Abstract
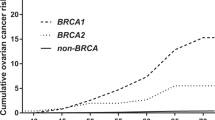
Women who carry a mutation in the BRCA1 gene (on chromosome 17q21), have an 80% risk of breast cancer and a 40% risk of ovarian cancer by the age of 70 (ref. 1). The variable penetrance of BRCA1 suggests that other genetic and non-genetic factors play a role in tumourigenesis in these individuals. The HRAS1 variable number of tandem repeats (VNTR) polymorphism, located 1 kilobase (kb) downstream of the HRAS1 proto-oncogene (chromosome 11p15.5 ) is one possible genetic modifier of cancer penetrance. Individuals who have rare alleles of this VNTR have an increased risk of certain types of cancers, including breast cancer2–4. To investigate whether the presence of rare HRAS1 alleles increases susceptibility to hereditary breast and ovarian cancer, we have typed a panel of 307 female BRCA1 carriers at this locus using a PCR-based technique. The risk for ovarian cancer was 2.11 times greater for BRCA1 carriers harbouring one or two rare HRAS1 alleles, compared to carriers with only common alleles (P = 0.015). The magnitude of the relative risk associated with a rare HRAS1 allele was not altered by adjusting for the other known risk factors for hereditary ovarian cancer5. Susceptibility to breast cancer did not appear to be affected by the presence of rare HRAS1 alleles. This study is the first to show the effect of a modifying gene on the penetrance of an inherited cancer syndrome.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Easton, D., Ford, D., Bishop, D. & Consortium, B.C.L. Breast and Ovarian cancer incidence in BRCA1- mutation carriers. Am. J. Hum. Genet. 56, 265–271 (1995).
Lidereau, R. et al. High frequency of rare alleles of the human c-Ha-ras-1 proto-oncogene in breast cancer patients. J. Natl. Cancer Inst. 77, 697–701 (1986).
Krontiris, T., Devlin, B, Karp, D., Robert, N. & Risch, N. An association between the risk of cancer and mutations in the HRAS1 minisatellite locus. New Eng. J. Med. 329, 517–523 (1993).
Garret, P., Hulka, B., Kim, Y. & Farber, R. HRAS protooncogene polymorphism and breast cancer. Cancer Epi. Biomarkers Prevention 2, 131–138 (1993).
Narod, S. et al. Risk modifiers in carriers of BRCA1 mutations. Int. J. Cancer 64, 394–398 (1995).
Simard, J. et al. Common origins of BRCA1 mutations in Canadian breast and ovarian cancer families. Nature Genet. 8, 392–398 (1995).
Serova, O. et al. A high incidence of BRCA1 mutations in 20 breast-ovarian cancer families. Am. J. Hum. Genet. 58, 42–51 (1996).
Shattuck-Eidens, D. et al. A collaborative survey of 80 mutations in the BRCA1 breast and ovarian cancer susceptibility gene. JAMA 273, 535–541 (1995).
Hogervorst, F. et al. Rapid detection of BRCA1 mutations by the protein truncation test. Nature Genet. 10, 208–212 (1995).
Castilla, L. et al.Mutations in the BRCA1 gene in families with early-onset breast and ovarian cancer.. Nature Genet. 8, 387–391 (1995).
Capon, D., Chen, E., Levinson, A., Seeburg, P. & Goeddel, D. Complete nucleotide sequence of the T24 human bladder carcinoma oncogene and it's normal homologue. Nature 302, 33–37 (1983).
Cox, D. Regression models and life tables. J. R. Stat Soc. 1972 34, 187–220 (1972).
Trepicchio, W. & Krontiris, T. Members of the rel/NF-kB family of transcriptional regulatory factors bind the HRAS1 minisatellite DNA sequence. Nucl. Acids Res. 20, 2427–2434 (1992).
Green, M. & Krontiris, T. Allelic variation of reporter gene activation by the HRAS1 minisatellite. Genomics 17, 429–434 (1993).
Spandidos, D. & Holmes, L. Transcriptional enhancer activity in the variable tandem repeat DNA sequence downstream of the human Ha-ras-1 gene. FEBS Lett. 218, 41–46 (1987).
Ryberg, D., Lindstedt, B., Zienolddiny, S. & Haugen, A. A herditary genetic marker closely associated with microsatellite instability in lung cancer.Cancer Res. 55, 3996–3999 (1995).
Narod, S. et al. An evaluation of genetic heterogeneity in 145 breast-ovarian cancer families. Am. J. Hum. Genet. 56, 254–264 (1995).
Decorte, R., Cuppens, H., Marynen, P. & Cassiman, J.-J. Rapid detection of hypervariable regions by the polymerase chain reaction technique. DNA Cell Biol. 9, 461–469 (1990).
Ardern, J. et al. Polymerase chain reaction analysis of allele frequency and loss at the Harvey Ras locus in myeloid malignancies. Leukemia 7, 258–262 (1993).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Phelan, C., Rebbeck, T., Weber, B. et al. Ovarian cancer risk in BRCA1 carriers is modified by the HRAS1 variable number of tandem repeat (VNTR) locus. Nat Genet 12, 309–311 (1996). https://doi.org/10.1038/ng0396-309
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ng0396-309
This article is cited by
-
Serum levels of IGF-I and BRCA penetrance: a case control study in breast cancer families
Familial Cancer (2011)
-
Gene expression profiles and breast cancer metastasis: a genetic perspective
Clinical & Experimental Metastasis (2009)
-
Haplotype analysis of TP53 polymorphisms, Arg72Pro and Ins16, in BRCA1 and BRCA2 mutation carriers of French Canadian descent
BMC Cancer (2008)
-
MDM2 SNP309 accelerates breast and ovarian carcinogenesis in BRCA1 and BRCA2 carriers of Jewish–Ashkenazi descent
Breast Cancer Research and Treatment (2008)
-
Epigenetic regulation of RhoB loss of expression in lung cancer
BMC Cancer (2007)