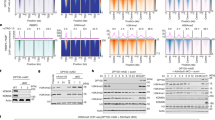
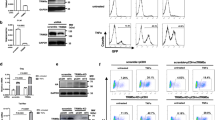
Abstract
PRDI-BF1, the human ortholog of mouse Blimp-1, is a DNA-binding protein involved in postinduction repression of interferon-β gene transcription in response to viral infection. PRDI-BF1 also has an essential function in driving terminal differentiation of B lymphocytes and therein silences multiple genes. Here we show PRDI-BF1 assembles silent chromatin over the interferon-β promoter in the osteosarcoma cell line U2OS through recruitment of the histone H3 lysine methyltransferase G9a. G9a is recruited only when in a complex with PRDI-BF1. G9a catalytic activity is required for the accumulation of methylated histone H3 and transcriptional silencing mediated by PRDI-BF1 in vivo. This establishes a mechanism for the recruitment of G9a, the main mammalian euchromatic methyltransferase, and defines nonembryonic targets of G9a.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Keller, A.D. & Maniatis, T. Identification and characterization of a novel repressor of β-interferon gene expression. Genes Dev. 5, 868–879 (1991).
Keller, A.D. & Maniatis, T. Only two of the five zinc fingers of the eukaryotic transcriptional repressor PRDI-BF1 are required for sequence-specific DNA binding. Mol. Cell. Biol. 12, 1940–1949 (1992).
Calame, K.L., Lin, K.I. & Tunyaplin, C. Regulatory mechanisms that determine the development and function of plasma cells. Annu. Rev. Immunol. 21, 205–230 (2003).
Angelin-Duclos, C., Cattoretti, G., Lin, K.I. & Calame, K. Commitment of B lymphocytes to a plasma cell fate is associated with Blimp-1 expression in vivo . J. Immunol. 165, 5462–5471 (2000).
Turner, C.A., Jr., Mack, D.H. & Davis, M.M. Blimp-1, a novel zinc finger-containing protein that can drive the maturation of B lymphocytes into immunoglobulin-secreting cells. Cell 77, 297–306 (1994).
Messika, E.J. et al. Differential effect of B lymphocyte-induced maturation protein (Blimp-1) expression on cell fate during B cell development. J. Exp. Med. 188, 515–525 (1998).
Lin, Y., Wong, K. & Calame, K. Repression of c-myc transcription by Blimp-1, an inducer of terminal B cell differentiation. Science 276, 596–599 (1997).
Shapiro-Shelef, M. et al. Blimp-1 is required for the formation of immunoglobulin secreting plasma cells and pre-plasma memory B cells. Immunity 19, 607–620 (2003).
Shaffer, A.L. et al. Blimp-1 orchestrates plasma cell differentiation by extinguishing the mature B cell gene expression program. Immunity 17, 51–62 (2002).
Lin, K., Angelin-Duclos, C., Kuo, T.C. & Calame, K. Blimp-1-dependent repression of Pax-5 is required for differentiation of B cells to immunoglobulin M-secreting plasma cells. Mol. Cell. Biol. 22, 4771–4780 (2002).
Piskurich, J.F. et al. BLIMP-I mediates extinction of major histocompatibility class II transactivator expression in plasma cells. Nat. Immunol. 1, 526–532 (2000).
Ghosh, N., Gyory, I., Wright, G., Wood, J. & Wright, K.L. Positive regulatory domain I binding factor 1 silences class II transactivator expression in multiple myeloma cells. J. Biol. Chem. 276, 15264–15268 (2001).
Funabiki, T., Kreider, B.L. & Ihle, J.N. The carboxyl domain of zinc fingers of the Evi-1 myeloid transforming gene binds a consensus sequence of GAAGATGAG. Oncogene 9, 1575–1581 (1994).
Xie, M., Shao, G., Buyse, I.M. & Huang, S. Transcriptional repression mediated by the PR domain zinc finger gene RIZ. J. Biol. Chem. 272, 26360–26366 (1997).
Abel, K.J. et al. Characterization of EZH1, a human homolog of Drosophila enhancer of zeste near BRCA1. Genomics 37, 161–171 (1996).
Jenuwein, T., Laible, G., Dorn, R. & Reuter, G. SET domain proteins modulate chromatin domains in eu- and heterochromatin. Cell. Mol. Life Sci. 54, 80–93 (1998).
Firestein, R., Cui, X., Huie, P. & Cleary, M.L. Set domain-dependent regulation of transcriptional silencing and growth control by SUV39H1, a mammalian ortholog of Drosophila Su(var)3-9. Mol. Cell. Biol. 20, 4900–4909 (2000).
Lachner, M. & Jenuwein, T. The many faces of histone lysine methylation. Curr. Opin. Cell Biol. 14, 286–298 (2002).
Kouzarides, T. Histone methylation in transcriptional control. Curr. Opin. Genet. Dev. 12, 198–209 (2002).
Fears, S. et al. Intergenic splicing of MDS1 and EVI1 occurs in normal tissues as well as in myeloid leukemia and produces a new member of the PR domain family. Proc. Natl. Acad. Sci. USA 93, 1642–1647 (1996).
Liu, L., Shao, G., Steele-Perkins, G. & Huang, S. The retinoblastoma interacting zinc finger gene RIZ produces a PR domain-lacking product through an internal promoter. J. Biol. Chem. 272, 2984–2991 (1997).
Gyory, I., Fejer, G., Ghosh, N., Seto, E. & Wright, K.L. Identification of a functionally impaired positive regulatory domain I binding factor 1 transcription repressor in myeloma cell lines. J. Immunol. 170, 3125–3133 (2003).
Fichelson, S. et al. Evi-1 expression in leukemic patients with rearrangements of the 3q25-q28 chromosomal region. Leukemia 6, 93–99 (1992).
He, L. et al. RIZ1, but not the alternative RIZ2 product of the same gene, is underexpressed in breast cancer, and forced RIZ1 expression causes G2-M cell cycle arrest and/or apoptosis. Cancer Res. 58, 4238–4244 (1998).
Jiang, G., Liu, L., Buyse, I.M., Simon, D. & Huang, S. Decreased RIZ1 expression but not RIZ2 in hepatoma and suppression of hepatoma tumorigenicity by RIZ1. Int. J. Cancer 83, 541–546 (1999).
Ren, B., Chee, K.J., Kim, T.H. & Maniatis, T. PRDI-BF1/Blimp-1 repression is mediated by corepressors of the Groucho family of proteins. Genes Dev. 13, 125–137 (1999).
Chen, G., Fernandez, J., Mische, S. & Courey, A.J. A functional interaction between the histone deacetylase Rpd3 and the corepressor groucho in Drosophila development. Genes Dev. 13, 2218–2230 (1999).
Yu, J., Angelin-Duclos, C., Greenwood, J., Liao, J. & Calame, K. Transcriptional repression by blimp-1 (PRDI-BF1) involves recruitment of histone deacetylase. Mol. Cell. Biol. 20, 2592–2603 (2000).
Tachibana, M., Sugimoto, K., Fukushima, T. & Shinkai, Y. Set domain-containing protein, G9a, is a novel lysine-preferring mammalian histone methyltransferase with hyperactivity and specific selectivity to lysines 9 and 27 of histone H3. J. Biol. Chem. 276, 25309–25317 (2001).
Rea, S. et al. Regulation of chromatin structure by site-specific histone H3 methyltransferases. Nature 406, 593–599 (2000).
Yang, L. et al. Molecular cloning of ESET, a novel histone H3-specific methyltransferase that interacts with ERG transcription factor. Oncogene 21, 148–152 (2002).
Schultz, D.C., Ayyanathan, K., Negorev, D., Maul, G.G. & Rauscher, F.J., 3rd. SETDB1: a novel KAP-1-associated histone H3, lysine 9-specific methyltransferase that contributes to HP1-mediated silencing of euchromatic genes by KRAB zinc-finger proteins. Genes Dev. 16, 919–932 (2002).
Ogawa, H., Ishiguro, K., Gaubatz, S., Livingston, D.M. & Nakatani, Y.A complex with chromatin modifiers that occupies E2F- and Myc-responsive genes in G0 cells. Science 296, 1132–1136 (2002).
Tachibana, M. et al. G9a histone methyltransferase plays a dominant role in euchromatic histone H3 lysine 9 methylation and is essential for early embryogenesis. Genes Dev. 16, 1779–1791 (2002).
Peters, A.H. et al. Loss of the Suv39h histone methyltransferases impairs mammalian heterochromatin and genome stability. Cell 107, 323–337 (2001).
Rice, J.C. et al. Histone methyltransferases direct different degrees of methylation to define distinct chromatin domains. Mol. Cell 12, 1591–1598 (2003).
Peters, A.H. et al. Partitioning and plasticity of repressive histone methylation states in mammalian chromatin. Mol. Cell 12, 1577–1589 (2003).
Jenuwein, T. & Allis, C.D. Translating the histone code. Science 293, 1074–1080 (2001).
Ayyanathan, K. et al. Regulated recruitment of HP1 to a euchromatic gene induces mitotically heritable, epigenetic gene silencing: a mammalian cell culture model of gene variegation. Genes Dev. 17, 1855–1869 (2003).
Lomvardas, S. & Thanos, D. Modifying gene expression programs by altering core promoter chromatin architecture. Cell 110, 261–271 (2002).
Zhang, X. et al. Structural basis for the product specificity of histone lysine methyltransferases. Mol. Cell 12, 177–185 (2003).
Zhang, X. et al. Structure of the Neurospora SET domain protein DIM-5, a histone H3 lysine methyltransferase. Cell 111, 117–127 (2002).
Wang, H. et al. Purification and functional characterization of a histone h3-lysine 4- specific methyltransferase. Mol. Cell 8, 1207–1217 (2001).
Nishioka, K. et al. Set9, a novel histone H3 methyltransferase that facilitates transcription by precluding histone tail modifications required for heterochromatin formation. Genes Dev. 16, 479–489 (2002).
Lachner, M., O'Sullivan, R.J. & Jenuwein, T. An epigenetic road map for histone lysine methylation. J. Cell Sci. 116, 2117–2124 (2003).
Huang, S. Histone methyltransferases, diet nutrients and tumour suppressors. Nat. Rev. Cancer 2, 469–476 (2002).
Turner, B.M. Memorable transcription. Nat. Cell Biol. 5, 390–393 (2003).
Vandel, L. et al. Transcriptional repression by the retinoblastoma protein through the recruitment of a histone methyltransferase. Mol. Cell. Biol. 21, 6484–6494 (2001).
Nielsen, S.J. et al. Rb targets histone H3 methylation and HP1 to promoters. Nature 412, 561–565 (2001).
Trimarchi, J.M. & Lees, J.A. Sibling rivalry in the E2F family. Nat. Rev. Mol. Cell. Biol. 3, 11–20 (2002).
Oberley, M.J., Inman, D.R. & Farnham, P.J. E2F6 negatively regulates BRCA1 in human cancer cells without methylation of histone H3 on lysine 9. J. Biol. Chem. 278, 42466–42476 (2003).
Yang, W.M., Tsai, S.C., Wen, Y.D., Fejer, G. & Seto, E. Functional domains of histone deacetylase-3. J. Biol. Chem. 277, 9447–9454 (2002).
Acknowledgements
Supported in part by the National Institutes of Health (CA80990), the American Cancer Society (ACS-IRG 032) and the Molecular Biology Core Facility and Molecular Imaging Core Facility at the H. Lee Moffitt Cancer Center and Research Institute.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Győry, I., Wu, J., Fejér, G. et al. PRDI-BF1 recruits the histone H3 methyltransferase G9a in transcriptional silencing. Nat Immunol 5, 299–308 (2004). https://doi.org/10.1038/ni1046
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni1046
This article is cited by
-
Loss of G9a does not phenocopy the requirement for Prdm12 in the development of the nociceptive neuron lineage
Neural Development (2024)
-
Establishment of H3K9-methylated heterochromatin and its functions in tissue differentiation and maintenance
Nature Reviews Molecular Cell Biology (2022)
-
Mucosal ribosomal stress-induced PRDM1 promotes chemoresistance via stemness regulation
Communications Biology (2021)
-
Transcriptional and epigenetic regulation of PD-1 expression
Cellular and Molecular Life Sciences (2021)
-
The BLIMP1—EZH2 nexus in a non-Hodgkin lymphoma
Oncogene (2020)