Abstract
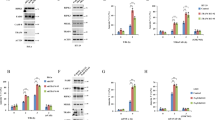
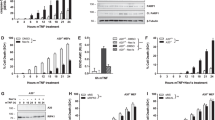
The physiological function of the adaptor protein TRADD remains unclear because of the unavailability of a TRADD-deficient animal model. By generating TRADD-deficient mice, we found here that TRADD serves an important function in tumor necrosis factor receptor 1 (TNFR1) signaling by orchestrating the formation of TNFR1 signaling complexes. TRADD was essential for TNFR1 signaling in mouse embryonic fibroblasts but was partially dispensable in macrophages; abundant expression of the adaptor RIP in macrophages may have allowed some transmission of TNFR1 signals in the absence of TRADD. Although morphologically normal, TRADD-deficient mice were resistant to toxicity induced by TNF, lipopolysaccharide and polyinosinic-polycytidylic acid. TRADD was also required for TRIF-dependent Toll-like receptor signaling in mouse embryonic fibroblasts but not macrophages. Our findings definitively establish the biological function of TRADD in TNF signaling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Tartaglia, L.A. & Goeddel, D.V. Two TNF receptors. Immunol. Today 13, 151–153 (1992).
Tracey, K.J. & Cerami, A. Tumor necrosis factor, other cytokines and disease. Annu. Rev. Cell Biol. 9, 317–343 (1993).
Baeuerle, P.A. & Baltimore, D. NF-κB: ten years after. Cell 87, 13–20 (1996).
Nagata, S. Apoptosis by death factor. Cell 88, 355–365 (1997).
Baud, V. & Karin, M. Signal transduction by tumor necrosis factor and its relatives. Trends Cell Biol. 11, 372–377 (2001).
Chen, G. & Goeddel, D.V. TNF-R1 signaling: a beautiful pathway. Science 296, 1634–1635 (2002).
Wajant, H., Pfizenmaier, K. & Scheurich, P. Tumor necrosis factor signaling. Cell Death Differ. 10, 45–65 (2003).
Locksley, R.M., Killeen, N. & Lenardo, M.L. The TNF and TNF receptor superfamilies: integrating mammalian biology. Cell 104, 487–501 (2001).
Hsu, H., Xiong, J. & Goeddel, D.V. The TNF receptor-1 associated protein TRADD signals cell death and NF-B activation. Cell 81, 495–504 (1995).
Hsu, H., Shu, H.B., Pan, M.P. & Goeddel, D.V. TRADD-TRAF2 and TRADD-FADD interactions define two distinct TNF signal transduction pathways. Cell 84, 299–308 (1996).
Hsu, H., Huang, J., Shu, H.B., Baichwal, V. & Goeddel, D.V. TNF-dependent recruitment of the protein kinase RIP to the TNF receptor-1 signaling complex. Immunity 4, 387–396 (1996).
Micheau, O. & Tschopp, J. Induction of TNF receptor I-mediated apoptosis via two sequential signaling complexes. Cell 114, 181–190 (2003).
Yeh, W.C. et al. Early lethality, functional NF-κB activation, and increased sensitivity to TNF-induced cell death in TRAF2-deficient mice. Immunity 7, 715–725 (1997).
Yeh, W.C. et al. FADD: Essential for embryo development and sygnaling from some, but not all, inducers of apoptosis. Science 279, 1954–1958 (1998).
Zhang, J., Cado, D., Chen, A., Kabra, N.H. & Winoto, A. Fas-mediated apoptosis and activation-induced T-cell proliferation are defective in mice lacking FADD/Mort1. Nature 392, 296–300 (1998).
Kelliher, M.A. et al. The death domain kinase RIP mediates the TNF-induced NF-κB signal. Immunity 8, 297–303 (1998).
Devin, A., Lin, Y. & Liu, Z.G. The role of the death-domain kinase RIP in tumour-necrosis-factor-induced activation of mitogen-activated protein kinases. EMBO Rep. 4, 623–627 (2003).
Lin, Y. et al. Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein-mediated cellular reactive oxygen species accumulation. J. Biol. Chem. 279, 10822–10828 (2004).
Holler, N. et al. Fas triggers an alternative, caspase-8-independent cell death pathway using the kinase RIP as effector molecule. Nat. Immunol. 1, 489–495 (2000).
Devin, A. et al. The distinct roles of TRAF2 and RIP in IKK activation by TNFRI: TRAF2 recruits IKK to TNFRI while RIP mediates IKK activation. Immunity 12, 419–429 (2000).
Zhang, S.Q., Kovalenko, A., Cantarella, G. & Wallach, D. Recruitment of the IKK signalosome to the p55 TNF receptor: RIP and A20 bind to NEMO (IKKγ) upon receptor stimulation. Immunity 12, 301–311 (2000).
Zhu, G., Wu, C.J., Zhao, Y. & Ashwell, J.D. Optineurin negatively regulates TNFα- induced NF-kappaB activation by competing with NEMO for ubiquitinated RIP. Curr. Biol. 17, 1438–1443 (2007).
Kim, Y., Morgan, M.J., Choksi, S. & Liu, Z.G. TNF-induced activation of the Nox1 NADPH oxidase and its role in the Induction of necrotic cell death. Mol. Cell 26, 769–771 (2007).
Jin, Z. & El-Deiry, W.S. Distinct signaling pathways in TRAIL- versus tumor necrosis factor-induced apoptosis. Mol. Cell. Biol. 26, 8136–8148 (2006).
Zheng, L. et al. Competetive Control of independent programs of tumor necrosis facor-induced cell death by TRADD and RIP1. Mol. Cell. Biol. 26, 3505–3513 (2006).
Lakso, M. et al. Efficient in vivo manipulation of mouse genomic sequences at the zygote stage. Proc. Natl. Acad. Sci. USA 93, 5860–5865 (1996).
Tiegs, G., Wolter, M. & Wendel, A. Tumor necrosis factor is a terminal mediator in galactosamine/endotoxin-induced hepatitis in mice. Biochem. Pharmacol. 38, 627–631 (1989).
Decker, K. & Keppler, D. Galactosamine hepatitis: key role of the nucleotide deficiency period in the pathogenesis of cell injury and cell death. Rev. Physiol. Biochem. Pharmaco. 71, 77–106 (1974).
Fiers, W., Beyaert, R., Declercq, W. & Vandenabeele, P. More than one way to die: apoptosis, necrosis and reactive oxygen damage. Oncogene 18, 7719–7730 (1999).
Lin, Y. et al. Tumor necrosis factor-induced nonapoptotic cell death requires receptor-interacting protein mediated reactive oxygen species accumulation. J. Biol. Chem. 279, 10822–10828 (2004).
Wertz, I.E. et al. De-ubiquitination and ubiquitin ligase domains of A20 downregulate NF-κB signalling. Nature 430, 694–699 (2004).
Tada, K. et al. Critical roles of TRAF2 and TRAF5 in tumor necrosis factor-induced NF-κB activation and protection from cell death. J. Biol. Chem. 276, 36530–36534 (2001).
Nowak, M. et al. LPS-induced liver injury in d-galactosamine-sensitized mice requires secreted TNFα and TNF-p55 receptor. Am. J. Physiol. Regul. Integr. Comp. Physiol. 278, R1202–R1209 (2000).
Dejager, L. & Libert, C. Tumor necrosis factor α mediates the lethal hepatotoxic effects of poly(I:C) in d-galactosamine-sensitized mice. Cytokine 42, 55–61 (2008).
Kawai, T., Adachi, O., Ogawa, T., Takeda, K. & Akira, S. Unresponsiveness of MyD88-deficient mice to endotoxin. Immunity 11, 115–122 (1999).
Yamamoto, M. et al. Role of adapter TRIF in the MyD88-independent Toll-like receptor signaling pathway. Science 301, 640–643 (2003).
Wesemann, D.R., Qin, H., Kokorina, N. & Benveniste, N.E. TRADD interacts with STAT1-α and influences interferon-γ signaling. Nat. Immunol. 5, 199–207 (2004).
Meylan, E. et al. RIP1 is essential mediator of Toll-like receptor 3–induced NF-κB activation. Nat. Immunol. 5, 503–507 (2004).
Cusson-Hermance, N., Khurana, S., Lee, T.H., Fitzgerald, K.A. & Kelliher, M. Rip1 mediates the Trif-dependent Toll-like receptor 3- and 4-induced NF-κB activation but does not contribute to interferon regulatory factor 3 activation. J. Biol. Chem. 280, 36560–36566 (2005).
Michallet, M.-C. TRADD protein is an essential component of the RIG-like helicase antiviral pathway. Immunity 28, 651–661 (2008).
Freshney, I.R. in Culture of Animal Cells 3rd edn. 131–132 (Wiley Liss, New York, 1994).
Acknowledgements
We thank W.C. Yeh and T.W. Mak (University of Toronto, Canada) for Traf2−/− MEFs; H. Nakano (Juntento University, Japan) for Traf5−/− and Traf2−/−Traf5−/− MEFs; L. Tessarollo (US National Cancer Institute) for the pLTM260 vector; A. Singer (US National Cancer Institute) for the pKO Scrambler 917 TK vector; and L. Pobezinsky for suggestions. Supported by the Intramural Research Program of Center for Cancer Research (US National Cancer Institute).
Author information
Authors and Affiliations
Corresponding author
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10, Table 1 and Methods (PDF 3485 kb)
Rights and permissions
About this article
Cite this article
Pobezinskaya, Y., Kim, YS., Choksi, S. et al. The function of TRADD in signaling through tumor necrosis factor receptor 1 and TRIF-dependent Toll-like receptors. Nat Immunol 9, 1047–1054 (2008). https://doi.org/10.1038/ni.1639
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.1639
This article is cited by
-
Immunogenic cell death in cancer: targeting necroptosis to induce antitumour immunity
Nature Reviews Cancer (2024)
-
RNA-Sequencing based analysis of bovine endometrium during the maternal recognition of pregnancy
BMC Genomics (2022)
-
Roles of RIPK3 in necroptosis, cell signaling, and disease
Experimental & Molecular Medicine (2022)
-
PLEKHO2 inhibits TNFα-induced cell death by suppressing RIPK1 activation
Cell Death & Disease (2021)
-
Necroptosis, pyroptosis and apoptosis: an intricate game of cell death
Cellular & Molecular Immunology (2021)