Abstract
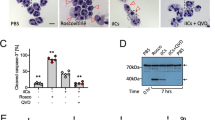
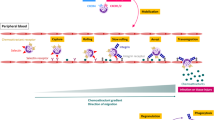
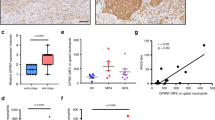
The β2 integrin LFA–1 (lymphocyte function associated antigen; CD11a/CD18) is the common ligand for the intercellular adhesion molecules1,2,3,4 (ICAMs). Integrins support cell function by providing co-stimulatory second signals that are a precondition for full cell activation first described for ICAM–1–binding to LFA–1 in lymphocytes3,4. Integrins can also serve to activate functions associated with distinct subunits of other integrins5. In addition to LFA–1, neutrophils express the β2 integrin Mac–1 (CD11b/CD18; CR3) that apparently contains multiple sites that bind invading microbes directly or through surface–fixed C3 (ref. 6), resulting in activation of the phagocyte function7,8,9,10,11. Expression of the LFA–1 counter–receptor ICAM–1 on endothelial cells occurs only at the site of inflammation12,13. Therefore, in neutrophils, ICAM–1 ligand binding could, as with lymphocytes, also play a part as a co–stimulatory signal to induce full phagocytotic function. We show that in neutrophils, the LFA–1 ligand interaction is the stimulatory signal to express full phagocytotic activation. This is best demonstrated by the rapid association of Streptococcus pyogenes with neutrophils, followed by ingestion, strong oxidative–burst induction and enhanced killing of these bacteria, which are well–known for their resistance to human neutrophil defense. These findings may contribute to the development of therapeutic strategies targeting the modulation of ICAM–1–leukocyte interaction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Hynes, R.O. Integrins: Versatility, modulation, and signalling in cell adhesion. Cell 69, 11–25 ( 1992).
Springer, T.A. Traffic signals for lymphocyte recirculation and leukocyte emigration: the multistep paradigm. Cell 76, 301– 314 (1994).
Marlin, S.D. & Springer, T.A. Purified intercellular adhesion molecule–1 (ICAM–1) is a ligand for lymphocyte function–associated antigen 1 (LFA–1). Cell 51, 813– 819 (1987).
van de Stolpe, A. & van der Saag, P.T. Intercellular adhesion molecule–1. J. Mol. Med. 74, 13–33 (1996).
Petruzzelli, L., Maduzia, L. & Springer, T.A. Activation of lymphocyte function–associated molecule–1 (CD11a/CD18) and Mac–1 (CD11b/CD18) mimicked by an antibody directed against CD18. J. Immunol. 155, 854–866 (1995).
Ross, G.D., Cain, J.A. & Lachmann, P.J. Membrane complement receptor type three (CR3) has lectin–like properties analogous to bovine conglutinin and functions as a receptor for zymosan and rabbit erythrocytes as well as a receptor for iC3b. J. Immunol. 134, 3307– 3315 (1985).
Graham, I.L., Gresham, H.D. & Brown, E.J. An immobile subset of plasma membrane CD11b/CD18 (Mac–1) is involved in phagocytosis of targets recognized by multiple receptors. J. Immunol. 142, 2352–2358 (1989).
Isberg, R.R. Discrimination between intracellular uptake and surface adhesion of bacterial pathogens. Science 252, 934– 938 (1991).
Tuomanen, E.I. in Leukocyte Adhesion: Basic and Clinical aspects. Proceedings of the 6th Novo Nordisk Foundation Symposium "Leukocyte Adhesion", Copenhagen, Denmark (eds. Gahmberg, C.G., Mandrup–Poulson, T. & Wogensen Bach, L.) 297–306 (Elsevier Science Publishers B. V., Amsterdam, 1992).
Nathan, C. et al. Cytokine–induced respiratory burst of human neutrophils: dependence on extracellular matrix proteins and CD11/CD18 integrins. J. Cell. Biol. 109, 1341–1349 (1989).
Berton, G., Yan, S. R., Fumagalli, L. & Lowell, C. A. Neutrophil activation by adhesion: mechanisms and pathophysiological implications. Int. J. Clin. Lab. Res. 26, 160– 177 (1996).
Ledebur, H.C. & Parks, T.P. Transcriptional regulation of the intercellular adhesion molecule–1 gene by inflammatory cytokines in human endothelial cells. Essential roles of a variant NF–kappa B site and p65 homodimers. J. Biol. Chem. 270, 933–943 (1995).
Cabanas, C. & Hogg, N. Ligand intercellular adhesion molecule 1 has a necessary role in activation of integrin lymphocyte function–associated molecule 1. Proc. Natl. Acad. Sci. USA 90, 5838–5842 (1993).
Leucocyte Typing V (eds. Schlossmann, S. F. et al.) 1548–1587 (Oxford University Press, Oxford, 1995).
Diamond, M.S., Garcia–Aguilar, J., Bickford, J.K., Corbi, A.L. & Springer, T.A. The I domain is a major recognition site on the leukocyte integrin Mac–1 (CD11b/CD18) for four distinct adhesion ligands. J. Cell. Biol. 120, 1031–1043 (1993).
Blackmore, T.K., Fischetti, V.A., Sadlon, H.M. & Gordon, D.L. M protein of group A streptococcus binds to the seventh short consensus repeat of human complement factor H. Infect. Immun. 66, 1427–1431 (1998).
Schmidt, K.H. et al. Susceptibility of chicken embryos to group A streptococci: correlation with fibrinogen binding. FEMS Immunol. Med. Microbiol. 7, 231–240 (1993).
Verhoef, J., Peterson, P.K. & Quie, P.G. Kinetics of staphylococcal opsonization, attachment, ingestion and killing by human polymorphnuclear leukocytes: a quantitative assay using 3H Thymidine labeled bacteria. J. Immunol. Methods 14, 303–311 (1977).
Mayer, C.L., Diamond, R.D. & Edwards, J.J.E. Recognition of binding sites on Candida albicans by monoclonal antibodies to human leucocyte antigens. Infect. Immun. 58, 3765–3769 (1990).
Rozdzinski, E. et al. Inhibition of leukocyte–endothelial cell interactions and inflammation by peptides from a bacterial adhesin which mimic coagulation factor X. J. Clin. Invest. 95, 1078– 1085 (1995).
Van Seventer, G.A., Shimizu, Y., Horgan, K.J. & Shaw, S. The LFA–1 ligand ICAM–1 provides an important costimulatory signal for T cell receptor–mediated activation of resting T cells. J. Immunol. 144, 4579–4586 (1990).
Podbielski, A., Schnitzler, N., Beyhs, P. & Boyle, M.D. M–related protein (Mrp) contributes to group A streptococcal resistance to phagocytosis by human granulocytes. Mol. Microbiol. 19, 429–441 (1996).
Kühnemund, O., Schumacher, F., Casaretto, M., Holm, S.E. & Lütticken, R. Estimation of type–specific anti–streptococcal M protein antibodies with biotinylated peptides in human sera. APMIS 106, 288– 292 (1998).
Baltimore, R.S., Kasper, D.L., Baker, C.J. & Goroff, D.K. Antigenic specificity of opsonophagocytic antibodies in rabbit anti–sera to Group B Streptococci. J. Immunol. 118, 673–678 (1977).
Salvadori, L.G., Blake, M.S., McCarty, M., Tai, J.Y. & Zabriskie, J.B. Group A Streptococcus–liposome ELISA antibody titers to Group A polysaccharide and opsonophagocytic capabilities of the antibodies. J. Infect. Dis. 171, 593– 600 (1994).
Acknowledgements
We thank R. Holland for technical assistance. We are grateful to O. Kühnemund for testing for anti–M antibodies and to C.R. Jr. Cooper for critical reading of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Schnitzler, N., Haase, G., Podbielski, A. et al. A co-stimulatory signal through ICAM-β2 integrin-binding potentiates neutrophil phagocytosis. Nat Med 5, 231–235 (1999). https://doi.org/10.1038/5597
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/5597
This article is cited by
-
Neutrophil recruitment limited by high-affinity bent β2 integrin binding ligand in cis
Nature Communications (2016)
-
Garenoxacin-induced increase of CD11b expression on human polymorphonuclear neutrophils does not affect phagocytosis and killing of Staphylococcus aureus
Journal of Infection and Chemotherapy (2011)
-
Streptococcal pyrogenic exotoxin B causes mitochondria damage to polymorphonuclear cells preventing phagocytosis of Group A streptococcus
Medical Microbiology and Immunology (2006)
-
Adhesion of human neutrophils to fibronectin is inhibited by comaruman, pectin of marsh cinquefoil comarum palustre L., and by its fragments
Biochemistry (Moscow) (2005)
-
Adhesion of human neutrophils to fibronectin is inhibited by comaruman, pectin of marsh cinquefoil Comarum palustre L., and by its fragments
Biochemistry (Moscow) (2005)