Abstract
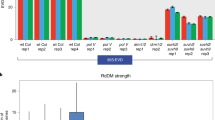
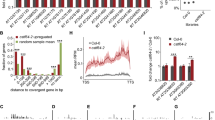
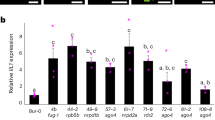
Small RNAs have several important biological functions1. MicroRNAs (miRNAs) and trans-acting small interfering RNAs (tasiRNAs) regulate mRNA stability and translation, and siRNAs cause post-transcriptional gene silencing of transposons, viruses and transgenes and are important in both the establishment and maintenance of cytosine DNA methylation2. Here, we study the role of the four Arabidopsis thaliana DICER-LIKE genes (DCL1–DCL4) in these processes. Sequencing of small RNAs from a dcl2 dcl3 dcl4 triple mutant showed markedly reduced tasiRNA and siRNA production and indicated that DCL1, in addition to its role as the major enzyme for processing miRNAs, has a previously unknown role in the production of small RNAs from endogenous inverted repeats. DCL2, DCL3 and DCL4 showed functional redundancy in siRNA and tasiRNA production and in the establishment and maintenance of DNA methylation. Our studies also suggest that asymmetric DNA methylation can be maintained by pathways that do not require siRNAs.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Meister, G. & Tuschl, T. Mechanisms of gene silencing by double-stranded RNA. Nature 431, 343–349 (2004).
Chan, S.W., Henderson, I.R. & Jacobsen, S.E. Gardening the genome: DNA methylation in Arabidopsis thaliana. Nat. Rev. Genet. 6, 351–360 (2005).
Xie, Z. et al. Genetic and functional diversification of small RNA pathways in plants. PLoS Biol. 2, E104 (2004).
Xie, Z., Allen, E., Wilken, A. & Carrington, J.C. DICER-LIKE 4 functions in trans-acting small interfering RNA biogenesis and vegetative phase change in Arabidopsis thaliana. Proc. Natl. Acad. Sci. USA 102, 12984–12989 (2005).
Yoshikawa, M., Peragine, A., Park, M.Y. & Poethig, R.S. A pathway for the biogenesis of trans-acting siRNAs in Arabidopsis. Genes Dev. 19, 2164–2175 (2005).
Gasciolli, V., Mallory, A.C., Bartel, D.P. & Vaucheret, H. Partially redundant functions of Arabidopsis DICER-like enzymes and a role for DCL4 in producing trans-acting siRNAs. Curr. Biol. 15, 1494–1500 (2005).
Dunoyer, P., Himber, C. & Voinnet, O. DICER-LIKE 4 is required for RNA interference and produces the 21-nucleotide small interfering RNA component of the plant cell-to-cell silencing signal. Nat. Genet. 37, 1356–1360 (2005).
Schauer, S.E., Jacobsen, S.E., Meinke, D.W. & Ray, A. DICER-LIKE1: blind men and elephants in Arabidopsis development. Trends Plant Sci. 7, 487–491 (2002).
Hamilton, A., Voinnet, O., Chappell, L. & Baulcombe, D. Two classes of short interfering RNA in RNA silencing. EMBO J. 17, 4671–4679 (2002).
Qi, Y., Denli, A.M. & Hannon, G.J. Biochemical specialization within Arabidopsis RNA silencing pathways. Mol. Cell 19, 421–428 (2005).
Liu, B. et al. Loss of function of OsDCL1 affects microRNA accumulation and causes developmental defects in rice. Plant Physiol. 139, 296–305 (2005).
Margulies, M. et al. Genome sequencing in microfabricated high-density picolitre reactors. Nature 437, 376–380 (2005).
Allen, E., Xie, Z., Gustafson, A.M. & Carrington, J.C. microRNA-directed phasing during trans-acting siRNA biogenesis in plants. Cell 121, 207–221 (2005).
Allen, E. et al. Evolution of microRNA genes by inverted duplication of target gene sequences in Arabidopsis thaliana. Nat. Genet. 36, 1282–1290 (2004).
Cam, H.P. et al. Comprehensive analysis of heterochromatin- and RNAi-mediated epigenetic control of the fission yeast genome. Nat. Genet. 37, 809–819 (2005).
Li, J., Yang, Z., Yu, B., Liu, J. & Chen, X. Methylation protects miRNAs and siRNAs from a 3′-end uridylation activity in Arabidopsis. Curr. Biol. 15, 1501–1507 (2005).
Yu, B. et al. Methylation as a crucial step in plant microRNA biogenesis. Science 307, 932–935 (2005).
Shen, B. & Goodman, H.M. Uridine addition after microRNA-directed cleavage. Science 306, 997 (2004).
Cao, X. & Jacobsen, S.E. Role of the Arabidopsis DRM methyltransferases in de novo DNA methylation and gene silencing. Curr. Biol. 12, 1138–1144 (2002).
Chan, S.W. et al. RNA silencing genes control de novo DNA methylation. Science 303, 1336 (2004).
Soppe, W.J. et al. The late flowering phenotype of fwa mutants is caused by gain-of-function epigenetic alleles of a homeodomain gene. Mol. Cell 6, 791–802 (2000).
Jacobsen, S.E., Sakai, H., Finnegan, E.J., Cao, X. & Meyerowitz, E.M. Ectopic hypermethylation of flower-specific genes in Arabidopsis. Curr. Biol. 10, 179–186 (2000).
Jackson, J.P., Lindroth, A.M., Cao, X. & Jacobsen, S.E. Control of CpNpG DNA methylation by the KRYPTONITE histone H3 methyltransferase. Nature 416, 556–560 (2002).
Malagnac, F., Bartee, L. & Bender, J. An Arabidopsis SET domain protein required for maintenance but not establishment of DNA methylation. EMBO J. 21, 6842–6852 (2002).
Lu, C. et al. Elucidation of the small RNA component of the transcriptome. Science 309, 1567–1569 (2005).
Yoder, J.A., Soman, N.S., Verdine, G.L. & Bestor, T.H. DNA (cytosine-5)-methyltransferases in mouse cells and tissues. Studies with a mechanism-based probe. J. Mol. Biol. 270, 385–395 (1997).
Pradhan, S., Bacolla, A., Wells, R.D. & Roberts, R.J. Recombinant human DNA (cytosine-5) methyltransferase. I. Expression, purification, and comparison of de novo and maintenance methylation. J. Biol. Chem. 274, 33002–33010 (1999).
Zilberman, D. et al. Role of Arabidopsis ARGONAUTE4 in RNA-directed DNA methylation triggered by inverted repeats. Curr. Biol. 14, 1214–1220 (2004).
Valoczi, A. et al. Sensitive and specific detection of microRNAs by northern blot analysis using LNA-modified oligonucleotide probes. Nucleic Acids Res. 32, e175 (2004).
Acknowledgements
We thank S. Chan for the dcl2 dcl3 double mutant, J. Carrington for drawing our attention to the IR-71 locus and members of the Jacobsen laboratory for critical reading of the manuscript. I.R.H. was supported by a European Molecular Biology Organization (EMBO) Long Term Postdoctoral Fellowship. S.E.J. is an investigator of the Howard Hughes Medical Institute. This work was supported by US National Science Foundation grants 0439186 and 0548569 to P.J.G. and B.C.M., and NIH grant GM60398 and funds from the Howard Hughes Medical Institute to S.E.J.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Repeat structure and siRNA accumulation at the large inverted repeat IR-71. (PDF 272 kb)
Supplementary Fig. 2
Distribution of methylation at repeat loci. (PDF 200 kb)
Supplementary Table 1
Composition of small RNAs sequenced from wild-type Columbia (Col) and the dcl2-1 dcl3-1 dcl4-2 triple mutant. (PDF 55 kb)
Supplementary Table 2
Sodium bisulfite sequencing analysis of repeat loci. (PDF 44 kb)
Supplementary Table 3
Adaptor, primer and probe sequences. (PDF 43 kb)
Supplementary Table 4
Small RNA sequences cloned from Columbia that show a perfect match to the Arabidopsis genome. (PDF 417 kb)
Supplementary Table 5
Small RNA sequences cloned from dcl2 dcl3 dcl4 that show a perfect match to the Arabidopsis genome. (PDF 285 kb)
Rights and permissions
About this article
Cite this article
Henderson, I., Zhang, X., Lu, C. et al. Dissecting Arabidopsis thaliana DICER function in small RNA processing, gene silencing and DNA methylation patterning. Nat Genet 38, 721–725 (2006). https://doi.org/10.1038/ng1804
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ng1804
This article is cited by
-
Differential expression of the genes encoding immune system components in response to Pseudomonas syringae and Pseudomonas aeruginosa in Arabidopsis thaliana
3 Biotech (2024)
-
A cryo-EM structure of KTF1-bound polymerase V transcription elongation complex
Nature Communications (2023)
-
The MOM1 complex recruits the RdDM machinery via MORC6 to establish de novo DNA methylation
Nature Communications (2023)
-
Genome-wide identification, characterization and expression analysis of AGO, DCL, and RDR families in Chenopodium quinoa
Scientific Reports (2023)
-
Conserved and non-conserved RNA–target modules in plants: lessons for a better understanding of Marchantia development
Plant Molecular Biology (2023)