Harth, G. Protective immunity against tuberculosis induced by vaccination with major extracellular proteins of Mycobacterium tuberculosis. Proc. Natl. Acad. Sci. USA 92, 1530–1534 (1995).
Boyum, A. Separation of white blood cells. Nature 204, 793 (1964).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Walsh, G., Tan, E., Dela Cruz, E. et al. The Philippine cynomolgus monkey (Macaca fasicularis) provides a new nonhuman primate model of tuberculosis that resembles human disease. Nat Med 2, 430–436 (1996). https://doi.org/10.1038/nm0496-430
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0496-430
This article is cited by
-
Full-length 16S rDNA sequencing based on Oxford Nanopore Technologies revealed the association between gut-pharyngeal microbiota and tuberculosis in cynomolgus macaques
Scientific Reports (2024)
-
CT and 18F-FDG PET abnormalities in contacts with recent tuberculosis infections but negative chest X-ray
Insights into Imaging (2022)
-
High-dose Mycobacterium tuberculosis aerosol challenge cannot overcome BCG-induced protection in Chinese origin cynomolgus macaques; implications of natural resistance for vaccine evaluation
Scientific Reports (2021)
-
Three decades of Philippine nonhuman primate studies: research gaps and opportunities for Philippine primatology
Primates (2021)
-
Detection of tuberculosis in cynomolgus macaques (Macaca fascicularis) using a supplementary Monkey Interferon Gamma Releasing Assay (mIGRA)
Scientific Reports (2020)