Abstract
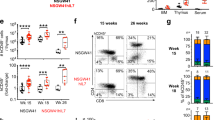
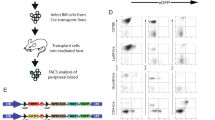
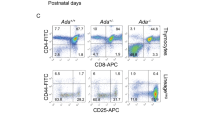
Janus kinase-3 (JAK3) deficiency has recently been identified as a cause of severe combined immunodeficiency (SCID) in humans. We used a mouse model of Jak3-deficient SCID to test a gene therapy approach for treatment of this disease. Transfer of a Jak3 retroviral vector to repopulat-ing hematopoietic stem cells resulted in increased numbers of T and B lymphocytes, reversal of hypogammaglobulinemia, restoration of T-cell activation upon stimulation with mitogens, and development of an antigen-specific immune response after immunization. Analysis for vector copy number in lymphoid and myeloid populations showed a large in vivo selective advantage for Jak3-expressing lymphoid cells. These results show that gene replacement is a feasible treatment strategy for this disease and that naturally occurring in vivo selection of corrected cells is an important advantage of this approach.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Hirschhorn, R. Overview of biochemical abnormalities and molecular genetics of adenosine deaminase deficiency. Pediatr. Res. 33, S35–S41 (1993).
Noguchi, M. et al. Interleukin-2 receptor γ chain mutation results in X-linked severe combined immunodeficiency in humans. Cell 73, 147 (1993).
Macchi, P. et al. Mutations of Jak-3 gene in patients with autosomal severe combined immunodeficiency (SCID). Nature 377, 65–68 (1995).
Russell, S. et al. Mutation of Jak3 in a patient with SCID: Essential role of Jak3 in lymphoid development. Science 270, 797–800 (1995).
Buckley, R. et al. Haploidentical bone marrow stem cell transplantation in human severe combined immunodeficiency. Semin. Hematol. 30, 92–104 (1993).
Dror, Y. et al. Immune reconstitution in severe combined immunodeficiency disease after lectin-treated, T-cell-depleted haplocompatible bone marrow transplantation. Blood 81, 2021–2030 (1993).
van Leeuwen, J. et al. Relationship between patterns of engraftment in peripheral blood and immune reconstitution after allogeneic bone marrow transplantation for (severe) combined immunodeficiency. Blood 84, 3936–3947 (1994).
Blaese, R. et al. T lymphocyte-directed gene therapy for ADA-SCID: Initial trial results after 4 years. Science 270, 475–480 (1995).
Bordignon, C. et al. Gene therapy in peripheral blood lymphocytes and bone marrow for ADA-immunodeficient patients. Science 270, 470–475 (1995).
Cao, X. et al. Defective lymphoid development in mice lacking expression of the common cytokine receptor γ chain. Immunity 2, 223–238 (1995).
Johnston, J. et al. Phosphorylation and activation of the Jak-3 Janus kinase in response to interleukin-2. Nature 370, 151–153 (1994).
Witthuhn, B. et al. Involvement of the Jak-3 Janus kinase in signalling by interleukins 2 and 4 in lymphoid and myeloid cells. Nature 370, 153–157 (1994).
Park, S. et al. Developmental defects of lymphoid cells in Jak3 kinase-deficient mice. Immunity 3, 771–782 (1995).
Nosaka, T. et al. Defective lymphoid development in mice lacking Jak3. Science 270, 800–802 (1995).
Thomis, D., Gurniak, C., Tivol, E., Sharpe, A. & Berg, L. Defects in B lymphocyte maturation and T lymphocyte activation in mice lacking Jak3. Science 270, 794–797 (1995).
Candotti, F. et al. Retroviral-mediated gene correction for X-linked severe combined immunodeficiency. Blood 87, 3097–3102 (1996).
Taylor, N. et al. Correction of interleukin-2 receptor function in X-SCID lymphoblastoid cells by retrovirally mediated transfer of the γc gene. Blood 87, 3103–3107 (1996).
Candotti, F. et al. In vitro correction of JAK3-deficient severe combined immunodeficiency by retroviral-mediated gene transduction. J. Exp. Med. 183, 2687–2692 (1996).
Allay, J., Dumenco, L., Koc, O., Liu, L. & Gerson, S. Retroviral transduction and expression of the human alkyltransferase cDNA provides nitrosourea resistance to hematopoietic cells. Blood 85, 3342–3351 (1995).
Laker, C. et al. Autocrine stimulation after transfer of the granulocyte/macrophage colony-stimulating factor gene and autonomous growth are distinct but interdependent steps in the oncogenic pathway. Proc. Natl. Acad. Sci. USA 84, 8458–8462 (1987).
Spencer, H., Sleep, S., Rehg, J., Blakley, R. & Sorrentino, B. A gene transfer strategy for making bone marrow cells resistant to trimetrexate. Blood 87, 2579–2587 (1996).
Buckley, R. et al. Human severe combined immunodeficiency: Genetic, phenotypic, and functional diversity in one hundred eight infants. J. Pediatr. 130, 378–387 (1997).
Orkin, S. & Motulsky, A. (study panel chairs). Report and recommendations of the panel to assess the NIH investment in research on gene therapy (http:// www.nih.gov/news/panelrep.html), access date 5/20/97.
Mardiney, M. III, et al. Enhanced host defense after gene transfer in the murine p47phox-deficient model of chronic granulomatous disease. Blood 89, 2268–2275 (1997).
Hirschhorn, R. et al. Spontaneous in vivo reversion to normal of an inherited mutation in a patient with adenosine deaminase deficiency. Nature Genet. 13, 290–295 (1996).
Stephan, V. et al. Atypical X-linked severe combined immunodeficiency due to possible spontaneous reversion of the genetic defect in T cells. N. Engl. J. Med. 335, 1563–1567 (1996).
Luo, H. et al. Mutation in the Jak kinase JH2 domain hyperactivates Drosophila and mammalian Jak-STAT pathways. Mol. Cell. Biol. 17, 1562–1571 (1997).
Luo, H., Hanratty, W. & Dearolf, C. An amino acid substitution in the Drosophila hop Tum-1 Jak kinase causes leukemia-like hematopoietic defects. EMBO J. 14, 1412–1420 (1995).
Smith, M., Duhe, R., Liu, Y. & Farrar, W. Microinjected cDNA encoding JAK2 protein-tyrosine kinase induces DNA synthesis in NIH 3T3 cells. FEBS Lett. 408, 327–330 (1997).
Miller, A. & Buttimore, C. Redesign of retrovirus packaging cell lines to avoid recombination leading to helper virus production. Mol. Cell. Biol. 6, 2895–2902 (1986).
Markowitz, D., Goff, S. & Bank, A. A safe packaging line for gene transfer: Separating viral genes on two different plasmids. J. Virol. 62, 1120–1124 (1988).
Bodine, D., McDonagh, K., Seidel, N. & Nienhuis, A. Survival and retrovirus infection of murine hematopoietic stem cells in vitro: Effects of 5-FU and method of infection. Exp. Hematol. 19, 206–212 (1991).
Bodine, D., Karlsson, S. & Nienhuis, A. Combination of interleukins 3 and 6 preserves stem cell function in culture and enhances retrovirus-mediated gene transfer into hematopoietic stem cells. Proc. Natl. Acad. Sci. USA 86, 8897–8901 (1989).
Sorrentino, B. et al. Selection of drug-resistant bone marrow cells in vivo after retroviral transfer of human MDR1. Science 257, 99–103 (1992).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bunting, K., Sangster, M., Ihle, J. et al. Restoration of lymphocyte function in Janus Kinase 3-deficient mice by retroviral-mediated gene transfer. Nat Med 4, 58–64 (1998). https://doi.org/10.1038/nm0198-058
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nm0198-058