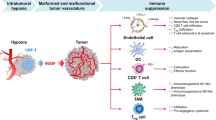
Abstract
Although angiogenesis inhibitors have provided substantial clinical benefit as cancer therapeutics, their use is limited by resistance to their therapeutic effects. While ample evidence indicates that such resistance can be influenced by the tumor microenvironment, the underlying mechanisms remain incompletely understood. Here, we have uncovered a paracrine signaling network between the adaptive and innate immune systems that is associated with resistance in multiple tumor models: lymphoma, lung and colon. Tumor-infiltrating T helper type 17 (TH17) cells and interleukin-17 (IL-17) induced the expression of granulocyte colony-stimulating factor (G-CSF) through nuclear factor κB (NF-κB) and extracellular-related kinase (ERK) signaling, leading to immature myeloid-cell mobilization and recruitment into the tumor microenvironment. The occurrence of TH17 cells and Bv8-positive granulocytes was also observed in clinical tumor specimens. Tumors resistant to treatment with antibodies to VEGF were rendered sensitive in IL-17 receptor (IL-17R)-knockout hosts deficient in TH17 effector function. Furthermore, pharmacological blockade of TH17 cell function sensitized resistant tumors to therapy with antibodies to VEGF. These findings indicate that IL-17 promotes tumor resistance to VEGF inhibition, suggesting that immunomodulatory strategies could improve the efficacy of anti-angiogenic therapy.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Ferrara, N. & Kerbel, R.S. Angiogenesis as a therapeutic target. Nature 438, 967–974 (2005).
Bergers, G. & Hanahan, D. Modes of resistance to anti-angiogenic therapy. Nat. Rev. Cancer 8, 592–603 (2008).
Ellis, L.M. & Hicklin, D.J. VEGF-targeted therapy: mechanisms of anti-tumour activity. Nat. Rev. Cancer 8, 579–591 (2008).
Tlsty, T.D. & Coussens, L.M. Tumor stroma and regulation of cancer development. Annu. Rev. Pathol. 1, 119–150 (2006).
Ebos, J.M., Lee, C.R. & Kerbel, R.S. Tumor and host-mediated pathways of resistance and disease progression in response to antiangiogenic therapy. Clin. Cancer Res. 15, 5020–5025 (2009).
Hanahan, D. & Coussens, L.M. Accessories to the crime: functions of cells recruited to the tumor microenvironment. Cancer Cell 21, 309–322 (2012).
Crawford, Y. & Ferrara, N. VEGF inhibition: insights from preclinical and clinical studies. Cell Tissue Res. 335, 261–269 (2009).
Maniati, E., Soper, R. & Hagemann, T. Up for mischief? IL-17/Th17 in the tumour microenvironment. Oncogene 29, 5653–5662 (2010).
Grivennikov, S.I. et al. Adenoma-linked barrier defects and microbial products drive IL-23/IL-17–mediated tumour growth. Nature 491, 254–258 (2012).
Fridman, W.H., Pages, F., Sautes-Fridman, C. & Galon, J. The immune contexture in human tumours: impact on clinical outcome. Nat. Rev. Cancer 12, 298–306 (2012).
Shojaei, F. et al. Tumor refractoriness to anti-VEGF treatment is mediated by CD11b+Gr1+ myeloid cells. Nat. Biotechnol. 25, 911–920 (2007).
Crawford, Y. et al. PDGF-C mediates the angiogenic and tumorigenic properties of fibroblasts associated with tumors refractory to anti-VEGF treatment. Cancer Cell 15, 21–34 (2009).
Shojaei, F. et al. G-CSF–initiated myeloid cell mobilization and angiogenesis mediate tumor refractoriness to anti-VEGF therapy in mouse models. Proc. Natl. Acad. Sci. USA 106, 6742–6747 (2009).
Iwakura, Y., Ishigame, H., Saijo, S. & Nakae, S. Functional specialization of interleukin-17 family members. Immunity 34, 149–162 (2011).
Korn, T., Bettelli, E., Oukka, M. & Kuchroo, V.K. IL-17 and Th17 Cells. Annu. Rev. Immunol. 27, 485–517 (2009).
Qu, X., Zhuang, G., Yu, L., Meng, G. & Ferrara, N. Induction of Bv8 expression by granulocyte colony-stimulating factor in CD11b+Gr1+ cells: key role of Stat3 signaling. J. Biol. Chem. 287, 19574–19584 (2012).
Shojaei, F. et al. Bv8 regulates myeloid-cell–dependent tumour angiogenesis. Nature 450, 825–831 (2007).
Hu, Y. et al. IL-17RC is required for IL-17A– and IL-17F–dependent signaling and the pathogenesis of experimental autoimmune encephalomyelitis. J. Immunol. 184, 4307–4316 (2010).
Metcalf, D. The molecular control of cell division, differentiation commitment and maturation in haemopoietic cells. Nature 339, 27–30 (1989).
Gabrilovich, D.I. & Nagaraj, S. Myeloid-derived suppressor cells as regulators of the immune system. Nat. Rev. Immunol. 9, 162–174 (2009).
Gabrilovich, D.I., Ostrand-Rosenberg, S. & Bronte, V. Coordinated regulation of myeloid cells by tumours. Nat. Rev. Immunol. 12, 253–268 (2012).
Shojaei, F. & Ferrara, N. Refractoriness to antivascular endothelial growth factor treatment: role of myeloid cells. Cancer Res. 68, 5501–5504 (2008).
Sinha, P. et al. Proinflammatory S100 proteins regulate the accumulation of myeloid-derived suppressor cells. J. Immunol. 181, 4666–4675 (2008).
Mazzieri, R. et al. Targeting the ANG2/TIE2 axis inhibits tumor growth and metastasis by impairing angiogenesis and disabling rebounds of proangiogenic myeloid cells. Cancer Cell 19, 512–526 (2011).
Erez, N., Truitt, M., Olson, P., Arron, S.T. & Hanahan, D. Cancer-associated fibroblasts are activated in incipient neoplasia to orchestrate tumor-promoting inflammation in an NF-κB–dependent manner. Cancer Cell 17, 135–147 (2010).
Bettelli, E., Korn, T., Oukka, M. & Kuchroo, V.K. Induction and effector functions of TH17 cells. Nature 453, 1051–1057 (2008).
Weaver, C.T., Hatton, R.D., Mangan, P.R. & Harrington, L.E. IL-17 family cytokines and the expanding diversity of effector T cell lineages. Annu. Rev. Immunol. 25, 821–852 (2007).
Dong, C. Diversification of T-helper-cell lineages: finding the family root of IL-17–producing cells. Nat. Rev. Immunol. 6, 329–333 (2006).
Tosolini, M. et al. Clinical impact of different classes of infiltrating T cytotoxic and helper cells (Th1, Th2, Treg, Th17) in patients with colorectal cancer. Cancer Res. 71, 1263–1271 (2011).
Liu, J. et al. IL-17 is associated with poor prognosis and promotes angiogenesis via stimulating VEGF production of cancer cells in colorectal carcinoma. Biochem. Biophys. Res. Commun. 407, 348–354 (2011).
Chen, X. et al. Increased IL-17–producing cells correlate with poor survival and lymphangiogenesis in NSCLC patients. Lung Cancer 69, 348–354 (2010).
Dirkx, A.E. et al. Anti-angiogenesis therapy can overcome endothelial cell anergy and promote leukocyte-endothelium interactions and infiltration in tumors. FASEB J. 20, 621–630 (2006).
Shrimali, R.K. et al. Antiangiogenic agents can increase lymphocyte infiltration into tumor and enhance the effectiveness of adoptive immunotherapy of cancer. Cancer Res. 70, 6171–6180 (2010).
Manning, E.A. et al. A vascular endothelial growth factor receptor-2 inhibitor enhances antitumor immunity through an immune-based mechanism. Clin. Cancer Res. 13, 3951–3959 (2007).
McGeachy, M.J. et al. TGF-β and IL-6 drive the production of IL-17 and IL-10 by T cells and restrain TH-17 cell-mediated pathology. Nat. Immunol. 8, 1390–1397 (2007).
Tartour, E. et al. Interleukin 17, a T-cell–derived cytokine, promotes tumorigenicity of human cervical tumors in nude mice. Cancer Res. 59, 3698–3704 (1999).
Numasaki, M. et al. Interleukin-17 promotes angiogenesis and tumor growth. Blood 101, 2620–2627 (2003).
Chung, A.S., Lee, J. & Ferrara, N. Targeting the tumour vasculature: insights from physiological angiogenesis. Nat. Rev. Cancer 10, 505–514 (2010).
He, D. et al. IL-17 promotes tumor development through the induction of tumor promoting microenvironments at tumor sites and myeloid-derived suppressor cells. J. Immunol. 184, 2281–2288 (2010).
Shojaei, F. et al. HGF/c-Met acts as an alternative angiogenic pathway in sunitinib-resistant tumors. Cancer Res. 70, 10090–10100 (2010).
Gerber, H.P. et al. VEGF regulates haematopoietic stem cell survival by an internal autocrine loop mechanism. Nature 417, 954–958 (2002).
Lee, S. et al. Autocrine VEGF signaling is required for vascular homeostasis. Cell 130, 691–703 (2007).
Purwar, R. et al. Robust tumor immunity to melanoma mediated by interleukin-9–producing T cells. Nat. Med. 18, 1248–1253 (2012).
Curd, L.M., Favors, S.E. & Gregg, R.K. Pro-tumour activity of interleukin-22 in HPAFII human pancreatic cancer cells. Clin. Exp. Immunol. 168, 192–199 (2012).
Liang, W.C. et al. Cross-species vascular endothelial growth factor (VEGF)-blocking antibodies completely inhibit the growth of human tumor xenografts and measure the contribution of stromal VEGF. J. Biol. Chem. 281, 951–961 (2006).
Zhong, C., Qu, X., Tan, M., Meng, Y.G. & Ferrara, N. Characterization and regulation of Bv8 in human blood cells. Clin. Cancer Res. 15, 2675–2684 (2009).
Brey, E.M. et al. Automated selection of DAB-labeled tissue for immunohistochemical quantification. J. Histochem. Cytochem. 51, 575–584 (2003).
Acknowledgements
We thank R. Khosla, I. Mellman, J. Kim, V. Phan, M. Yan and M. Junttila for helpful discussions and comments. We thank L. Komuves, C. Chalouni, M. Gonzales Edick and M. Sagolla for providing microscopy expertise and Genentech microarrays (in particular, Z. Modrusan), the Genentech animal and FACS facilities (in particular, A. Paler-Martinez) and D. Kallop and R. Weimer in animal imaging at Genentech. We thank N. Ota for help with Il17rc−/− mice.
Author information
Authors and Affiliations
Contributions
A.S.C. designed, planned and coordinated the experiments, analyzed the data and wrote the manuscript. N.F. coordinated and supervised the project and wrote the manuscript. A.S.C., X.W. and G.Z. performed the experiments. W.O. provided study design. Z.J. performed microarrays and bioinformatics analysis. H.N. and I.K. assisted with image acquisition and performed the analyses of clinical and preclinical samples, respectively. F.V.P. provided pathologist review and assessment of clinical samples. J.Z. and Y.G.M. performed mouse Bv8 ELISA. J.-M.V. and Y.G.M. performed intratumoral antibody ELISAs.
Ethics declarations
Competing interests
A.S., X.W., G.Z., H.N., I.K., J.Z., J.-M.V., Z.J., Y.G.M., F.V., W.O. and N.F. are present or formers employees of Genentech/Roche.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–8 (PDF 806 kb)
Rights and permissions
About this article
Cite this article
Chung, A., Wu, X., Zhuang, G. et al. An interleukin-17–mediated paracrine network promotes tumor resistance to anti-angiogenic therapy. Nat Med 19, 1114–1123 (2013). https://doi.org/10.1038/nm.3291
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nm.3291
This article is cited by
-
Abnormal generation of IL-17A represses tumor infiltration of stem-like exhausted CD8+ T cells to demote the antitumor immunity
BMC Medicine (2023)
-
Biology and therapeutic targeting of vascular endothelial growth factor A
Nature Reviews Molecular Cell Biology (2023)
-
Targeting angiogenesis in oncology, ophthalmology and beyond
Nature Reviews Drug Discovery (2023)
-
Design, construction and in vivo functional assessment of a hinge truncated sFLT01
Gene Therapy (2023)
-
Current status and future of anti-angiogenic drugs in lung cancer
Clinical and Experimental Medicine (2023)