Abstract
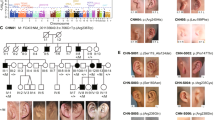
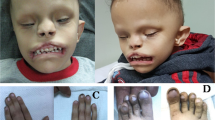
Holoprosencephaly (HPE) is a common developmental defect of the forebrain and frequently the midface in humans, with both genetic and environmental causes. HPE has a prevalence of 1:250 during embryogenesis and 1:16,000 newborn infants, and involves incomplete development and septation of midline structures in the central nervous system (CMS) with a broad spectrum of clinical severity1–3. Alobar HPE, the most severe form which is usually incompatible with postnatal life, involves complete failure of division of the forebrain into right and left hemispheres and is characteristically associated with facial anomalies including cyclopia, a primitive nasal structure (proboscis) and/or midfacial clefting. At the mild end of the spectrum, findings may include microcephaly, mild hypotelorism, single maxillary central incisor and other defects (Fig. 1). This phenotypic variability also occurs between affected members of the same family. The molecular basis underlying HPE is not known, although teratogens, non-random chromosomal anomalies, and familial forms with autosomal dominant and recessive inheritance have been described4. HPE3 on chromosome 7q36 is one of at least four different loci implicated in HPE5–9. Here, we report the identification of human Sonic Hedgehog (SHH) as HPE3 — the first known gene to cause HPE. Analyzing 30 autosomal dominant HPE (ADHPE) families, we found five families that segregate different heterozygous SHH mutations. Two of these mutations predict premature termination of the SHH protein, whereas the others alter highly conserved residues in the vicinity of the alpha-helix-1 motif or signal cleavage site.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Cohen, M.M. Jr., Perspectives on holoprosencephaly, Part I. Epidemiology, genetics, and syndromology. Teratology 40, 211–235 (1989).
Cohen, M.M.,Jr. Perspectives on holoprosencephaly, Part III. Spectra, distinctions, continuities, and discontinuities. Am. J. Med. Genet. 34, 271–288 (1989).
Cohen, M.M.,Jr. & Sulik, K.K. Perspectives on holoprosencephaly. Part II. Central nervous system, craniofacial anatomy, syndrome commentary, diagnostic approach, and experimental studies. J. Craniofac. Genet. Dev. Biol. 12, 196–244 (1992).
Muenke, M. Holoprosencephaly as a genetic model for normal craniofacial development. Sem. Dev. Biol. 5, 293–301 (1994).
Muenke, M. et al. Linkage of a human malformation, familial holoprosencephaly, to chromosome 7 and evidence for genetic heterogeneity. Proc. Nat Acad. Sci. U.S.A. 91, 8102–8106 (1994).
Muenke, M. et al. Physical mapping of the holoprosencephaly critical region in 21q22.3, exclusion of SIM2 as a candidate gene for holoprosencephaly, and mapping of SIM2 to a region of chromosome 21 important for Down syndrome. Am. J. Hum. Genet. 57, 1074–1079 (1995).
Schell, U. et al. Molecular characterization of breakpoints in patients with holoprosencephaly and definition of the HPE2 critical region 2p21. Hum. Mol. Genet. 5, 223–229 (1995).
Gurrieri, F. et al. Physical mapping of the holoprosencephaly critical region of 7q36. Nature Genet. 3, 247–251 (1993).
Overhauser, J. et al. Physical mapping of the holoprosencephaly critical region in 18p11.3. Am. J. Hum. Genet. 57, 1080–1085 (1995).
Belloni, E. et al. Identification of Sonic Hedgehog as a canddiate gene for holoprosencephaly. Nature Genet. 14, 353–356 (1996).
Marigo, V. et al. Cloning, expression, and chromosomal location of SHH and IHH: two human homologues of the Drosophila segment polarity gene Hedgehog. Genomics 28, 44–51 (1995).
Nüsslein-Volhard, C. & Wieschaus, E. Mutations affecting segment number and polarity in Drosophila. Nature 287, 795–801 (1980).
Lee, J.J., von Kessler, D.P., Parks, S. & Beachy, P.A. Secretion and localized transcription suggest a role in positional signaling for products of the segmentation Gene hedgehog. Cell 71, 33–50 (1992).
Ekker, S.C. et al. Distinct expression and shared activities of members of the hedgehog gene of Xenopus laevis. Development 121, 2337–2347 (1995).
Riddle, R.D., Johnson, R.L., Laufer, E. & Tabin, C. Sonic hedgehog mediates the polarizing activity of the ZPA. Cell 75, 1401–1416 (1993).
Echelard, Y. et al. Sonic hedgehog, a member of a family of putative signaling molecules, is implicated in the regulation of CNS polarity. Cell 75, 1417–1430 (1993).
Roelink, H. et al. Floor plate and motor neuron induction by vhh-1, a vertebrate homolog of hedgehog expressed by the notochord. Cell 76, 761–775 (1995).
Krauss, S., Concordet, J.P. & Ingham, P.W. A functionally conserved homolog of the Drosophila segment polarity gene hh is expressed in tissues with polarizing activity in zebrafish embryos. Cell 75, 1431–1444 (1993).
Fan, C.-M., Porter, J.A., Chiang, C., Chang, D.T., Beachy, P.A. & Tessier-Lavigne, M. Long-range sclerotome induction by sonic hedgehog: direct role of the amino-terminal cleavage product and modulation by the cyclic AMP signalling pathway. Cell 81, 457–465 (1995).
Tanabe, Y., Roelink, H. & Jessell, T.M. Induction of motor neurons by Sonic hedgehog independent of floor plate differentiation. Curr. Biol. 5, 651–658 (1995).
Fietz, M.J., Jacinto, A., Taylor, A.M., Alexandre, C. & Ingham, P.W. Secretion of the amino-terminal fragment of the Hedgehog protein is necessary and sufficient for hedgehog signalling in Drosophila. Curr. Biol. 5, 643–649 (1995).
Porter, J.A. et al. The product of hedgehog autoproteolytic cleavage is active in local and long-range signalling. Nature 374, 363–366 (1995).
Marti, E., Bumcrot, D.A., Takada, R. & McMahon, A.P. Requirement of the 19K form of Sonic hedgehog for induction of distinct ventral cell types in CNS explants. Nature 375, 322–325 (1995).
Ericson, J., Muhr, J., Piaczek, M., Lints, T., Jessell, T.M. & Edlund, T. Sonic hedgehog induces the differentiation of ventral forebrain neurons: a common signal for ventral patterning within the neural tube. Cell 81, 747–756 (1995).
Johnson, R.L., Laufer, E., Riddle, R.D. & Tabin, C. Ectopic expression of Sonic hedgehog alters dorsal-ventral patterning of somites. Cell 79, 1165–1173 (1994).
Huang, Z. & Krauss, S. Hedgehog, transmitted along retinal axons, triggers neurogenesis in the developing visual centers of the Drosophila brain. Cell 86, 411–122 (1996).
Macdonald, R., Barth, K.A., Xu, Q., Holder, N., Mikkola, I. & Wilson, S.W. Midline signalling is required for Pax gene regulation and patterning of the eyes. Development 121, 3267–3278 (1995).
Hall, T.M.T., Porter, J.A., Beachy, P.A. & Leahy, D.J. A potential catalytic site revealed by the 1.7-A crystal structure of the amino-terminal signalling domain of Sonic hedgehog. Nature 378, 212–216 (1995).
Chiang, C. et al. Cyclopia and defective axial patterning in mice lacking Sonic hedgehog gene function. Nature 383, 407–413 (1996).
Roelink, H. et al. Floor plate and motor neuron induction by different concentrations of the amino-terminal cleavage product of Sonic hedgehog autoproteolysis. Cell 81, 445–455 (1995).
Hatta, K., Kimmel, C.B., Ho, R.K. & Walker, C. The cyclops mutation blocks specification of the floor plate of the zebrafish central nervous system. Nature 350, 339–341 (1991).
Schulte-Merker, S. van Eeden, F.J.M., Halpern, M.E., Kimmel, C.B. & Nüsslein-Volhard, C. No tail (nt) is the zebrafish homologue of the mouse T (Brachyury) gene. Development 120, 1009–1015 (1994).
Lynch, S.A. et al. A gene for autosomal dominant sacral agenesis maps to the holoprosencephaly region at 7q36. Nature Genet. 11, 93–95 (1995).
Hammerschmidt, M., Bitgood, M.J. & McMahon, A.P. Protein kinase A is a common negative regulator of Hedgehog signaling in the vertebrate embryo. Genes & Devel. 10, 647–658 (1996).
Muenke, M. et al. A common mutation in the fibroblast growth factor receptor 1 gene in Pfeiffer syndrome. Nature Genet. 8, 269–274 (1994).
Ardinger, H.H. & Bartley, J.A. Microcephaly in familial holoprosencephaly. J. Craniofac. Genet. Dev. Biol. 8, 53–61 (1988).
Johnson, V.P. Holoprosencephaly: a developmental field defect. Am J. Med. Genet. 34, 258–264 (1989).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Roessler, E., Belloni, E., Gaudenz, K. et al. Mutations in the human Sonic Hedgehog gene cause holoprosencephaly. Nat Genet 14, 357–360 (1996). https://doi.org/10.1038/ng1196-357
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng1196-357
This article is cited by
-
Non-canonical non-genomic morphogen signaling in anucleate platelets: a critical determinant of prothrombotic function in circulation
Cell Communication and Signaling (2024)
-
Disruption of Sonic Hedgehog Signaling Accelerates Age-Related Neurogenesis Decline and Abolishes Stroke-Induced Neurogenesis and Leads to Increased Anxiety Behavior in Stroke Mice
Translational Stroke Research (2022)
-
Aniridia-related keratopathy relevant cell signaling pathways in human fetal corneas
Histochemistry and Cell Biology (2022)
-
Forebrain Shh overexpression improves cognitive function and locomotor hyperactivity in an aneuploid mouse model of Down syndrome and its euploid littermates
Acta Neuropathologica Communications (2021)
-
Association of Sonic Hedgehog with the extracellular matrix requires its zinc-coordination center
BMC Molecular and Cell Biology (2021)