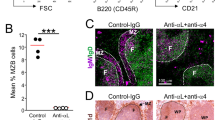
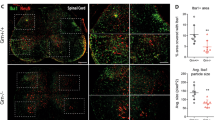
Abstract
New-variant Creutzfeldt–Jakob disease and scrapie are typically initiated by extracerebral exposure to the causative agent, and exhibit early prion replication in lymphoid organs1,2. In mouse scrapie, depletion of B-lymphocytes prevents neuropathogenesis after intraperitoneal inoculation3,4, probably due to impaired lymphotoxin-dependent maturation of follicular dendritic cells5 (FDCs), which are a major extracerebral prion reservoir6. FDCs trap immune complexes with Fc-γ receptors and C3d/C4b-opsonized antigens with CD21/CD35 complement receptors. We examined whether these mechanisms participate in peripheral prion pathogenesis. Depletion of circulating immunoglobulins or of individual Fc-γ receptors had no effect on scrapie pathogenesis if B-cell maturation was unaffected. However, mice deficient in C3, C1q, Bf/C2, combinations thereof7,8 or complement receptors9 were partially or fully protected against spongiform encephalopathy upon intraperitoneal exposure to limiting amounts of prions. Splenic accumulation of prion infectivity and PrPSc was delayed, indicating that activation of specific complement components is involved in the initial trapping of prions in lymphoreticular organs early after infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Hill, A.F., Zeidler, M., Ironside, J. & Collinge, J. Diagnosis of new variant Creutzfeldt-Jakob disease by tonsil biopsy. Lancet 349, 99 (1997).
Fraser, H. & Dickinson, A.G. Pathogenesis of scrapie in the mouse: the role of the spleen. Nature 226, 462–463 (1970).
Klein, M.A. et al. A crucial role for B cells in neuroinvasive scrapie. Nature 390, 687–690 (1997).
Klein, M.A. et al. PrP expression in B lymphocytes is not required for prion neuroinvasion. Nature Med. 4, 1429–1433 (1998).
Montrasio, F. et al. Impaired prion replication in spleens of mice lacking functional follicular dendritic cells. Science 288, 1257–1259 (2000).
Kitamoto, T., Muramoto, T., Mohri, S., Dohura, K. & Tateishi, J. Abnormal isoform of prion protein accumulates in follicular dendritic cells in mice with Creutzfeldt-Jakob disease. J.Virol. 65, 6292–6295 (1991).
Botto, M. et al. Homozygous C1q deficiency causes glomerulonephritis associated with multiple apoptotic bodies. Nature Genet. 19, 56–59 (1998).
Taylor, P.R. et al. A targeted disruption of the murine complement factor B gene resulting in loss of expression of three genes in close proximity, factor B, C2, and D17H6S45. J. Biol. Chem. 273, 1699–1704 (1998).
Molina, H. et al. Markedly impaired humoral immune response in mice deficient in complement receptors 1 and 2. Proc. Natl. Acad. Sci. USA 93, 3357–3361 (1996).
Kitamura, D., Roes, J., Kuhn, R. & Rajewsky, K. A B cell-deficient mouse by targeted disruption of the membrane exon of the immunoglobulin mu chain gene. Nature 350, 423–426 (1991).
Park, S.Y. et al. Resistance of Fc receptor-deficient mice to fatal glomerulonephritis. J. Clin. Invest. 102, 1229–1238 (1998).
Takai, T., Ono, M., Hikida, M., Ohmori, H. & Ravetch, J.V. Augmented humoral and anaphylactic responses in Fc gamma RII-deficient mice. Nature 379, 346–349 (1996).
Hazenbos, W.L. et al. Impaired IgG-dependent anaphylaxis and Arthus reaction in Fc γ xsRIII (CD16) deficient mice. Immunity 5, 181–188 (1996).
Szakal, A.K. & Hanna, M.G. Jr. The ultrastructure of antigen localization and viruslike particles in mouse spleen germinal centers. Exp. Mol. Pathol. 8, 75–89 (1968).
Carroll, M.C. CD21/CD35 in B cell activation. Semin. Immunol. 10, 279–286 (1998).
Yamakawa, M. & Imai, Y. Complement activation in the follicular light zone of human lymphoid tissues. Immunology 76, 378–384 (1992).
Ahearn, J.M. et al. Disruption of the Cr2 locus results in a reduction in B-1a cells and in an impaired B cell response to T-dependent antigen. Immunity 4, 251–262 (1996).
Büeler, H. et al. High prion and PrPSc levels but delayed onset of disease in scrapie-inoculated mice heterozygous for a disrupted PrP gene. Mol. Med. 1, 19–30 (1994).
Croix, D.A. et al. Antibody response to a T-dependent antigen requires B cell expression of complement receptors. J. Exp. Med. 183, 1857–1864 (1996).
Ebenbichler, C.F. et al. Human immunodeficiency virus type 1 activates the classical pathway of complement by direct C1 binding through specific sites in the transmembrane glycoprotein gp41. J. Exp. Med . 174, 1417–1424 (1991).
Klickstein, L.B., Barbashov, S.F., Liu, T., Jack, R.M. & Nicholson-Weller, A. Complement receptor type 1 (CR1, CD35) is a receptor for C1q. Immunity 7, 345–355 (1997).
Fischer, M.B. et al. Dependence of germinal center B cells on expression of CD21/CD35 for survival. Science 280, 582–585 (1998).
Wu, X. et al. Impaired affinity maturation in Cr2−/− mice is rescued by adjuvants without improvement in germinal center development. J. Immunol. 165, 3119–3127 (2000).
Hill, A.F. et al. Investigation of variant Creutzfeldt–Jakob disease and other human prion diseases with tonsil biopsy samples. Lancet 353, 183–189 (1999).
Nemazee, D.A. & Burki, K. Clonal deletion of B lymphocytes in a transgenic mouse bearing anti-MHC class I antibody genes. Nature 337, 562–566 (1989).
Sitia, R., Neuberger, M.S. & Milstein, C. Regulation of membrane IgM expression in secretory B cells: translational and post-translational events. EMBO J. 6, 3969–3977 (1987).
Seiler, P. et al. Enhanced virus clearance by early inducible lymphocytic choriomeningitis virus-neutralizing antibodies in immunoglobulin-transgenic mice. J. Virol. 72, 2253–2258 (1998).
Büeler, H.R. et al. Mice devoid of PrP are resistant to scrapie. Cell 73, 1339–1347 (1993).
Fischer, M. et al. Prion protein (PrP) with amino-proximal deletions restoring susceptibility of PrP knockout mice to scrapie. EMBO J. 15, 1255–1264 (1996).
Prusiner, S.B. et al. Measurement of the scrapie agent using an incubation time interval assay. Ann. Neurol. 11, 353–358 (1982).
Brandner, S. et al. Normal host prion protein necessary for scrapie-induced neurotoxicity. Nature 379, 339–343 (1996).
Acknowledgements
We thank M. Kosco for the FDC-M1 antibody; K. Rajewsky for μMT mice; C. Farquhar for the 1B3 antiserum; and C. Weissmann for critical reading of the manuscript. This work is supported by the Kanton of Zürich, the Bundesamt für Bildung und Wissenschaft, the Migros and Coop foundations, and by grants of the Swiss National Foundation to AA and RMZ.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Klein, M., Kaeser, P., Schwarz, P. et al. Complement facilitates early prion pathogenesis. Nat Med 7, 488–492 (2001). https://doi.org/10.1038/86567
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/86567
This article is cited by
-
Alternative complement pathway is activated in the brains of scrapie-infected rodents
Medical Microbiology and Immunology (2020)
-
Complement 3+-astrocytes are highly abundant in prion diseases, but their abolishment led to an accelerated disease course and early dysregulation of microglia
Acta Neuropathologica Communications (2019)
-
Unaltered intravenous prion disease pathogenesis in the temporary absence of marginal zone B cells
Scientific Reports (2019)
-
Analysis of RNA Expression Profiles Identifies Dysregulated Vesicle Trafficking Pathways in Creutzfeldt-Jakob Disease
Molecular Neurobiology (2019)
-
Molecular Alterations in the Cerebellum of Sporadic Creutzfeldt–Jakob Disease Subtypes with DJ-1 as a Key Regulator of Oxidative Stress
Molecular Neurobiology (2018)