Abstract
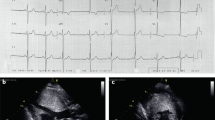
Familial hypertrophic cardiomyopathy (FHC) is caused by missense mutations in the β cardiac myosin heavy chain (MHC) gene in less than half of affected individuals. To identify the location of another gene involved in this disorder, a large family with FHC not linked to the β MHC gene was studied. Linkage was detected between the disease in this family and a locus on chromosome 1q3 (maximum multipoint lod score = 8.47). Analyses in other families with FHC not linked to the β MHC gene, revealed linkage to the chromosome 1 locus in two and excluded linkage in six. Thus mutations in at least three genetic loci can cause FHC. Three sarcomeric contractile proteins — troponin I, tropomyosin and actin — are strong candidate FHC genes at the chromosome 1 locus.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Jarcho, J.A. et al. Mapping a gene for familial hypertrophic cardiomyopathy to chromosome 14q1. New Eng. J. Med. 321, 1372–1378 (1989).
Solomon, S.D. et al. Familial hypertrophic cardiomyopathy is a genetically heterogeneous disease. J. clin. Invest. 86, 993–939 (1990).
Maron, B.J., Bonow, R.O., Cannon, R.O., Leon, M.B. & Epstein, S.E. Hypertrophic cardiomyopathy: interrelations of clinical manifestations, pathophysiology, and therapy. New Engl. J. Med. 316, 780–789 (1987).
McKenna, W.J. Hypertrophic cardiomyopathy. In Diseases of the Heart, (eds Julian, D.G., Camm, A.J., Fox, K.M., Hall, R.J.C., Poole-Wilson. P.A.) (Bailliere Tindall, London, 1989).
Watkins, H. et al. Characteristics and prognostic significance of myosin missense mutations in familial hypertrophic cardiomyopathy. New Engl. J. Med. 326, 1108–1114 (1992).
Geisterfer-Lowrance, A.A.T. et al. A molecular basis for familial hypertrophic cardiomyopathy: A β cardiac myosin heavy chain gene missense mutation. Cell 62, 999–1006 (1990).
Schwartz, K. et al. Exclusion of cardiac myosin heavy chain and actin gene involvement in hypertrophic cardiomyopathy of several French families. Circ. Res. 71, 3–8 (1992).
Kurabayashi, M., Komuro, I., Tsuchimochi, H., Takaku, F. & Yazaki, Y. Molecular cloning and characterization of human atrial and ventricular myosin alkali light chain cDNA clones. J. biol. Chem. 263, 13930–13936 (1988).
Weber, J.L. & May, P.E. Abundant class of human DNA polymorphisms which can be typed using the polymerase chain reaction. Am. J. hum. Genet. 44, 388–396 (1989).
Litt, M. & Luty, J.A. A hypervariable microsatellite revealed by in vitro amplification of a dinucleotide repeat within the cardiac muscle actin gene. Am. J. hum. Genet. 44, 397–401 (1989).
Macera, M.J., Szabo, P., Wadgaonkar, R., Siddiqui, M.A.Q. & Verma, R.S. Localization of the gene coding for ventricular myosin regulatory light chain (MYL2) to chromosome 12 q23-q24.3. Genomics 13, 829–831 (1992).
Webb, G.C., Coggan, M., Ichinose, A. & Board, P.G. Localization of the factor XIIIB subunit gene (F13B) to chromosomal bands 1q31-32. 1 and restriction fragment polymorphism at the locus. Hum. Genet. 81, 157–160 (1989).
Nishimura, D.Y. & Murray, J.C. A tetranucleotide repeat for the F13B locus. Nucl. Acids Res. 20, 1167 (1992).
Cox, D.W., Nakamura, Y. & Gedde-Dahl, T. Report of the committee on the genetic constitution of chromosome 14. Cytogenet. Cell Genet. 55, 183–188 (1990).
Dracopoli, N.C. et al. The CEPH consortium linkage map of human chromosome 1. Genomics 9, 686–700 (1991).
Collins, A., Keats, B.J., Dracopoli, N., Shields, D.C. & Motion, N.E. Integration of gene maps: Chromosome 1. Proc. natn. Acad. Sci. U.S.A. 89, 4598–4602 (1992).
Edwards, A., Hammond, H.A., Jin, L., Caskey, C.T. & Chakraborty, R. Genetic variation at five trimeric and tetrameric tandem repeat loci in four human population groups. Genomics 12, 241–253 (1992).
Nishimura, D.Y., Leysens, N.J. & Murray, J.C. A dinucleotide repeat for the D1S53 locus. Nucl. Acids Res. 20, 1167 (1992).
Epstein, N.E., Cohn, G.M., Cyran, F. & Fananapazir, L. Differences in clinical expression of hypertrophic cardiomyopathy associated with two distinct mutations in the β-myosin heavy chain gene. Circulation 86, 345–352 (1992).
Gunning, P., Ponte, P., Kedes, L., Eddy, R. & Shows, T. Chromosomal location of the co-expressed human skeletal and cardiac actin genes. Proc. natn. Acad. Sci. U.S.A. 81, 1813–1817 (1984).
Boheler, K.R. et al. Skeletal actin mRNA increases in the human heart during ontogenic development and is the major isoform of control and failing adult hearts. J. clin. Invest. 88, 323–30 (1991).
Bakerman, P.R., Stenmark, K.R. & Fisher, J.H. α-skeletal actin messenger RNA increases in acute right ventricular hypertrophy. Am. J. Physiol. 258, L173–L178 (1990).
Radice, P. The human tropomyosin gene involved in the generation of the TRK oncogene maps to chromosome 1q31. Oncogene 6, 2145–2148 (1991).
Reinach, F.C. & MacLeod, A.R. Tissue-specific expression of the human tropomyosin gene involved in the generation of the trk oncogene. Nature 322, 648–650 (1986).
Wade, R., Eddy, R., Shows, T.B. & Kedes, L. cDNA sequence, tissue-specific expression, and chromosomal mapping of the human slow-twitch skeletal muscle isoform of troponin I. Genomics 7, 346–367 (1990).
Bhavsar, P.K. et al. Developmental expression of troponin I isoforms in fetal human heart. FEBS Letters 292, 5–8 (1991).
Hunkeler, N.M., Kullman, J. & Murphy, A.M. Troponin I isoform expression in human heart. Circ. Res. 69, 1409–1414 (1991).
Rosenzweig, A. et al. Preclinical diagnosis of familial hypertrophic cardiomyopathy by genetic analysis of blood lymphocytes. New Engl. J. Med. 326, 1753–1760 (1991).
Watkins, H. et al. Sporadic hypertrophic cardiomyopathy due to De Novo myosin mutations. J. clin. Invest. 90, 1666–1671 (1992).
Chou, Y-H., et al. The gene responsible for familial hypocalciuric hypercalcaemia maps to chromosome 3q in four unrelated families. Nature Genet. 1, 295–300 (1992).
Polymeropoulos, M.H., Xiao, H., Rath, D.S. & Merrill, C.R. Dinucleotide repeat polymorphism at the human cardiac beta-myosin gene. Nucl. Acids Res. 19, 4019 (1991).
Lathrop, G.M., Lalouel, J.M., Julier, C. & Ott, J. Strategies for multilocus linkage analysis in humans. Proc. natn. Acad. Sci. U.S.A. 81, 3443–3446 (1984).
Ott, J. Analysis of human linkage 2nd edn (The Johns Hopkins University Press, Baltimore, 1992).
Kaplan, E.L. & Meier, P. Nonparametric estimation from incomplete observations. J. Am. Statist. Assoc. 53, 457–481 (1958).
Lee, E.T. Statistical methods for survival data analysis. (Lifetime Learning Publications, Belmont: 1980).
Cox, D.R., Oakes, D. Analysis of survival data. (Chapman and Hall, New York, 1984).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Watkins, H., MacRae, C., Thierfelder, L. et al. A disease locus for familial hypertrophic cardiomyopathy maps to chromosome 1q3. Nat Genet 3, 333–337 (1993). https://doi.org/10.1038/ng0493-333
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0493-333
This article is cited by
-
Predictive genomic tools in disease stratification and targeted prevention: a recent update in personalized therapy advancements
EPMA Journal (2022)
-
Personalized medicine in cardiovascular disease: review of literature
Journal of Diabetes & Metabolic Disorders (2021)
-
Genetic, clinical, molecular, and pathogenic aspects of the South Asian–specific polymorphic MYBPC3Δ25bp variant
Biophysical Reviews (2020)
-
When signalling goes wrong: pathogenic variants in structural and signalling proteins causing cardiomyopathies
Journal of Muscle Research and Cell Motility (2017)