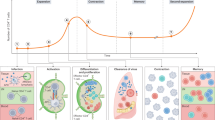
Abstract
The strength with which complexes of self peptide and major histocompatibility complex (MHC) proteins are recognized by the T cell antigen receptor (TCR) dictates the homeostasis of naive CD8+ T cells, but its effect on reactivity to foreign antigens is controversial. As expression of the negative regulator CD5 correlates with self-recognition, we studied CD5lo and CD5hi naive CD8+ T cells. Gene-expression characteristics suggested CD5hi cells were better poised for reactivity and differentiation than were CD5lo cells, and we found that the CD5hi pool also exhibited more efficient clonal recruitment and expansion, as well as enhanced reactivity to inflammatory cues, during the recognition of foreign antigen. However, the recognition of complexes of foreign peptide and MHC was similar for both subsets. Thus, CD8+ T cells with higher self-reactivity dominate the immune response to foreign antigens, with implications for T cell repertoire diversity and autoimmunity.
Similar content being viewed by others
Main
The nature of the interaction of the T cell antigen receptor (TCR) with complexes of foreign peptide and major histocompatibility complex (MHC) proteins (foreign peptide–MHC) determines the magnitude of the response and differentiation characteristics of antigen-specific T cells1,2,3,4. In addition, studies suggest that interactions of the TCR with self peptide–MHC also affect the response of naive T cells to foreign peptide–MHC5,6,7,8,9,10,11. Thymic positive selection and naive T cell homeostasis require low-affinity recognition of self peptide–MHC by the TCR12,13,14,15,16, but there is controversy about how such interactions affect the subsequent response to foreign peptide–MHC: published studies indicate that the recognition of self peptide–MHC enhances6 or diminishes7 the response to foreign antigens or selectively impairs sensitivity to low-affinity foreign ligands14. However, those reports investigated the effect of withdrawal of self peptide–MHC rather than studying how the degree of sensitivity to self peptide–MHC influences the T cell response to foreign peptide–MHC.
Homeostatic interactions of the TCR with self peptide–MHC are thought to be of very low affinity and to involve the recognition of multiple self peptides by an individual T cell clone, which would preclude direct assessment of the characteristics of self peptide–MHC recognition in the polyclonal T cell pool. However, differences in expression of the cell surface protein CD5 have proven to be a valuable surrogate for the strength of TCR–self peptide–MHC interactions14,17,18,19,20,21. CD5 expression on naive T cells can be used to accurately predict basal TCR signaling intensity and the ability of T cells to rapidly engage key TCR signaling pathways9,10,11, and it correlates with the ability of naive CD8+ T cells to respond to homeostatic cues22,23,24,25,26. However, the underlying basis for the distinct response characteristics of naive CD5lo and CD5hi populations is unclear, as is the effect of these differences on reactivity to foreign peptide–MHC.
Published studies have used CD5 expression on naive CD4+ T cells to correlate the strength of self peptide–MHC interactions with reactivity to foreign peptide–MHC9,10,11. In one study, analysis of mice with transgenic expression of the TCR suggested a direct correlation between the abundance of CD5 on the cell surface and the ability of the cell to bind tetramers of cognate foreign peptide–MHC9, which suggested that that the affinity of the TCR for self peptide–MHC can be used to predict its affinity for foreign peptide–MHC. Those authors observed more vigorous responses by CD5hi naive CD4+ T cells than by their CD5lo counterparts to foreign peptide–MHC. Another report failed to observe any correlation between CD5 expression and affinity of the TCR for foreign peptide–MHC ligands, however, and found that CD5lo T cell populations expanded more efficiently than their CD5hi counterparts during the primary response to foreign antigen10,11. Hence, whether and how CD5 expression can be used to predict the capacity with which naive T cells bind to and/or respond toward foreign peptide–MHC ligands is unclear.
Here we report that CD5hi and CD5lo naive CD8+ T cells differed in gene-expression characteristics and that the CD5hi population manifested improved clonal recruitment and expansion in response to foreign peptide–MHC. These differences in response did not correlate with the strength of the interaction of the TCR with foreign peptide–MHC, but CD5hi naive CD8+ T cells showed superior use of in vivo inflammatory signals. Our data suggest that predetermined heterogeneity among naive T cells dictates the capacity of their response to foreign antigens, with consequences for diversity of the functional T cell repertoire. Moreover, the finding that T cells with strong reactivity to self peptide–MHC dominated the response to foreign peptide–MHC has implications for the outgrowth of autoreactive T cells.
Results
Distinct phenotypes of CD5hi versus CD5lo CD8+ T cells
We first assessed phenotypic differences between CD5lo naive (CD44loCD122lo) CD8+ T cells and their CD5hi counterparts. In an extension of published work24,26,27, we found that the CD5hi cells were slightly larger, had higher expression of the adhesion molecule CD44 and modestly higher expression of the cytokine receptors IL-2Rβ (CD122) and IL-7Rα (CD127), but slightly lower expression of the TCR, coreceptor CD8 and adhesion molecule CD62L, than the CD5lo cells had (Fig. 1a and Supplementary Fig. 1a–c). The CD5hi naive CD8+ T cell population also showed elevated expression of T-bet and eomesodermin (transcription factors associated with the differentiation of activated CD8+ T cells28), and a subset of CD5hi cells expressed the chemokine receptor CXCR3 (Fig. 1a). The phenotypic characteristics of CD5hi naive CD8+ T cells had some similarities to those of memory CD8+ T cells. However, the frequency and phenotype of CD5hi naive CD8+ T cells was similar in wild-type mice and mice deficient in interleukin 15 (IL-15), which lack typical CD8+ memory T cells29 (Fig. 1b and Supplementary Fig. 1b,c). Hence, the CD5hi naive CD8+ T cell population neither derived from nor depended on memory-phenotype CD8+ T cells.
(a,b) Flow cytometry of cells pooled from the spleen and lymph nodes of wild-type mice (a) or Il15−/− mice (b), stained for CD44, T-bet, Eomes and CXCR3 (right), gated on naive (CD44loCD122lo) CD8+ T cells with the lowest 20% (lo) and highest 20% (hi) CD5 expression (gating at left). Gray shaded curves (right), cells with memory phenotype (CD44hiCD122hi). FSC, forward scatter (a measure of cell size). Numbers adjacent to outlined areas (far left) indicate percent CD44loCD122lo cells (bottom left) or CD44hiCD122hi cells (top right). (c,d) Flow cytometry of naive CD8+ T cells sorted by CD5 expression (as in a,b) and transferred together as congenically distinct populations into wild-type congenic recipient mice, then analyzed 4–8 weeks later without immunization of recipients. In d, right (compiled data), each symbol represents data from an individual mouse; small horizontal lines indicate the mean (± s.e.m.). Numbers adjacent to outlined areas (far left, d) indicate percent CD45.2hiCD45.1lo cells (bottom left) or CD45.2loCD45.1hi cells (top right). (e) Flow cytometry of CD4+CD25− and CD8+ T cells from Nur77GFP mice, gated on CD5 expression (as in a,b), followed by analysis of Nur77-GFP expression (gray curves as in a,b). (f) Flow cytometry of H-Y or OT-I CD8+ T cells and of polyclonal naive CD8+ T cells (gray), gated on the CD44lo population, assessing expression CD5 and Nur77-GFP. Data are representative of four (a,c,d), two (b,f) or three to four (e) independent experiments (n = 10 mice (a), 9 mice (c,d), 4 mice (b), 6–7 mice (f) or 6–11 mice (e)).
To determine whether the CD5hi and CD5lo populations were stable, we sorted polyclonal naive CD8+ T cells into CD5hi or CD5lo populations (which reflected the upper and lower 20% of distribution of CD5 expression, respectively) and transferred congenically distinct cell populations together into normal recipients. Both transferred populations maintained distinct CD5 expression and persisted for at least 8 weeks (Fig. 1c,d and data not shown), indicative of equivalent steady-state survival (similar to results obtained in studies of naive CD4+ T cells9). The majority of donor cells maintained a naive phenotype, although a fraction of the CD5hi cells converted to a CD44hi phenotype (Supplementary Fig. 1d), consistent with their enhanced response to homeostatic cues22,23,24,25,26.
Biochemical approaches indicate a correlation between CD5 abundance and the degree of basal TCR signaling5,9,11; however, such methods do not permit assessment of TCR signal strength in individual cells. Hence, we studied Nur77GFP transgenic reporter mice, in which expression of green fluorescent protein (GFP) from the immediate-early gene encoding the nuclear hormone receptor Nur77 (Nr4a1; called 'Nur77' here) provides a sensitive 'readout' of TCR signaling30. In CD8+ and CD4+ naive T cell subsets, CD5hi cells showed higher GFP expression than that of the CD5lo population (Fig. 1e), and this correlation held for Nur77GFP expression in CD8+ T cells from H-Y mice (which have transgenic expression of a male antigen–specific MHC class I–restricted TCR) and OT-I mice (which have transgenic expression of an ovalbumin-specific, MHC class I–restricted TCR), which reflect CD5lo populations and CD5hi populations, respectively)9,22,25 (Fig. 1f). Thus, CD5hi naive CD8+ T cells and their CD5lo counterparts were distinct, stable populations, with CD5hi cells displaying characteristics of cells that undergo more intense or frequent interactions of the TCR with self peptide–MHC.
Transcriptional profiles of CD5hi versus CD5lo CD8+ T cells
We next conducted gene-expression analysis of polyclonal CD5hi and CD5lo naive CD8+ T cells. In total, 57 genes showed a significant difference in expression of at least twofold in CD5hi naive CD8+ T cells relative to their expression in CD5lo naive CD8+ T cells (47 were upregulated and 10 were downregulated; Table 1). Among genes upregulated in CD5hi cells were those encoding the transcription factors Eomes, T-bet, Helios and Id3, many of which have key roles in the differentiation of activated T cells28, and molecules associated with the trafficking and adhesion of effector T cells (CXCR3, XCL1 and CD44). Conversely, the gene encoding the kinase Itk (which can serve as a negative regulator of T-bet31,32) was downregulated in CD5hi cells.
We further investigated the expression of XCL1, since it has been associated with efficient in vivo activation of CD8+ T cells (via enhancing the colocalization of T cells and dendritic cells (DCs))33. After brief in vitro stimulation of splenocytes, expression of XCL1 protein was biased to a subpopulation of CD5hi naive CD8+ T cells (Fig. 2a,b and Supplementary Fig. 2a). Expression of CXCR3 and T-bet also marked a subset of CD5hi naive CD8+ T cells (Fig. 2b and Supplementary Fig. 2a). However, although memory-phenotype (CD44hi) CD8+ T cells typically coexpressed these proteins, there was little coordinated expression of these in the naive CD5hi pool (Supplementary Fig. 2b,c) We further investigated the expression of XCL1, since it has been associated with efficient in vivo activation of CD8+ T cells (via enhancing the colocalization of T cells and dendritic cells (DCs))33. After brief in vitro stimulation of splenocytes, expression of XCL1 protein was biased to a subpopulation of CD5hi naive CD8+ T cells (Fig. 2a,b and Supplementary Fig. 2a). Expression of CXCR3 and T-bet also marked a subset of CD5hi naive CD8+ T cells (Fig. 2b and Supplementary Fig. 2a). However, although memory-phenotype (CD44hi) CD8+ T cells typically coexpressed these proteins, there was little coordinated expression of these in the naive CD5hi pool (Supplementary Fig. 2b,c), which indicated considerable heterogeneity within the CD5hi naive CD8+ T cell population.
(a) Expression of XCL1 (right) by a memory phenotype (CD44hi) CD8+ T cell population (far right) and by naive (CD44lo) CD8+ T cells among splenocytes left unstimulated (US) or stimulated (PMA + iono) with the phorbol ester PMA and ionomycin in the presence of brefeldin A and then stained intracellularly for XCL1, gated on CD5 expression (as in Fig. 1a,b). Numbers adjacent to outlined areas (far left) indicate percent CD44hiCD8hi cells (top) or CD44loCD8hi cells (bottom); numbers above bracketed lines (right) indicate percent XCL1+ cells. (b) Frequency of cells of various subsets (key) expressing XCL1, assessed following activation as in a, or expressing CXCR3 or T-bet, assessed in unstimulated cells pooled from C57BL/6 (B6) mice and T-bet reporter mice (in which GFP is expressed under control of the gene encoding T-bet)50. (c) Ratio of the expression of genes in clusters II, III and X by CD5lo cells relative to their expression in CD5hi cells, among CD44lo CD8+ T cells gated on CD5 expression (as in Fig. 1a,b); gene-expression data were aligned with gene clusters that are associated with stages of the in vivo CD8+ T cell response34. A ratio of <1 (left of the vertical dashed line) indicates 'preferential' expression in the CD5hi population. *P < 0.001 (unpaired t-test). Data are representative of four experiments with nine mice (a) or are from four experiments (n = 9 mice) for XCL1, three experiments (n = 7 mice) for T-bet or five experiments (n = 13 mice) for CXCR3 (b; error bars, s.e.m.) or three independent experiments (c).
Most differences between the CD5hi and CD5lo naive CD8+ T cell populations in their expression of individual genes were subtle (Table 1); hence, we explored whether there was enrichment in the expression of specific gene clusters. For a focused comparison, we used a χ2 test to compare differences in gene transcription by CD5hi and CD5lo cells with a database generated by Immunological Genome Project Consortium, which has comprehensively defined patterns of gene expression after the activation and differentiation of CD8+ T cells34. In that work, temporal analysis of gene expression over the course of the immune response allowed the characterization of ten clusters of correlated gene expression (clusters I–X)34. We investigated how expression of genes in these clusters was regulated in the CD5hi and CD5lo naive CD8+ T cell subsets. This analysis revealed that the CD5hi population expressed a significantly higher proportion of genes that characterize two early stages of the CD8+ T cell response and are associated with preparation for cell cycle (cluster II) and active cell cycle and division (cluster III) (Fig. 2c and Table 2). We also found a more moderate (but still highly significant) correlation of genes 'preferentially' expressed in the CD5hi population with cluster X, which defines genes expressed at late effector and memory stages (Fig. 2c and Table 2). Together these data suggested the CD5hi population would be better poised for initial activation than the CD5lo population.
Enhanced expansion of the CD5hi population after infection
We next directly investigated whether CD5hi and CD5lo naive CD8+ T cells differed in their primary immune response to foreign antigen. In initial studies, we assayed polyclonal CD8+ T cells specific for the H-2Kb-restricted epitope derived from amino acids 20–27 of the vaccinia virus glycoprotein B8R (peptide called simply 'B8R' here), which are present at a frequency of ∼1 cell per 1 × 104 to 2 × 104 CD8+ T cells in unimmunized C57BL/6 mice35. We sorted naive CD44lo CD8+ T cells by flow cytometry into congenically distinct CD5lo and CD5hi populations and transferred ∼1.5 × 106 cells of each population together into congenic wild-type recipient mice that we subsequently infected with LM-B8R, a recombinant attenuated Listeria monocytogenes strain that expresses the B8R epitope and the H-2Kb-restricted peptide of ovalbumin amino acids 257–264 (called simply 'OVA' here). With the assumption of ∼20% engraftment following adoptive transfer, this would be expected to seed ∼20 B8R–H-2Kb–specific cells from each donor. At day 7 following infection, we used staining with peptide-MHC (pMHC) tetramers to identify responsive CD5hi and CD5lo donor cells and determined the ratio (Fig. 3a) and absolute number (Fig. 3b) of each population. In most cases, the CD5hi donor population dominated the response, on average accounting for ∼95% of the B8R–H-2Kb–specific population (Fig. 3a,b), although occasionally progeny of the CD5lo donor cells were more frequent (Fig. 3a,b). Tetramer binding may fail to identify all functionally responsive cells, but we obtained similar results by using peptide-induced production of interferon-γ to identify antigen-specific T cells (Supplementary Fig. 3a,b). At the memory phase following priming and also during a recall response, the progeny of CD5hi donor cells maintained dominance over those from the CD5lo pool (Fig. 3a). The finding that this skewing was not exacerbated during the recall response indicated that memory cells generated from CD5lo and CD5hi cells had similar population-reexpansion potential.
(a) Ratio of donor CD5hi B8R–H-2Kb–specific cells to their CD5lo counterparts in CD45.1+CD45.2+ host mice given transfer of 1.25 × 106 to 1.5 × 106 each of congenically distinct CD5lo and CD5hi cells (generated from CD44lo CD8+ T cells gated on CD5 expression as in Fig. 1a,b) and infected the next day with LM-B8R, followed by isolation of B8R–H-2Kb–specific cells from the spleen by B8R–H-2Kb tetramer enrichment and analysis at day 7 or 30 after primary infection or 5 d after rechallenge of recipient mice (at >30 d after the primary infection) with virulent LM-B8R (Day 5 recall). Filled symbols indicate mice for which a B8R–H-2Kb–specific response was not detected for CD5lo donor cells and the ratio was arbitrarily set as 102. (b) Total donor cells at day 7 in mice as in a. Each symbol represents an individual mouse; ‡ indicates an outlier (exclusion of this group did not alter the significance of these results). (c) Ratio of bulk LM-B8R-specific donor CD5hi CD8+ T cells to their CD5lo counterparts in mice as in a. (d,e) B8R–H-2Kb–specific responses (d) and bulk LM-B8R-specific responses (e) in congenic recipients given a mixture of sorted CD8+ CD44lo cells plus congenically distinct CD44lo CD5lo or CD44lo CD5hi populations (1.25 × 106 to 1.5 × 106 cells of each population), followed by infection of recipients with LM-B8R 1 d later and analysis 7 d after infection. (f) Ratio of cells derived from CXCR3hi donor cells to their CXCR3lo counterparts in the B8R–H-2Kb–specific and bulk LM-B8R-responsive populations (left), and quantification of B8R–H-2Kb–specific CXCR3hi and CXCR3lo donor cells (right), in CD45.1+CD45.2+ recipients given 1 × 106 to 2 × 106 congenically distinct CXCR3lo or CXCR3hi CD5hi cells (CD5hi naive CD8+ T cells (CD44lo cells in the top 20% for CD5 expression) sorted as cells with the lowest or highest 30% of CXCR3 expression), followed by infection of recipients with LM-B8R 1 d later and isolation of B8R–H-2Kb–specific cells from the spleen 7 d after infection by tetramer enrichment. (g) Quantification of B8R–H-2Kb–specific donor cells in the spleen of congenic recipients given congenically distinct combinations of wild-type (WT) and CD25 (IL-2Rα)-deficient (KO) CD44lo CD5lo and CD5hi cells sorted from chimeras reconstituted with congenic wild-type and CD25 (IL-2Rα)-deficient bone marrow, followed by infection of recipients with LM-B8R 1 d later and analysis at day 7 following infection. Each symbol represents an individual mouse; small horizontal lines indicate the geometric mean (a,c,f) or mean (d,e,g) (± 95% confidence interval in a,c,g or ± s.e.m. in d,e)). NS, not significant; *P < 0.05, **P < 0.01 and ***P < 0.001 (unpaired t-test). Data were pooled from four experiments at day 7 (n = 11 mice), three experiments at day 30 (n = 9 mice) or two experiments for day 5 recall (n = 6 mice) (a–c), three experiments (d,f; n = 11–12 mice (d) or n = 10 mice (f)) or three independent experiments (n = 6 mice) (g) or are from two experiments (n = 8 mice) (e).
The dominance of CD5hi naive CD8+ T cell responses was not unique to B8R–H-2Kb–specific T cells or to infection with L. monocytogenes: The OVA–H-2Kb–specific response induced by infection with LM-B8R, as well as the response specific to the H-2Dd-restricted epitope from lymphocytic choriomeningitis virus (LCMV) glycoprotein amino acids 33–41 (gp33), induced by infection with LCMV, were also biased to the CD5hi donor cells (Supplementary Fig. 3c,d). Beyond individual antigen specificities, the bulk pathogen-specific response, identified as donor CD8+ T cells that had acquired an antigen-experienced CD44hiCD8loCD11ahi phenotype, also showed an advantage for the CD5hi donor pool, albeit less pronounced than that observed for individual pMHC-specific responses (Fig. 3c and Supplementary Fig. 3e).
The 'preferential' expansion of the CD5hi donor population did not reflect a greater intrinsic capacity of these cells for TCR-induced proliferation, as CD5lo and CD5hi naive CD8+ T cells proliferated similarly upon in vitro stimulation with antibody to the invariant signaling protein CD3 (anti-CD3) plus antibody to the coreceptor CD28 (anti-CD28) (Supplementary Fig. 3f), consistent with published studies9,20,24. We also noted the enhanced CD5hi T cell B8R–H-2Kb–specific response in studies of recipient mice deficient in recombination-activating gene 1, which are deficient in mature B cells and T cells (Supplementary Fig. 3g); this ruled out the possibility of a required contribution from host T lymphocytes or B lymphocytes.
Since naive CD5hi cells have intermediate expression of CD44, it was formally possible that some memory-phenotype T cells had contaminated the CD5hi donor population. However, when we sorted CD5hi and CD5lo donor populations to have equally low CD44 expression, the CD5hi donor population still dominated the response to LM-B8R (Supplementary Fig. 3h), which argued against the possibility of contamination by memory cells in our studies. The differences in the responses of CD5hi and CD5lo naive CD8+ T cells suggested that reactivity improved proportionally to increasing CD5 expression. To explore this, we measured the response of CD5lo and CD5hi populations to infection, relative to that of the total naive CD8+ T cell pool (containing the full spectrum of CD5 expression). The CD5lo population expanded less than bulk naive CD8+ T cells did, while the CD5hi cells proliferated more (Fig. 3d,e), which suggested that CD5lo and CD5hi populations represent the extremes of a continuum in reactivity to foreign antigen.
Given that the CD5hi population was heterogeneous (Fig. 2b and Supplementary Fig. 2c), it was possible that a small subset within this pool was responsible for their superior antigen-specific responses. We investigated this hypothesis by subdividing the naive CD5hi population on the basis of CXCR3 expression, since this chemokine receptor has been associated with enhanced detection of antigen in vivo by memory CD8+ T cells36,37. We sorted congenic populations of naive CD5hi cells into CXCR3lo and CXCR3hi populations and assessed their response toward infection with LM-B8R using the co-transfer model described above. We observed that expansion of the CXCR3hi CD5hi population was significantly greater than that of the CXCR3lo CD5hi subset, in both the B8R–H-2Kb–specific responder population and bulk CD44hi responder population (Fig. 3f). However, these differences were of lower magnitude than those between the CD5hi and CD5lo populations (Fig. 3a,b,f), which challenged the hypothesis that a small subset of CD5hi cells accounted for all the enhanced reactivity of this population.
In addition to being influenced by TCR signals, CD8+ T cell responses are strongly influenced by cytokines. Naive CD5hi CD8+ T cells are more reactive to common γ-chain cytokines than are their CD5lo counterparts24,26, and they have enhanced ability to produce IL-2 following stimulation via the TCR11, which might induce autocrine signaling via IL-2R. Also, the ability of inflammatory cues to augment the magnitude of CD8+ T cell responses involves sustained expression of the T cell activation–marker (and cytokine receptor chain) CD25 (ref. 38). To determine whether CD25 expression affected the differences in responses of CD5lo and CD5hi naive CD8+ T cells, we assessed the reactivity of CD25-deficient polyclonal CD8+ T cells, generated in mixed–bone marrow chimeras to avoid the lymphoproliferation and autoimmunity of CD25-deficient mice39. The distribution of CD5 expression was similar in wild-type and Cd25−/− resting naive CD8+ T cells (Supplementary Fig. 3i). As expected, the B8R–H-2Kb–specific wild-type CD5hi population expanded more than the wild-type CD5lo population did (Fig. 3g), but, while CD25 deficiency did not have a significant effect on the response of B8R–H-2Kb–specific CD5lo cells, the response of Cd25−/− CD5hi cells was modestly but significantly impaired (Fig. 3g). While these data suggested the CD5hi pool might have been more reliant on responsiveness to IL-2, the responses of CD5hi and CD5lo cells were not 'normalized' by CD25 deficiency, which indicated that elevated sensitivity to IL-2 could not fully account for the differences between these populations. Together these data indicated that the antigen-specific response to pathogens was dominated by CD5hi naive CD8+ T cells.
Distinct clonal responses of CD5hi versus CD5lo CD8+ T cells
Our analyses of bulk naive CD8+ T cell responses were unable to determine whether the differences in the expansion of CD5hi and CD5lo naive CD8+ T cell populations reflected enhanced responses by all antigen-reactive CD5hi cells or dominance by a small number of CD5hi CD8+ T cell clones. This is relevant because the expansion characteristics of progeny from individual antigen-specific naive T cells can vary considerably2,4,40,41,42. Accordingly, we reduced the number of adoptively transferred naive CD44lo CD5hi or CD5lo polyclonal naive CD8+ T cells to 25 × 103 to 30 × 103 cells. Given the frequency of B8R–H-2Kb–specific precursor cells35, 20% engraftment would seed approximately one B8R–H-2Kb–specific donor CD8+ T cell per three to five donor cell cohorts, which would result in an average predicted response rate of ∼27.5%. To increase the efficiency of detecting a clonal response, we used simultaneous transfer of up to eight congenically distinct donor populations into a single recipient, as described before2,4,40 (Supplementary Fig. 4). In studies of CD5hi naive CD8+ T cells, ∼24% (46 of 188) of the transfers led to a B8R–H-2Kb–specific response, which was not significantly different from the predicted frequency (Fig. 4a) and was consistent with studies of naive OT-I T cells40. In contrast, adoptive transfer of 25 × 103 CD5lo cells led to no detectable B8R–H-2Kb–specific donor responses (0 of 40 transfers), which was significantly below the predicted rate (Fig. 4a). Increasing the input of CD5lo donor cells to 100 × 103 led to detectable responses, but only in 14% (18 of 125) of transfers (Fig. 4a). This response rate suggested that less than one seventh of the expected number of CD5lo CD8+ T cell precursors were able to mount a detectable response. Examination of nonclonal responses by 375 × 103 CD5hi or CD5lo donor cells showed that >95% of CD5hi cells mounted a response (23 of 24 transfers), but only 70% of donor CD5lo cells did so (14 of 20 transfers) (Fig. 4b), which further demonstrated the reduced response rate of the CD5lo pool.
(a) Response rate of B8R–H-2Kb–specific donor cells (above the limit of detection of five cells) in the spleen of congenic recipients given sorted CD5lo or CD5hi cells (number of cells transferred, key), followed by infection of recipients with LM-B8R and analysis 7 d after infection (congenic marker scheme, Supplementary Fig. 4). NS, not significant (P = 0.2), response rate for 2.5 × 104 to 3.0 × 104 CD5hi cells versus expected response (∼27.5%), and *P < 0.0001, response rate of 2.5 × 104 CD5lo cells (0/40) or 1 × 105 CD5lo cells (18/125) versus expected response (25% or 100%, respectively) (one-sided binomial test). (b) Quantification of B8R–H-2Kb–specific CD8+ T cells derived from each donor population in a, as well as responses of host cells (key), presented as log-transformed values; horizontal dashed line indicates limit of detection (donor populations that did not mount a detectable response are grouped on that line); arrows indicate largest CD5lo clonal response. Each symbol represents an individual donor cohort; small horizontal lines indicate the geometric mean (± 95% confidence interval). *P < 0.05, for responses above the limit of detection (Mann-Whitney test). Data are from two to four independent experiments (error bars (a), s.d.).
Furthermore, the mean magnitude of the clonal expansion ('burst size') of the responding CD5lo population (from the transfer of 100 × 103 cells) was significantly smaller than that of the CD5hi population (from the transfer of 25 × 103 to 30 × 103 cells) (Fig. 4b). It was also notable that the two largest clonal responses were derived from CD5hi precursor cells and were 10- to 100-fold greater than the largest CD5lo clonal response (Fig. 4b). Modeling of the outcome if all the CD5hi and CD5lo clonal responses measured had occurred in a single mouse showed that those two clones would account for nearly 80% of the B8R–H-2Kb–specific population (data not shown).
Hence, clonal analysis revealed two ways in which the responses of CD5hi and CD5lo T cells differed. First, the CD5hi population displayed a markedly greater response rate. Second, even among cells that did engage in the B8R–H-2Kb–specific response, the average clonal burst size from the CD5lo pool was less than that from the CD5hi pool. Together, these differences accounted for much of the expansion advantage of the CD5hi pool.
Efficient recruitment of CD5hi CD8+ T cell responses
The greater clonal recruitment of CD5hi cells than of CD5lo cells might have reflected 'preferential' initial activation of CD5hi cells or a similar initial response by both populations, followed by improved proliferation and/or survival of the CD5hi population. The superior response by CD5hi cells was already apparent at days 3–4 of the in vivo response to LM-B8R (Fig. 5a); hence, we next investigated whether CD5hi cells were 'preferentially' activated during the initial response to infection. This was not feasible by adoptive transfer of polyclonal cells, and to determine the response of endogenous CD5hi and CD5lo cells, it was first necessary to determine whether CD5 expression changed during short-term activation in vivo. We gave Nur77GFP mice intravenous injection of anti-CD3 and, 5 h later, assessed T cell activation as induction of expression of the activation marker CD69 and Nur77GFP (as indicated by expression of the protein encoded (Nur77-GFP)). Despite robust activation, naive CD8+ T cells showed no change in CD5 expression (Fig. 5b,c), which indicated that CD5hi and CD5lo naive populations could still be distinguished. Next we infected Nur77GFP mice with LM-B8R and, 5 h later, enriched splenic CD8+ T cell populations specific for B8R–H-2Kb and for an irrelevant antigen (the H-2Kb-restricted epitope from amino acids 816–824 of mouse cytomegalovirus protein M57 (M57–H-2Kb)) by capture with MHC class I tetramers. Following infection with LM-B8R, activated naive CD8+ T cells were evident among the B8R–H-2Kb–specific population but not among the control M57–H-2Kb–specific population (Fig. 5d), and the activated B8R–H-2Kb–specific population showed enrichment for CD5hi cells (Fig. 5e,f). These data suggested that initial recruitment and/or activation favored the CD5hi naive CD8+ T cell pool during the response to foreign antigen.
(a) Responses of donor-derived B8R–H-2Kb–specific cells (assessed as in Fig. 3a) in the spleen of congenic recipients given congenically distinct CD44lo CD5lo and CD5hi cells (sorted and transferred together as in Fig. 3), followed by infection of recipients with LM-B8R and analysis on day 3 or 4 after infection. Each symbol represents an individual recipient mouse; small horizontal lines indicate the geometric mean (± 95% confidence interval). (b,c) Expression of Nur77-GFP and CD69 (b) and CD5 (c) on CD44lo CD8+ T cells in the spleen of Nur77GFP mice 5 h after intravenous injection of 50 μg anti-CD3 (α-CD3) or PBS. (d) Expression of Nur77-GFP and CD69 on B8R–H-2Kb– or M57–H-2Kb–specific CD8+ T cells isolated by tetramer-based enrichment from the spleen of Nur77GFP mice 5 h after infection with 1 × 107 colony-forming units of LM-B8R. (e) CD5 expression by B8R–H-2Kb–specific cells obtained as in d, with a non-activated (Non-act) (Nur77-GFPloCD69−) or activated (act) (Nur77-GFPhiCD69+) phenotype. (f) CD5 expression on non-activated and activated B8R–H-2Kb–specific naive CD8+ T cells as in e, presented as geometric mean fluorescence intensity (gMFI). Numbers adjacent to outlined areas (b,d) indicate percent CD44loCD8+ cells (left plots) or Nur77-GFPloCD69− cells (left) or Nur77-GFPhiCD69+ cells (right) (right plots). *P < 0.01 (unpaired t-test). Data are pooled from three experiments (one at day 3 (n = 5 mice) and two at day 4 (n = 9 mice)) (a), are from three independent experiments (n = 6 mice) (b,c) or are representative of or pooled from three experiments for B8R–H-2Kb–specific cells (n = 13 mice) or two experiments for M57–H-2Kb–specific cells (n = 9 mice) (d–f).
Similar TCR-binding characteristics of CD5lo and CD5hi cells
Some studies have suggested that CD5 expression on naive CD4+ T cells with transgenic TCR expression correlates with the affinity of the TCR for foreign peptide–MHC, indicated by greater labeling of CD5hi clones than CD5lo clones with pMHC tetramers9. However, we found similar intensities of staining with pMHC tetramers on CD5hi and CD5lo naive CD8+ T cell populations isolated from unimmunized mice by tetramer enrichment (Fig. 6a), which suggested similar capacities for binding foreign peptide–MHC ligands. Furthermore, the intensity of staining with B8R–H-2Kb tetramers was not significantly different on effector cells derived from clonal CD5lo responses and those derived from CD5hi responses, which revealed that intensity did not differ significantly for antigen-specific progeny of CD5lo clones versus those of CD5hi clones, whereas the burst size of CD5hi clones was significantly greater than that of CD5lo clones (Fig. 6b). Thus, we observed minimal correlation between the intensity of staining with pMHC tetramers and either CD5 expression or clonal-expansion characteristics of specific CD8+ T cells.
(a) Tetramer-staining intensity of B8R–H-2Kb– and M57–H-2Kb–specific CD8+ T cells (key) enriched from unimmunized mice with gating of CD44lo precursor cells on the lower and upper 20% of CD5 expression; results are presented as a ratio (CD5hi/CD5lo) to normalize results. Each symbol represents an individual mouse; small horizontal lines indicate the mean (± s.e.m.). (b) B8R–H-2Kb tetramer staining (left vertical axis) and burst size (right vertical axis) of expanded clonal populations derived from donor cells in recipient mice given 1 × 105 CD5lo cells and 2.5 × 104 to 3.0 × 104 CD5hi cells (as in Fig. 4), followed by infection of host mice with LM-B8R and analysis at day 7 after infection; tetramer staining on donor cells was normalized to that on B8R–H-2Kb–specific host cells from the same mouse to yield relative fluorescence intensity (RFI). ‡, outlier. (c) CD5 expression on bulk CD44lo CD8+ T cells and on B8R–H-2Kb–specific and M57–H-2Kb–specific CD44lo CD8+ T cell populations (key) isolated by tetramer enrichment from the spleen and lymph nodes of unimmunized mice. (d) CD5 expression on CD8+ T cells from C57BL/6 mice (B6) and various strains of mice with transgenic expression of MHC class I–restricted TCRs (key); right, results normalized to the average expression on the bulk CD44lo population and are presented relative to those of polyclonal C57BL/6 CD44lo CD8+ T cells (B6), set as 100. (e) Binding of various concentrations of MHC class I tetramers (horizontal axis) to cells of strains as in d (in parentheses in key), presented relative to maximum intensity, set as 100. (f) CD69 expression (in vitro activation) of CD8+ T cells obtained from mice of strains as in d (key), then incubated for 6 h with titrated doses of peptide (horizontal axis), assessed by flow cytometry; results are presented relative to maximum CD69 expression, set as 100% (horizontal dotted line indicates 50% response). (g) Expression of H-2Kb or H-2Db (H-2b) on RMA-S mouse tumor cells incubated with various doses (horizontal axis) of the peptide recognized by the TCR transgenically expressed by each strain of T cell (key); results are presented as staining of MHC class I relative to maximum staining intensity, set as 100 (horizontal dotted line indicates 50% of maximum). (h) CD5 expression by host CD44lo cells or donor OT-I cells (key) from congenic recipients of 1 × 103 cells of each population (wild-type or mutant) of congenically distinct CD44lo OT-I CD8+ T cells enriched from chimeras (generated by transfer of OT-I bone marrow into irradiated wild-type (WT) or β2-microglobulin-deficient (B2m−/−) hosts), followed by no further treatment of recipients or infection 1–3 d later with attenuated OVA-expressing L. monocytogenes ΔactA (ΔactA LM-OVA) (diagram at right). (i) Ratio of CD5hi OT-I donor dell populations to CD5lo OT-I donor cell populations, normalized to the average ratio of engraftment ('take') in uninfected recipients (Day 0), and at 7 d and 12 d after infection with OVA-expressing L. monocytogenes as in h. Each symbol represents an individual mouse; small horizontal lines indicate the mean (± s.e.m.). NS (b), P > 0.05, with or without inclusion of the outlier (‡); *P < 0.05 (unpaired t-test (b) and **P < 0.001 (paired t-test (c)). Data were pooled from four experiments (n = 14 mice (B8R–H-2Kb) or n = 9 mice (M57–H-2Kb)) (a), are from at least three experiments with 18 (CD5lo) or 46 (CD5hi) clonal responses (b), were pooled from six experiments for B8R–H-2Kb–specific cells (n = 21 mice) or two experiments for M57–H-2Kb–specific cells (n = 8 mice) (c; mean and s.e.m.), were pooled from two to three experiments (n = 4-6) (d; mean and s.e.m.), are from one experiment representative of two independent experiments with similar results (e,f), are representative of three independent experiments (g) or are from one experiment with four independent sets of chimeras (h,i).
It was also possible that foreign antigen–specific T cells were selectively under-represented in the CD5lo pool. We did observe modest, but in some cases significant, skewing toward higher CD5 expression in the pool of naive CD8+ T cells that bound foreign peptide–MHC tetramers (Fig. 6c) and, accordingly, there were slightly more B8R–H-2Kb–specific cells in sorted CD5hi populations than in sorted CD5lo populations (Supplementary Fig. 5). However, such skewing contributed an average increase of only ∼1.5-fold in antigen-specific precursor cells in the CD5hi population, relative to that in the CD5lo population (Supplementary Fig. 5), which could not explain the larger differences in the clonal recruitment or population expansion of antigen-specific CD5hi cells relative to that of CD5lo cells (Figs. 3 and 4).
To avoid potential artifacts from the tetramer-enrichment protocol, we analyzed four lines of mice with transgenic TCR expression that differ in surface expression of CD5, in the order H-Y < F5 < P14 < OT-I (lowest to highest CD5 expression)22,23,26 (Fig. 6d). All the strains bound cognate pMHC tetramers with similar efficiency in dose titration (Fig. 6e). which indicated that in contrast to studies of CD4+ T cells with transgenic TCR expression9, low CD5 expression versus high CD5 expression could not be used to predict the strength of the binding of foreign peptide–MHC ligands to CD8+ T cells with transgenic TCR expression. Interpreting tetramer staining may be complicated by the findings that the expression of CD8 and TCR was lower on CD5hi naive CD8+ T cells than on CD5lo naive CD8+ T cells (Supplementary Fig. 1a) and that CD8 contributes to binding of peptide–MHC class I tetramers43. Hence, we also assessed the reactivity (as induction of CD69 expression) of CD5hi and CD5lo T cells with transgenic TCR expression to their cognate foreign ligands in response to dose titration. Although we noted differences in antigen sensitivity, they did not correlate with the level of CD5 expression; for example, CD5hi OT-I CD8+ T cells and CD5lo F5 CD8+ T cells showed similar antigen sensitivity (Fig. 6f). Instead, dose sensitivity corresponded with binding of peptide to the relevant MHC molecules (Fig. 6g). Hence, these data indicated that CD5 expression could not be used to predict tetramer binding or in vitro antigen sensitivity of naive CD8+ T cells.
An expectation from our findings would be that CD5hi and CD5lo naive CD8+ T cells with identical TCRs would display distinct response characteristics. This hypothesis was supported by published studies of CD8+ T cells with transgenic TCR expression, sorted into CD5hi and CD5lo pools24, but as CD5 levels are typically determined during thymic development, we sought to manipulate positive selection to produce cells with distinct levels of CD5 expression. We generated chimeras by reconstituting wild-type or β2-microglobulin-deficient hosts with OT-I donor marrow; in the latter hosts, positive selection is mediated by hematopoietic cells, which resulted in generation of OT-I cells with lower CD5lo expression (Fig. 6h and data not shown). When we assessed their response to infection with OVA-expressing L. monocytogenes, we found that the CD5hi OT-I population expanded approximately three- to fourfold more than the CD5lo OT-I population (Fig. 6i), which indicated that the level of CD5 expression correlated with the magnitude of the immune response, even when TCR specificity was normalized. In aggregate, our data suggested that the advantage of CD5hi naive CD8+ T cells over CD5lo naive CD8+ T cells in their response to foreign antigen could not be explained by differences in precursor frequency or avidity for foreign peptide–MHC ligands.
Use of inflammatory cues by CD5hi CD8+ T cells
In addition to being influenced by TCR signals, the magnitude of the CD8+ T cell response is influenced by inflammatory cues44,45; hence, we next assessed the effect of inflammation on the response of CD5hi and CD5lo naive CD8+ T cells. Since the population expansion of CD8+ T cells is reduced in the absence of innate cues46,47, this system was not suitable for analysis of rare antigen-specific polyclonal CD8+ T cells; hence, we used H-Y T cells and OT-I T cells as examples of CD5lo clones and CD5hi clones, respectively. We transferred small numbers of H-Y and OT-I naive CD8+ T cells into congenic hosts and stimulated the cells by injection of DCs loaded with cognate peptides, with or without simultaneous infection with the attenuated L. monocytogenes strain ΔactA (which expresses no stimulatory antigens for either transgenically expressed TCR) as a source of inflammatory stimulation. We used OVA to stimulate OT-I cells, while we stimulated H-Y T cells with C2A, a variant of the Smcy peptide that enhances recognition of the H-Y TCR without altering MHC binding48 (Supplementary Fig. 6a–c), to minimize differences in the sensitivity of H-Y and OT-I T cell clones to ligand dose.
As expected, antigen-bearing DCs alone elicited modest responses by both H-Y cells and OT-I cells (Fig. 7a) and, after correction for donor-cell engraftment, there was moderately more expansion of the OT-I pool than of the H-Y pool (Fig. 7b). The addition of simultaneous infection with L. monocytogenes ΔactA enhanced expansion of the OT-I population, as anticipated on the basis of published studies38,49, but did not increase the population expansion of H-Y T cells and in fact caused a slight reduction in cell numbers (Fig. 7a). Accordingly, the addition of simultaneous infection with L. monocytogenes ΔactA greatly increased the difference between the H-Y population and OT-I population in terms of their size (Fig. 7b). We noted similar effects by the addition of infection with LCMV (Fig. 7c,d), and preliminary studies with coadministration of oligonucleotides containing the dinucleotide CpG (a Toll-like receptor 9 agonist), yielded comparable results (Supplementary Fig. 6d). These data suggested that while the CD5hi OT-I clone responded to proinflammatory signals with enhanced expansion, this pathway was not operative in CD5lo H-Y CD8+ T cells. The proinflammatory cytokines IL-12 and type I interferon act as a 'third signals' to promote CD8+ T cell responses46,47, but preliminary in vitro experiments did not suggest differences in the responses of H-Y T cells versus OT-I T cells to those cytokines (data not shown), which indicated a more complex basis for the altered response. Nevertheless, our data suggested qualitative differences in the responses of CD5hi CD8+ T cells versus those of CD5lo CD8+ T cells when foreign-antigen stimulation was delivered in the context of innate immune cues in vivo.
(a) Total donor CD8+ T cells in the spleen of congenic recipients of a mixture of congenically distinct CD44lo H-Y and OT-I CD8+ T cells (key), followed by immunization of recipients the next day with a mixture of matured DCs that were independently pulsed with OVA or C2A, with (DC + LM) or without (DC alone) infection with L. monocytogenes ΔactA and analysis 7 d later. (b) Ratio of OT-I cells to H-Y cells 1 d after adoptive transfer as in a, normalized to the 'take' ratio in unimmunized mice. (c) Total donor CD8+ T cells congenic recipients immunized as in a, with (DC + LCMV) or without (DC alone) infection with LCMV Armstrong strain. (d) Ratio of OT-I cells to H-Y cells 1 d after adoptive transfer as in c, normalized as in b. *P < 0.01 and **P < 0.001 (unpaired t-test). Data are pooled from three independent experiments (n = 10 mice) (a,b; mean and s.e.m.) or are from two experiments (n = 6 mice (DC alone) or n = 7 mice (DC + LCMV)) (c,d; mean and s.e.m.).
Discussion
Our data showed that naive CD8+ T cells with heightened recognition of self peptide–MHC ligands displayed enhanced reactivity toward foreign peptide–MHC antigens. We confirmed and extended the utility of CD5 expression as a measure of the strength of encounter with self ligand, showing that CD5hi cells exhibited increased expression of the Nur77GFP reporter (a surrogate of TCR signaling) and changes in gene expression indicative of enhanced response sensitivity. Comparison of the in vivo response to foreign antigen revealed multiple steps at which the CD5hi population of naive CD8+ T cells manifested an advantage over their CD5lo counterparts: their initial activation and response rates were more efficient, their clonal burst size was greater, and their sensitivity to inflammatory cues was enhanced. On the other hand, we did not observe a consistent difference in the capacity of polyclonal CD5hi cells or CD5hi cells with transgenic TCR expression to bind to foreign peptide–MHC tetramers versus that of their CD5lo counterparts, nor did models of transgenic TCR expression suggest a difference in sensitivity of the response to foreign peptide–MHC. Together our studies support a model in which the differences between CD5hi naive CD8+ T cells and CD5lo naive CD8+ T cells are established before encounter with foreign antigen and various properties of the CD5hi population make their responses more efficient and competitive.
Our data differ from two published reports that used CD5 expression to characterize heterogeneity in the responses of naive CD4+ T cells. While one study found that CD5hi cells had enhanced engagement of the TCR with foreign peptide–MHC ligands and superior responses to antigen in vivo9, another reported that CD5lo and CD5hi cells had similar engagement with pMHC ligands and that CD5lo cells showed greater population expansion in vivo than that of their CD5hi counterparts10,11. Although we observed some skewing in the size of the population that bound foreign peptide–MHC tetramers in favor of the CD5hi pool, this effect was modest, and average tetramer-binding intensity was similar for antigen-specific CD5lo cells and CD5hi cells. Hence, our data and published data10,11 challenge the conceptually complex model that the structural ability to bind foreign peptide–MHC ligands is dictated by the sensitivity of T cells to self peptide–MHC. These discrepancies may reflect distinct properties of CD4+ T cells and CD8+ T cells9, although this argument does not pertain to the divergent conclusions reached by studies of CD4+ T cells9,11. In any case, our findings reinforce the concept that, at least for naive CD8+ T cells, the distinct responses of the CD5hi and CD5lo populations probably reflect preexisting, intrinsic properties of the cells rather than arising from differences in perception of foreign antigen.
In contrast to our findings and other published results9, some reports have found that CD5lo populations expand more effectively than do CD5hi populations, despite having similar properties for the recognition of foreign ligands10,11. Notably, published studies have argued that CD5hi naive CD4+ T cells exhibit a strong response to stimulation via the TCR, but this leads to increased susceptibility to activation-driven cell death11. Our data suggested that optimal population expansion of activated CD5hi naive CD8+ T cells was dependent on CD25 expression, and enhanced sensitivity to IL-2 (through improved signaling via its receptor CD122) has been reported in studies of the homeostasis of CD5hi naive CD8+ T cells24. Enhanced sensitivity to IL-2 might be a boon to responding CD5hi CD8+ T cells yet may be detrimental to CD5hi CD4+ T cells (at least in some situations) by making them more vulnerable to induced cell death. We note that the range of CD5 expression (and associated basal TCR signaling) is greater in naive CD4+ T cells than in naive CD8+ T cells9,10,11, which is perhaps indicative of distinct functional thresholds in these subsets.
Our studies build on considerable work suggesting that CD5 levels correlate with engagement of the TCR by self peptide–MHC9,10,11,18,19,20,21. Our work has defined the properties of the CD5hi population before antigen encounter, as well as the mechanisms by which these cells outcompete other naive CD8+ T cell populations during an active immune response. It is unclear whether CD5 itself contributes to the distinct functions of CD5hi cells and CD5lo cells; studies of Cd5−/− mice do not support that concept9,11, although this does not negate the value of CD5 expression as a marker. As we have shown here, the CD5hi population differed from the CD5lo population in the expression of several genes. However, even within the CD5hi pool there was heterogeneity in the expression of T-bet and CXCR3 and induced expression of XCL1; hence, there may be other features of CD5hi cells that better correlate with their improved functional prowess. We found a modest but significant advantage of CXCR3hi CD5hi populations over CXCR3lo CD5hi populations, which suggested that CXCR3 expression may be a core feature of the optimal reactivity of CD5hi naive CD8+ T cells to foreign antigen.
Together these findings suggest that naive CD8+ T cells with the greatest self-reactivity are the cells most efficiently recruited into the foreign peptide–MHC–specific response. Since sensitivity to TCR signals may change following the activation of naive T cells, it is possible that progeny of some CD5hi clones could exhibit overt self-reactivity following activation, with relevance for the induction of autoimmune disease following the response to infection. At the same time, our findings leave open the question of why the CD5lo pool is maintained in the naive CD8+ T cell repertoire. CD5lo cells are relatively resistant to IL-7 deprivation26, which indicates it possible that these cells are efficiently maintained during the competition of naive T cells for homeostatic cytokines. Alternatively, CD5lo naive CD8+ T cells may show superior responses to pathogens in certain situations; as shown for naive CD4+ T cells, heightened initial reactivity may accompany increased sensitivity to cell death10,11. Whether some immune responses favor the CD5lo population of naive CD8+ T cells awaits further investigation.
Methods
Mice.
We purchased 6- to 12-week-old female C57BL/6 and B6.SJL mice from the National Cancer Institute. For recipients of adoptively transferred cells, we used F1 CD45.1/2 females generated from C57BL/6J mice (Jackson Laboratories) crossed with B6.SJL mice (National Cancer Institute). Il15−/− mice and P14 mice (with transgenic expression of a TCR specific for LCMV gp33)51 were gifts from D. Masopust. P14 and OT-I mice52 were maintained on a C57BL/6N and B6.PL (Thy-1.1+) backgrounds. H-Y mice were maintained on a background deficient in recombination-activating gene 2 (Rag2−/−) (apart from initial cell surface phenotype studies, in which cells from female Rag2+/+ H-Y mice were analyzed with monoclonal antibody T3.70 to identify H-Y-specific CD8+ T cells). F5 mice deficient in recombination-activating gene 1 (Rag1−/− mice) were a gift from L. Cauley, and Cd25−/− mice were obtained from Jackson Labs. The Nur77GFP transgenic reporter mice have been described30 and were maintained on a C57BL/6N background. T-bet–ZsGreen reporter mice50 were initially obtained from J. Zhu and were maintained on the C57BL/6N background. All mice were maintained in specific pathogen–free conditions, and all mouse protocols were approved by the University of Minnesota Institutional Animal Care and Use Committee. No samples or animals were excluded from the analysis. The investigators were not blinded to group allocations or assessment.
Bacterial and viral infection.
The attenuated L. monocytogenes strain ΔactA (DP-L1942)53 and OVA-expressing L. monocytogenes ΔactA were provided by J. Harty, and LM-B8R (both virulent and ΔactA), which contains both the H-2Kb-restricted CD8+ epitopes B8R (amino acids 20–27) and OVA (amino acids 257–264), was a gift from R. Kedl. LCMV, Armstrong strain, was a gift from D. Masopust. L. monocytogenes was grown in tryptic soy broth containing 50 μg/ml streptomycin to a absorbance of ∼0.1 at 600 nm. For primary infection with attenuated LM-B8R, 3 × 106 colony-forming units were injected intravenously into mice. For secondary infection with virulent LM-B8R, mice were given intravenous injection of 1 × 106 colony-forming units. In experiments in which wild-type L. monocytogenes ΔactA was used to induce inflammation, 3 × 106 to 6 × 106 colony-forming units of bacteria were mixed with peptide-pulsed DCs and the mixture was injected intravenously into mice. For infection with LCMV, mice were given intraperitoneal injection of 2 × 105 plaque-forming units.
Immunization with DCs.
Splenic DCs were prepared as described49. To generate splenic DCs, mice were given subcutaneous injection of 5 × 106 B16 mouse melanoma cells expressing the cytokine Flt3L (provided by M. Prlic and M. Bevan, via J. Harty). When tumors were palpable (5 mm × 5 mm), mice were given intravenous injection of 2 μg lipopolysaccharide to mature the DCs, and spleens were collected ∼16 h later. Following digestion for 20 min at 37 °C with collagenase D, red blood cells were lysed, and splenocytes were resuspended in medium composed of two parts complete RPMI medium (RPMI-1640 medium containing 10% fetal calf serum, 5 mM HEPES, 50 U/ml penicillin (sodium salt), 50 μg/ml streptomycin sulfate, 2 mM L-glutamine, 50 μM 2-mercaptoethanol and 50 μg/ml gentamycin sulfate) mixed with one part complete RPMI medium conditioned by Flt3L-expressing B16 cells, 50 ng/ml granulocyte-macrophage colony-stimulating factor and 2 μM peptide. The C2A mutant of the Smcy peptide54 was used for the stimulation of H-Y CD8+ T cells, while OVA peptide was used for the stimulation of OT-I cells. Splenocytes were pulsed for 2 h at 37 °C with peptide, then were thoroughly washed, and DCs were purified with Miltenyi CD11c microbeads. Mice were given intravenous injection of 1 × 106 DCs pulsed with each peptide along with coadministration of L. monocytogenes, LCMV or CpG.
Flow cytometry.
Cells were stained with the following antibodies (all antibodies from eBioscience or BD Biosciences unless noted otherwise): anti-CD4 (RM4-5), anti-CD8 (53-6.7), anti-CD5 (53-7.3), anti-CD27 (LG.7F9), anti-CD44 (IM7), anti-CD45.1 (A20), anti-CD45.2 (104), anti-Thy-1.1 (HIS51 or OX-7), anti-Thy-1.2 (53-2.1), anti-CD62L (MEL-14), anti-CD69 (H1.2F3), anti-CD122 (TM-b1), anti-CD127 (A7R34), anti-TCRβ (H57-597), anti-CXCR3 (CXCR3-173), anti-interferon-γ (XMG1.2), anti–MHC class II (M5/114.15.2) and anti-F4/80 (BM8). The B8R–H-2Kb and OVA–H-2Kb tetramers were generated as described43. The mouse cytomegalovirus M57–H-2Kb, LCMV gp33–H-2Db, influenza virus NP68–H-2Db and H-Y Smcy–H-2Db tetramers were provided by the NIH Tetramer Facility. For intracellular staining of transcription factors, cells were fixed and permeabilized with Foxp3 Fixation and Permeabilization Buffers (eBioscience) and were stained with anti-T-bet (4B10) and anti-eomesodermin (Dan11mag) in Permeabilization Solution. Data were collected on an LSR II or Fortessa flow cytometer (BD Biosciences), and data were analyzed with FlowJo analysis software (TreeStar).
XCL1 expression assay.
Peripheral lymphocytes were stained with monoclonal antibody to XCL1 (MTAC-2)55 provided by R. Kroczek (Robert Koch-Institute, Berlin, Germany). For analysis of XCL1 production, bulk splenocytes were stimulated for 3–5 h at 37 °C with PMA (phorbol 12-myristate 13-acetate) and ionomycin in the presence of brefeldin A. Cells were stained for cell surface markers, then were fixed and permeabilized with BD Cytofix/Cytoperm or eBiosciences Foxp3/transcription factor fixation/permeabilization solutions, before intracellular staining for XCL1.
Cell sorting and adoptive transfer.
For adoptive-transfer experiments, spleens and lymph nodes from C57BL/6 (CD45.2+CD45.2+) mice and B6.SJL (CD45.1+CD45.1+) mice were digested with collagenase D (Roche) and underwent negative enrichment for CD8+ T cells with Miltenyi enrichment antibody 'cocktail' and beads. Cells were then stained with anti-CD8, anti-CD5 and anti-CD44 (antibodies identified above), and CD8+CD44lo cells (i.e., samples from which CD44hi cells were excluded) were sorted on the lower or upper 20% of CD5 expression with a FACSAria I (BD). In some studies, the CD5hi CD44lo population was further gated on the lower or upper 30% of CXCR3 expression before sorting. Approximately 1.25 × 106 to 1.5 × 106 cells of each of the congenically mismatched CD5lo and CD5hi cell populations were transferred together into CD45.1+CD45.2+ recipients that that infected with LM-B8R the next day. For recall experiments, CD5lo and CD5hi recipients that had been infected with B8R-expressing L. monocytogenes ΔactA >40 d previously were challenged with virulent LM-B8R. Various combinations of congenic backgrounds for donor and host mice were used in transfer studies.
In experiments in which we transferred single B8R–H-2Kb–specific CD8+ T cell clones, CD8+ T cells underwent negative enrichment from the spleens and lymph node cells of 4 to 8 congenically distinct donors through the use of different combinations of CD45.1 and CD45.2 as well as CD90.1 and CD90.2 (Supplementary Fig. 4) (antibodies identified above). Equal numbers of CD8+ T cells from each congenic donor group were mixed, stained with anti-CD8, anti-CD5 and anti-CD44 (antibodies identified above)and sorted as naive CD8+ T cells in the lower or upper 20% of CD5 expression. The appropriate number of CD5lo and CD5hi cells from each congenic group was then transferred into congenic recipients. Mice were infected 1–2 d after transfer with attenuated LM-B8R, and the response to B8R–H-2Kb was assessed 7 d later. Background staining for congenic markers was very low (one event or less; data not shown), and we set our limit of detection at three or more flow cytometry events in the antigen-specific population, which is equal to about five total B8R–H-2Kb–specific CD8+ T cells.
In adoptive-transfer experiments with CD8+ T cells with transgenic TCR expression, CD44lo Thy-1.1+ OT-I cells (Rag1+/+ or Rag1−/−) underwent enrichment by negative selection as described56. Rag2−/− H-Y CD8+ T cells from female mice, which are all CD44lo cells, underwent negative enrichment with Miltenyi beads. Mixtures containing 1 × 103 cells of the OT-I population and 1 × 103 cells of the H-Y population were transferred intravenously into B6.SJL mice, and these recipients were immunized 1 d later. For analysis of the 'take', 2 × 105 cells from the same mixture of OT-I and H-Y cells was transferred into recipients, and cells from these mice were then analyzed by flow cytometry on the day of immunization. Similar 'take' ratios were observed for mice that received 1 × 103 OT-I and H-Y T cells that underwent enrichment by magnetic beads on the day of immunization (data not shown).
Enrichment with MHC class I tetramers.
For analysis of antigen-specific precursors of CD8+ T cells or CD5lo or CD5hi donor responses following infection, enrichment by MHC class I tetramers was used as described35. Spleens and lymph nodes (for analysis of precursors) or spleens only (for infection with L. monocytogenes) were digested with collagenase D. Cells were labeled with phycoerythrin- or allophycocyanin-conjugated tetramers and underwent enrichment over magnetic columns with anti-phycoerythrin or anti-allophycocyanin magnetic beads (Miltenyi). A small portion of the enriched fraction was added to AccuCheck counting beads (Invitrogen) for accurate back-calculation of total numbers. Fractions enriched for tetramers were then stained with additional extracellular antibodies (identified above) and were fixed with paraformaldehyde before analysis by flow cytometry.
Mixed bone marrow chimeras.
We generated mixed–bone marrow chimeras by obtained bone marrow from congenic strains, depleting the bone marrow of T cells and mixing the bone marrow from the different strains, and then injecting 5 × 106 to 10 × 106 cells into lethally irradiated (1,000 rads) host mice. For chimeras reconstituted with wild-type and Cd25−/− bone marrow, roughly equal numbers of cells from wild-type (CD45.1+CD45.2+) mice and Cd25−/− (CD45.2+) mice (6–8 weeks of age) were injected into wild-type (CD45.1+) hosts. For OT-I chimeras, OT-I (RAG+) bone marrow with disparate allelic expression of the alloantigen Thy-1 was injected into congenically distinct wild-type or β2-microglobulin-deficient recipients. Cells from chimeras were used >10 weeks after transplantation.
In vitro stimulation.
For analysis of CD8+ T cell activation, 2 × 104 purified CD44lo CD8+ cells with transgenic TCR expression were incubated at 37 °C with 1 × 106 to 2 × 106 splenic antigen-presenting cells in 96-well round-bottomed plates with titrated doses of cognate peptide: OT-I cells were incubated with OVA–H-2Kb (OVA amino acids 257–264: SIINFEKL), P14 cells were incubated with gp33–H-2Db (gp amino acids 33–41: KAVYNFATC), F5 cells were incubated with NP366–H-2Db (NP amino acids 366–374; ASNENMDAM), and H-Y cells were incubated with Smcy–H-2Db (KCSRNRQYL) or C2A–H-2Db (KASRNRQYL). Cells were stimulated for 6 h and then stained for CD69 expression.
Assay of MHC class I stabilization in RMA-S cells.
RMA-S cells were cultured at 30 °C with 5% CO2 overnight in RPMI medium containing 10% FCS. In a 96-well round-bottomed plate, 1 × 105 RMA-S cells were incubated for 1 h with titrated doses of peptide and then the plate was moved to a 37 °C CO2 incubator for 3 h. Stable surface MHC class I molecules on cells were then stained with antibody to H-2Kb (Y3) or H-2Db (28.14.8) (both produced at the University of Minnesota).
Gene-transcription analysis.
Naive CD44lo CD8+ T cells from spleens and lymph nodes were sorted by flow cytometry as cells with the lower and upper 20% of CD5 expression as described above. For each sample, ≥1 × 106 cells were used for RNA extraction with an RNeasy microkit (Qiagen). RNA was used to generate biotinylated cRNA with a MessageAmpIII RNA Amplification kit according to the manufacturer's recommendations (Ambion). Samples were hybridized to Affymetrix murine 430 2.0 gene chips at the BioMedical Genomics Center (University of Minnesota) by standard procedures. RNA samples from three independent sorts were analyzed. Genespring software was used to generate gene-expression analysis for Table 1: data were normalized with the MAS5 algorithm and were filtered for present or absent 'calls' in at least one group and for a significant (P < 0.05) difference in expression of twofold or more. For enrichment analysis (Table 2), cluster genes expressed by either CD5hi cells or CD5lo cells were determined to be any genes with a change in expression of >0. Significance was determined by χ2, in which equal distribution was taken as the null hypothesis.
Statistics.
Unless indicated otherwise in the figure legend(s), a two-tailed, unpaired Student's t-test was performed on log-transformed data with Prism (GraphPad Software). For multiple comparisons, one-way analysis of variance with Dunnett's multiple-comparison post-test was used. Sample sizes were chosen on the basis of previous experience and similar studies. In the clonal analysis in Figure 4, the data distribution was not normal or log-normal, and the nonparametric Mann-Whitney test was applied.
Accession codes.
GEO: microarray data, GSE62142.
Accession codes
References
Zehn, D., Lee, S.Y. & Bevan, M.J. Complete but curtailed T-cell response to very low-affinity antigen. Nature 458, 211–214 (2009).
Tubo, N.J. et al. Single naive CD4+ T cells from a diverse repertoire produce different effector cell types during infection. Cell 153, 785–796 (2013).
Fazilleau, N., McHeyzer-Williams, L.J., Rosen, H. & McHeyzer-Williams, M.G. The function of follicular helper T cells is regulated by the strength of T cell antigen receptor binding. Nat. Immunol. 10, 375–384 (2009).
Plumlee, C.R., Sheridan, B.S., Cicek, B.B. & Lefrancois, L. Environmental cues dictate the fate of individual CD8+ T cells responding to infection. Immunity 39, 347–356 (2013).
Dorfman, J.R., Stefanova, I., Yasutomo, K. & Germain, R.N. CD4+ T cell survival is not directly linked to self-MHC-induced TCR signaling. Nat. Immunol. 1, 329–335 (2000).
Stefanová, I., Dorfman, J.R. & Germain, R.N. Self-recognition promotes the foreign antigen sensitivity of naive T lymphocytes. Nature 420, 429–434 (2002).
Bhandoola, A. et al. Peripheral expression of self-MHC-II influences the reactivity and self-tolerance of mature CD4+ T cells: evidence from a lymphopenic T cell model. Immunity 17, 425–436 (2002).
Hochweller, K. et al. Dendritic cells control T cell tonic signaling required for responsiveness to foreign antigen. Proc. Natl. Acad. Sci. USA 107, 5931–5936 (2010).
Mandl, J.N., Monteiro, J.P., Vrisekoop, N. & Germain, R.N. T cell-positive selection uses self-ligand binding strength to optimize repertoire recognition of foreign antigens. Immunity 38, 263–274 (2013).
Weber, K.S. et al. Distinct CD4+ helper T cells involved in primary and secondary responses to infection. Proc. Natl. Acad. Sci. USA 109, 9511–9516 (2012).
Persaud, S.P., Parker, C.R., Lo, W.L., Weber, K.S. & Allen, P.M. Intrinsic CD4 T cell sensitivity and response to a pathogen are set and sustained by avidity for thymic and peripheral complexes of self peptide and MHC. Nat. Immunol. 15, 266–274 (2014).
Starr, T.K., Jameson, S.C. & Hogquist, K.A. Positive and negative selection of T cells. Annu. Rev. Immunol. 21, 139–176 (2003).
Tanchot, C., Lemonnier, F.A., Perarnau, B., Freitas, A.A. & Rocha, B. Differential requirements for survival and proliferation of CD8 naive or memory T cells. Science 276, 2057–2062 (1997).
Takada, K. & Jameson, S.C. Self-class I MHC molecules support survival of naive CD8 T cells, but depress their functional sensitivity through regulation of CD8 expression levels. J. Exp. Med. 206, 2253–2269 (2009).
Takada, K. & Jameson, S.C. Naive T cell homeostasis: from awareness of space to a sense of place. Nat. Rev. Immunol. 9, 823–832 (2009).
Sprent, J. & Surh, C.D. Normal T cell homeostasis: the conversion of naive cells into memory-phenotype cells. Nat. Immunol. 12, 478–484 (2011).
Tarakhovsky, A. et al. A role for CD5 in TCR-mediated signal transduction and thymocyte selection. Science 269, 535–537 (1995).
Azzam, H.S. et al. CD5 expression is developmentally regulated by T cell receptor (TCR) signals and TCR avidity. J. Exp. Med. 188, 2301–2311 (1998).
Wong, P., Barton, G.M., Forbush, K.A. & Rudensky, A.Y. Dynamic tuning of T cell reactivity by self-peptide-major histocompatibility complex ligands. J. Exp. Med. 193, 1179–1187 (2001).
Smith, K. et al. Sensory adaptation in naive peripheral CD4 T cells. J. Exp. Med. 194, 1253–1261 (2001).
Seddon, B. & Zamoyska, R. TCR signals mediated by Src family kinases are essential for the survival of naive T cells. J. Immunol. 169, 2997–3005 (2002).
Kieper, W.C., Burghardt, J.T. & Surh, C.D. A role for TCR affinity in regulating naive T cell homeostasis. J. Immunol. 172, 40–44 (2004).
Ge, Q., Bai, A., Jones, B., Eisen, H.N. & Chen, J. Competition for self-peptide-MHC complexes and cytokines between naive and memory CD8+ T cells expressing the same or different T cell receptors. Proc. Natl. Acad. Sci. USA 101, 3041–3046 (2004).
Cho, J.H., Kim, H.O., Surh, C.D. & Sprent, J. T cell receptor-dependent regulation of lipid rafts controls naive CD8+ T cell homeostasis. Immunity 32, 214–226 (2010).
Johnson, L.D. & Jameson, S.C. Self-specific CD8+ T cells maintain a semi-naive state following lymphopenia-induced proliferation. J. Immunol. 184, 5604–5611 (2010).
Palmer, M.J., Mahajan, V.S., Chen, J., Irvine, D.J. & Lauffenburger, D.A. Signaling thresholds govern heterogeneity in IL-7-receptor-mediated responses of naive CD8+ T cells. Immunol. Cell Biol. 89, 581–594 (2011).
Park, J.H. et al. 'Coreceptor tuning': cytokine signals transcriptionally tailor CD8 coreceptor expression to the self-specificity of the TCR. Nat. Immunol. 8, 1049–1059 (2007).
Kaech, S.M. & Cui, W. Transcriptional control of effector and memory CD8+ T cell differentiation. Nat. Rev. Immunol. 12, 749–761 (2012).
Judge, A.D., Zhang, X., Fujii, H., Surh, C.D. & Sprent, J. Interleukin 15 controls both proliferation and survival of a subset of memory-phenotype CD8+ T cells. J. Exp. Med. 196, 935–946 (2002).
Moran, A.E. et al. T cell receptor signal strength in Treg and iNKT cell development demonstrated by a novel fluorescent reporter mouse. J. Exp. Med. 208, 1279–1289 (2011).
Chang, J.T. et al. Asymmetric proteasome segregation as a mechanism for unequal partitioning of the transcription factor T-bet during T lymphocyte division. Immunity 34, 492–504 (2011).
Miller, A.T., Wilcox, H.M., Lai, Z. & Berg, L.J. Signaling through Itk promotes T helper 2 differentiation via negative regulation of T-bet. Immunity 21, 67–80 (2004).
Dorner, B.G. et al. Selective expression of the chemokine receptor XCR1 on cross-presenting dendritic cells determines cooperation with CD8+ T cells. Immunity 31, 823–833 (2009).
Best, J.A. et al. Transcriptional insights into the CD8+ T cell response to infection and memory T cell formation. Nat. Immunol. 14, 404–412 (2013).
Haluszczak, C. et al. The antigen-specific CD8+ T cell repertoire in unimmunized mice includes memory phenotype cells bearing markers of homeostatic expansion. J. Exp. Med. 206, 435–448 (2009).
Sung, J.H. et al. Chemokine guidance of central memory T cells is critical for antiviral recall responses in lymph nodes. Cell 150, 1249–1263 (2012).
Kastenmüller, W. et al. Peripheral prepositioning and local CXCL9 chemokine-mediated guidance orchestrate rapid memory CD8+ T cell responses in the lymph node. Immunity 38, 502–513 (2013).
Starbeck-Miller, G.R., Xue, H.H. & Harty, J.T. IL-12 and type I interferon prolong the division of activated CD8 T cells by maintaining high-affinity IL-2 signaling in vivo. J. Exp. Med. 211, 105–120 (2014).
Malek, T.R. & Castro, I. Interleukin-2 receptor signaling: at the interface between tolerance and immunity. Immunity 33, 153–165 (2010).
Buchholz, V.R. et al. Disparate individual fates compose robust CD8+ T cell immunity. Science 340, 630–635 (2013).
Gerlach, C. et al. Heterogeneous differentiation patterns of individual CD8+ T cells. Science 340, 635–639 (2013).
van Heijst, J.W. et al. Recruitment of antigen-specific CD8+ T cells in response to infection is markedly efficient. Science 325, 1265–1269 (2009).
Daniels, M.A. & Jameson, S.C. Critical role for CD8 in T cell receptor binding and activation by peptide/major histocompatibility complex multimers. J. Exp. Med. 191, 335–346 (2000).
Haring, J.S., Badovinac, V.P. & Harty, J.T. Inflaming the CD8+ T cell response. Immunity 25, 19–29 (2006).
Cui, W. & Kaech, S.M. Generation of effector CD8+ T cells and their conversion to memory T cells. Immunol. Rev. 236, 151–166 (2010).
Badovinac, V.P., Messingham, K.A., Jabbari, A., Haring, J.S. & Harty, J.T. Accelerated CD8+ T-cell memory and prime-boost response after dendritic-cell vaccination. Nat. Med. 11, 748–756 (2005).
Curtsinger, J.M. & Mescher, M.F. Inflammatory cytokines as a third signal for T cell activation. Curr. Opin. Immunol. 22, 333–340 (2010).
Maile, R. et al. Peripheral “CD8 tuning” dynamically modulates the size and responsiveness of an antigen-specific T cell pool in vivo. J. Immunol. 174, 619–627 (2005).
Pham, N.L., Badovinac, V.P. & Harty, J.T. A default pathway of memory CD8 T cell differentiation after dendritic cell immunization is deflected by encounter with inflammatory cytokines during antigen-driven proliferation. J. Immunol. 183, 2337–2348 (2009).
Zhu, J. et al. The transcription factor T-bet is induced by multiple pathways and prevents an endogenous Th2 cell program during Th1 cell responses. Immunity 37, 660–673 (2012).
Pircher, H., Rohrer, U.H., Moskophidis, D., Zinkernagel, R.M. & Hengartner, H. Lower receptor avidity required for thymic clonal deletion than for effector T-cell function. Nature 351, 482–485 (1991).
Hogquist, K.A. et al. T cell receptor antagonist peptides induce positive selection. Cell 76, 17–27 (1994).
Brundage, R.A., Smith, G.A., Camilli, A., Theriot, J.A. & Portnoy, D.A. Expression and phosphorylation of the Listeria monocytogenes ActA protein in mammalian cells. Proc. Natl. Acad. Sci. USA 90, 11890–11894 (1993).
Maile, R. et al. Antigen-specific modulation of an immune response by in vivo administration of soluble MHC class I tetramers. J. Immunol. 167, 3708–3714 (2001).
Dorner, B.G. et al. MIP-1α, MIP-1β, RANTES, and ATAC/lymphotactin function together with IFN-γ as type 1 cytokines. Proc. Natl. Acad. Sci. USA 99, 6181–6186 (2002).
Kieper, W.C., Prlic, M., Schmidt, C.S., Mescher, M.F. & Jameson, S.C. Il-12 enhances CD8 T cell homeostatic expansion. J. Immunol. 166, 5515–5521 (2001).
Acknowledgements
We thank D. Masopust (University of Minnesota) for Il15−/− mice, P14 mice and LCMV Armstrong strain; L. Cauley (University of Connecticut) for F5 mice deficient in recombination-activating gene 1; J. Zhu (National Institute of Allergy and Infectious Diseases) for T-bet–ZsGreen reporter mice; J. Harty (University of Iowa) for L. monocytogenes strain ΔactA (DP-L1942) and OVA-expressing L. monocytogenes ΔactA; R. Kedl (National Jewish Medical Research Center) for LM-B8R (virulent and ΔactA); M. Prlic and M. Bevan (University of Washington) and J. Harty (University of Iowa) for Flt3L-expressing B16 cells; R. Kroczek (Robert Koch-Institute) for the conjugated monoclonal antibody MTAC-2; the University of Minnesota Flow Core personnel for flow-cytometry support and cell sorting; M. Jenkins and M. Mescher for critical review of the manuscript; G. Stritesky for insight on the analysis of Nur77 mice; J. Ding and S. Peery for technical assistance with mice; and members of the Jamequist laboratory for discussions. Supported by the US National Institutes of Health (R37 AI-38903 to S.C.J.; P30 CA77598, with the Biostatistics and Bioinformatics Core shared resource of the Masonic Cancer Center, University of Minnesota; UL1TR000114 from the National Center for Advancing Translational Sciences) and the Irvington Institute Fellowship Program of the Cancer Research Institute (R.B.F.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institutes of Health.
Author information
Authors and Affiliations
Contributions
R.B.F. and S.C.J. designed the experimental approaches; R.B.F. and S.E.H. conducted experiments; Y.X. and K.A.H. provided mouse strains and bone marrow chimeras; J.A.B. and A.W.G. analyzed gene-expression data; and R.B.F. and S.C.J. wrote and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 Phenotypic characteristics of naive CD5lo and CD5hi CD8+ T cells in wild-type and Il15−/− mice.
Peripheral lymphocytes were isolated from WT (a and c) or IL-15-/- (b and c) mice. Naive (CD44lo) CD8 T cells were gated on the lower (red) and upper (blue) 20% of CD5 expression and compared to memory phenotype (MP) cells (CD44hiCD122hi, gray shaded). Flow plots are representative of 2 separate experiments. (d) CD5hi and CD5lo naïve CD8 T cells were sorted and transferred into normal hosts (as in Fig 1c). The graph shows the percentage of each population that acquired CD44hi (memory)-phenotype, presented as mean ± SEM). Data in (a,c) are representative of 4 experiments and those in (b) 2 experiments. * indicates p<0.05.
Supplementary Figure 2 Naive CD5hi CD8+ T cells exhibit rapid production of the chemokine XCL1.
Splenocytes from T-bet-GFP reporter mice were left untreated or stimulated with PMA/ionomycin in the presence of brefeldin A. Cells were then stained for CD8, CD44 and CD5, and then fixed, permeabilized and stained intracellularly for XCL1 and also CXCR3 (since detectable surface expression of CXCR3 was reduced by stimulation). T-bet expression was determined using the T-bet-ZsGreen (enhanced GFP) reporter (note – some quenching of the ZsGreen reporter is apparent in stimulated cells). In other experiments (including those shown in Fig. 1a,b), T-bet was detected by intracellular staining, with similar results. (a) Expression of CXCR3 and T-bet (T-bet-ZsGreen reporter) was determined in unstimulated cells, while staining for XCL1 was assessed after PMA/Ionomycin stimulation. The cells were counterstained for CD5 and the data gated on CD44lo CD8+ T cells. (b,c) Co-expression of XCL-1, CXCR3 and T-bet in (b) CD44hi memory phenotype CD8+ T cells or (c) CD44lo CD5hi CD8+ T cells. Cells were stimulated with PMA/Ionomycin or not, as indicated. Using Boolean gating, the average frequency of (stimulated) CD44hi cells positive for CXCR3, T-bet AND XCL1 was 36.5%, while that for CD44lo, CD5hi cells was 1.2% (data not shown) in this experiment. These data are representative of 3 experiments (n=7), utilizing T-bet reporter cells.
Supplementary Figure 3 Additional characterization of the expansion potential of naive CD5hi and CD5lo CD8+ T cell populations.
(A and B) Detection of CD5lo/hi B8R/Kb-specific (A) and bulk LM-specific cells (B) by IFN-γ. Bulk splenocytes were stimulated with 1 μM B8R20-27 peptide (A) or PMA/ionomycin (B) for 5 hrs in the presence of BFA and then stained for intracellular IFN-γ. (C-E) The preferential expansion of naive CD5hi CD8 T cells is not unique to B8R/Kb specific cells or to LM infection. (C) shows the ratio of donor CD5hi: CD5lo OVA/Kb-specific cells on day 7 after primary LM-B8R infection or 5 days after re-challenge of memory recipients with virulent LM-B8R. Red symbols indicate mice where OVA/Kb-specific responses were not detected within the CD5lo donor population and the ratio was arbitrarily set to 100. (D and E) Ratio of donor CD5hi:CD5lo gp33/Db (D) or bulk pathogen-specific cells (E) on days 7 and 30+ after primary acute infection with LCMV Armstrong. (F) In vitro stimulation of CFSE-labeled polyclonal CD5lo/hi cells with plate-bound αCD3/αCD28 (2.5ug/mL) for 6 days. (G) CD5hi donor cells still have an advantage over CD5lo cells in RAG-/- recipients. Donor cells were transferred into RAG-/- recipients, infected the next day with LM-B8R, and the splenic responses were analyzed 7 days later. (H) CD5lo/hi cells were sorted on equally low CD44 expression and adoptively transferred into congenic wild type recipients. Mice were infected with LM-B8R and responses in peripheral blood were analyzed 7 days later. Red symbol indicates mice were the B8R/Kb-specific response was not detected within the CD5lo donor population and the ratio was arbitrarily set to 100. (I) CD5 expression levels on WT and CD25-/- naïve CD8+ T cells derived from bone marrow chimeras (described in Fig. 3g). Data in (a,b) are compiled from 1 to 4 experiments (n=4-11), (c) compiled from 2-4 experiments (n=6-11), (d) compiled from 2 experiments (n=4-7) (f) is representative of 2 experiments (g,h) from 1 experiment (n=4) (i) is representative of 3 chimera sets (n=6).
Supplementary Figure 4 Schematic for the simultaneous adoptive transfer of multiple congenic naive CD5lo or CD5hi CD8+ T cells.
Peripheral CD8 T cells from 4-8 congenic donors were negatively enriched and then equal numbers from each donor were combined. CD44lo CD5lo and CD5hi cells were flow sorted and then the indicated number of each individual donor population was transferred into congenic recipients. Mice were infected the next day with LM-B8R and the B8R/Kb-specific response was assessed 7 days later using tetramer enrichment
Supplementary Figure 5 Skewed CD5 distribution on naive LM-B8R–H-2Kb–specific CD8+ T cell precursors.
The gates demarcating the lower and upper 20% for CD5 expression on bulk CD44lo CD8+ T cells cells were applied to CD44lo B8R/Kb tetramer stained CD8+ T cells from the same sample. The data indicate what percentage of B8R/Kb specific cells are contained in the gates used for sorting CD5hi and CD5lo cells. Values above 20% indicate the B8R/Kb specific naïve CD8+ T cells was overrepresented in that sorted population, values less than 20% indicate B8R/Kb specific population were underrepresented in that group. Data are cumulative from 5 separate experiments (n=17) and are shown as mean ± SEM. Median values were used to calculate the fold-change from the bulk population (i.e. by how much the B8R/Kb-specific population is under- or overrepresented in that population).
Supplementary Figure 6 Characterization of the C2A peptide, and effect of CpG on the response of OT-I and H-Y T cells.
(a) Relative stabilization of H-2Db on RMA-S cells following incubation with titrated doses of the HY Smcy/Db peptide with or without a C2A substitution. Data are shown as mean ± SEM. (b) H-Y TCR transgenic MHC class I tetramer binding intensities (geometric MFI normalized to maximum intensity) over a range of tetramer concentrations. Data are representative of one out of two experiments. (c) In vitro activation of H-Y CD8 T cells. T cells were incubated with titrated doses of wild-type or altered C2A peptide for 6 hrs and CD69 expression was assessed by flow cytometry. Data is normalized to maximum CD69 expression and representative of two experiments. (d) HY and OT-I T cells were adoptively co-transferred and primed with peptide-pulsed DC +/- CpG administration, and the response of each population was measured on day 7 after priming. Data are presented in the same way as Fig. 7a,b. Data in (a) are compiled from 3 separate experiments, (b,c) are representative of 2 separate experiments; (d) is from a single experiment (n=3 for DC alone and n=4 for DC+CpG).
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–6 (PDF 922 kb)
Rights and permissions
About this article
Cite this article
Fulton, R., Hamilton, S., Xing, Y. et al. The TCR's sensitivity to self peptide–MHC dictates the ability of naive CD8+ T cells to respond to foreign antigens. Nat Immunol 16, 107–117 (2015). https://doi.org/10.1038/ni.3043
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.3043
This article is cited by
-
T-cell receptor-based therapy: an innovative therapeutic approach for solid tumors
Journal of Hematology & Oncology (2021)
-
Loss of the orphan nuclear receptor NR2F6 enhances CD8+ T-cell memory via IFN-γ
Cell Death & Disease (2021)
-
Self-reactivity controls functional diversity of naive CD8+ T cells by co-opting tonic type I interferon
Nature Communications (2021)
-
Infection-induced type I interferons critically modulate the homeostasis and function of CD8+ naïve T cells
Nature Communications (2021)
-
Rapid statistical discrimination of fluorescence images of T cell receptors on immobilizing surfaces with different coating conditions
Scientific Reports (2021)