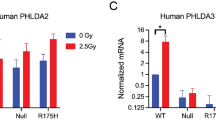
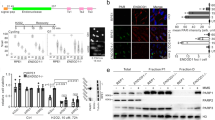
Abstract
Mutations in atm and p53 cause the human cancer-associated diseases ataxia-telangiectasia1 and Li-Fraumeni syndrome2,3, respectively. The two genes are believed to interact in a number of pathways4–6, including regulation of DNA damage–induced cell-cycle checkpoints7, apoptosis and radiation sensitivity8, and cellular proliferation9. Atm-null mice10–12, as well as those null for p5313,14, develop mainly T-cell lymphomas, supporting the view that these genes have similar roles in thymocyte development. To study the interactions of these two genes on an organismal level, we bred mice heterozygous for null alleles of both atm and p53 to produce all genotypic combinations. Mice doubly null for atm and p53 exhibited a dramatic acceleration of tumour formation relative to singly null mice, indicating that both genes collaborate in a significant manner to prevent tumorigenesis. With respect to their roles in apoptosis, loss of atm rendered thymocytes only partly resistant to irradiation-induced apoptosis, whereas additional loss of p53 engendered complete resistance. This implies that the irradiation-induced atm and p53 apoptotic pathways are not completely congruent. Finally—and in contrast to prior predictions4,6—atm and p53 do not appear to interact in acute radiation toxicity, suggesting a separate atm effector pathway for this DNA damage response and having implications for the prognosis and treatment of human tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Savitsky, K. et al. A single ataxia telangiectasia gene with a product similar to PI-3 kinase. Science 268, 1749–1753 (1995).
Srivastava, S., Zou, Z.Q., Pirollo, K., Blattner, W. & Chang, E.H. Germ-line transmission of a mutated p53 gene in a cancer-prone family with Li-Fraumeni syndrome. Nature 348, 747–749 (1990).
Malkin, D. et al. Germ line p53 mutations in a familial syndrome of breast cancer, sarcomas, and other neoplasms. Science 250, 1233–1238 (1990).
Meyn, M.S. Ataxia-telangiectasia and cellular responses to DNA damage. Cancer Res. 55, 5991–6001 (1995).
Hawley, R.S. & Friend, S.H. Strange bedfellows in even stranger places: the role of ATM in meiotic cells, lymphocytes, tumors, and its functional links to p53. Genes Dev. 10, 2383–2388 (1996).
Enoch, T. & Norbury, C. Cellular responses to DNA damage: cell-cycle checkpoints, apoptosis and the roles of p53 and ATM. Trends Biochem. Sci. 20, 426–430 (1995).
Kastan, M.B. et al. A mammalian cell cycle checkpoint pathway utilizing p53 and GADD45 is defective in ataxia-telangiectasia. Cell 71, 587–597 (1992).
Meyn, M.S., Strasfeld, L. & Allen, C. Testing the role of p53 in the expression of genetic instability and apoptosis in ataxia-telangiectasia. Int. J. Radiat. Biol. 66, S141–S149 (1994).
Westphal, C.H. et al. Genetic interactions between atm and p53 influence cellular proliferation and irradiation-induced cell cycle checkpoints. Cancer Res. 57, 1664–1667 (1997).
Xu, Y. & Baltimore, D. Dual roles of ATM in the cellular response to radiation and in cell growth control. Genes Dev. 10, 2401–2410 (1996).
Elson, A. et al. Pleiotropic defects in ataxia-telangiectasia protein-deficient mice. Proc. Natl. Acad. Sci. USA 93, 13084–13089 (1996).
Barlow, C. et al. Atm-deficient mice: a paradigm of ataxia telangiectasia. Cell 86, 159–171 (1996).
Donehower, L.A. et al. Mice deficient for p53 are developmentally normal but susceptible to spontaneous tumours. Nature 356, 215–221 (1992).
Jacks, T. et al. Tumor spectrum analysis in p53-mutant mice. Curr. Biol. 4, 1–7 (1994).
Clarke, A.R. et al. Thymocyte apoptosis induced by p53-dependent and independent pathways. Nature 362, 849–852 (1993).
Lowe, S.W., Schmitt, E.M., Smith, S.W., Osborne, B.A. & Jacks, T. p53 is required for radiation-induced apoptosis in mouse thymocytes. Nature 362, 847–849 (1993).
Lu, X. & Lane, D.P. Differential induction of transcriptionally active p53 following UV or ionizing radiation: defects in chromosome instability syndromes. Cell 75, 765–778 (1993).
Canman, C.E., Wolff, A.C., Chen, C.Y., Fornace, A.J., Jr. & Kastan, M.B. The p53-dependent G1 cell cycle checkpoint pathway and ataxia- telangiectasia. Cancer Res. 54, 5054–5058 (1994).
Lavin, M.F. & Shiloh, Y. The genetic defect in ataxia-telangiectasia. Annu. Rev. Immunol. 15, 177–202 (1996).
Donehower, L.A. et al. Effects of genetic background on tumorigenesis in p53-deficient mice. Mol. Carcinog. 14, 16–22 (1995).
Gotoff, S.P., Amirmokri, E. & Liebner, E.J. Ataxia telangiectasia: neoplasia, untoward response to X-irradiation, and tuberous sclerosis. Am. J. Dis. Child. 114, 617–625 (1967).
Cunliffe, P.M., Mann, J.R., Cameron, A.H. & Roberts, K.D. Radiosensitivity in ataxia-telangiectasia. Br. J. Radiol. 48, 374–376 (1975).
Morgan, J.L., Holcomb, T.M. & Morissey, R.W. Radiation reaction in ataxia telangiectasia. Am. J. Dis. Child. 116, 557–559 (1968).
Morgan, S.E., Lovly, C., Pandita, T.K., Shiloh, Y. & Kastan, M.B. Fragments of atm which have dominant-negative or complementing activity. Mol. Cell. Biol. 17, 2020–2029 (1997).
Fisher, D.E. Apoptosis in cancer therapy: crossing the threshold. Cell 78, 539–542 (1994).
Jacks, T. & Weinberg, R.A. Cell-cycle control and its watchman. Nature 381, 643–644 (1996).
Lowe, S.W., Ruley, H.E., Jacks, T. & Housman, D.E. p53-dependent apoptosis modulates the cytotoxicity of anticancer agents. Cell 74, 957–967 (1993).
Lowe, S.W. et al. p53 status and the efficacy of cancer therapy in vivo. Science 266, 807–810 (1994).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Westphal, C., Rowan, S., Schmaltz, C. et al. atm and p53 cooperate in apoptosis and suppression of tumorigenesis, but not in resistance to acute radiation toxicity. Nat Genet 16, 397–401 (1997). https://doi.org/10.1038/ng0897-397
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0897-397
This article is cited by
-
Histological analysis of sleep and circadian brain circuitry in cranial radiation-induced hypersomnolence (C-RIH) mouse model
Scientific Reports (2022)
-
AIMP3 Deletion Induces Acute Radiation Syndrome-like Phenotype in Mice
Scientific Reports (2018)
-
Synergy between Prkdc and Trp53 regulates stem cell proliferation and GI-ARS after irradiation
Cell Death & Differentiation (2017)
-
Pharmacologic ATM but not ATR kinase inhibition abrogates p21-dependent G1 arrest and promotes gastrointestinal syndrome after total body irradiation
Scientific Reports (2017)
-
ATM signalling and cancer
Oncogene (2014)