Abstract
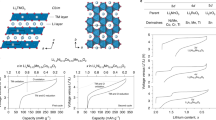
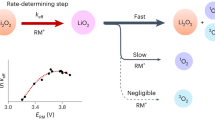
At the cathode of a Li–O2 battery, O2 is reduced to Li2O2 on discharge, the process being reversed on charge. Li2O2 is an insulating and insoluble solid, leading ultimately to low rates, low capacities and early cell death if formed on the cathode surface. Here we show that when using dual mediators, 2,5-Di-tert-butyl-1,4-benzoquinone [DBBQ] on discharge and 2,2,6,6-tetramethyl-1-piperidinyloxy [TEMPO] on charge, the electrochemistry at the cathode surface is decoupled from Li2O2 formation/decomposition in solution. Capacities of 2 mAh cmareal−2 at 1 mA cmareal−2 with low polarization on charge/discharge are demonstrated, and up to 40 mAh cmareal−2 at rates ≫1 mA cmareal−2 are anticipated if suitable gas diffusion electrodes can be devised. One of the major barriers to the progress of Li–O2 cells is decomposition of the carbon cathode. By forming/decomposing Li2O2 in solution and avoiding high charge potentials, the carbon instability is significantly mitigated (<0.008% decomposition per cycle compared with 0.12% without mediators).
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Khetan, A., Luntz, A. & Viswanathan, V. Trade-offs in capacity and rechargeability in nonaqueous Li–O2 batteries: solution-driven growth versus nucleophilic stability. J. Phys. Chem. Lett. 6, 1254–1259 (2015).
Johnson, L. et al. The role of LiO2 solubility in O2 reduction in aprotic solvents and its consequences for Li–O2 batteries. Nat. Chem. 6, 1091–1099 (2014).
McCloskey, B. D. et al. Twin problems of interfacial carbonate formation in nonaqueous Li–O2 batteries. J. Phys. Chem. Lett. 3, 997–1001 (2012).
Viswanathan, V. et al. Electrical conductivity in Li2O2 and its role in determining capacity limitations in non-aqueous Li–O2 batteries. J. Chem. Phys. 135, 214704 (2011).
Abraham, K. M. Prospects and limits of energy storage in batteries. J. Phys. Chem. Lett. 6, 830–844 (2015).
Gallagher, K. G. et al. Quantifying the promise of lithium–air batteries for electric vehicles. Energy Environ. Sci. 7, 1555–1563 (2014).
Grande, L. et al. The lithium/air battery: still an emerging system or a practical reality? Adv. Mater. 27, 784–800 (2015).
Feng, N., He, P. & Zhou, H. Critical challenges in rechargeable aprotic Li–O2 batteries. Adv. Energy Mater. 6, 1502303 (2016).
Imanishi, N., Luntz, A. C. & Bruce, P. G. The Lithium Air Battery: Fundamentals (Springer, 2014).
Lu, J. et al. Aprotic and aqueous Li–O2 batteries. Chem. Rev. 114, 5611–5640 (2014).
Lu, Y. C. et al. Lithium–oxygen batteries: bridging mechanistic understanding and battery performance. Energy Environ. Sci. 6, 750–768 (2013).
Luntz, A. C. & McCloskey, B. D. Nonaqueous Li–air batteries: a status report. Chem. Rev. 114, 11721–11750 (2014).
Black, R., Adams, B. & Nazar, L. F. Non-aqueous and hybrid Li–O2 batteries. Adv. Energy Mater. 2, 801–815 (2012).
Choi, J. W. & Aurbach, D. Promise and reality of post-lithium-ion batteries with high energy densities. Nat. Rev. Mater. 1, 16013 (2016).
Christensen, J. et al. A critical review of Li/air batteries. J. Electrochem. Soc. 159, R1–R30 (2012).
Aurbach, D., McCloskey, B. D., Nazar, L. F. & Bruce, P. G. Advances in understanding mechanisms underpinning lithium–air batteries. Nat. Energy 1, 16128 (2016).
Lu, J. et al. A lithium–oxygen battery based on lithium superoxide. Nature 529, 377–382 (2016).
Jian, Z. et al. Core–shell-structured CNT@RuO2 composite as a high-performance cathode catalyst for rechargeable Li–O2 batteries. Angew. Chem. Int. Edn 53, 442–446 (2014).
Wu, F. et al. Facile synthesis of boron-doped rGO as cathode material for high energy Li–O2 batteries. ACS Appl. Mater. Interfaces 8, 23635–23645 (2016).
Ottakam Thotiyl, M. M. et al. A stable cathode for the aprotic Li–O2 battery. Nat. Mater. 12, 1050–1056 (2013).
Kundu, D., Black, R., Berg, E. J. & Nazar, L. F. A highly active nanostructured metallic oxide cathode for aprotic Li–O2 batteries. Energy Environ. Sci. 8, 1292–1298 (2015).
Aetukuri, N. B. et al. Solvating additives drive solution-mediated electrochemistry and enhance toroid growth in non-aqueous Li–O2 batteries. Nat. Chem. 7, 50–56 (2015).
Burke, C. M., Pande, V., Khetan, A., Viswanathan, V. & McCloskey, B. D. Enhancing electrochemical intermediate solvation through electrolyte anion selection to increase nonaqueous Li–O2 battery capacity. Proc. Natl Acad. Sci. USA 112, 9293–9298 (2015).
Gao, X., Chen, Y., Johnson, L. & Bruce, P. G. Promoting solution phase discharge in Li–O2 batteries containing weakly solvating electrolyte solutions. Nat. Mater. 15, 882–888 (2016).
Sun, D. et al. A solution-phase bifunctional catalyst for lithium–oxygen batteries. J. Am. Chem. Soc. 136, 8941–8946 (2014).
Bergner, B. J., Schurmann, A., Peppler, K., Garsuch, A. & Janek, J. TEMPO: a mobile catalyst for rechargeable Li–O2 batteries. J. Am. Chem. Soc. 136, 15054–15064 (2014).
Lim, H. D. et al. Superior rechargeability and efficiency of lithium–oxygen batteries: hierarchical air electrode architecture combined with a soluble catalyst. Angew. Chem. Int. Edn 53, 3926–3931 (2014).
Kundu, D., Black, R., Adams, B. & Nazar, L. F. A Highly active low voltage redox mediator for enhanced rechargeability of Lithium–oxygen batteries. ACS Central Sci. 1, 510–515 (2015).
Chen, Y., Freunberger, S. A., Peng, Z., Fontaine, O. & Bruce, P. G. Charging a Li–O2 battery using a redox mediator. Nat. Chem. 5, 489–494 (2013).
Lim, H.-D. et al. Rational design of redox mediators for advanced Li–O2 batteries. Nat. Energy 1, 16066 (2016).
Kwak, W.-J. et al. Li–O2 cells with LiBr as an electrolyte and a redox mediator. Energy Environ. Sci. 9, 2334–2345 (2016).
Bryantsev, V. S. et al. Predicting the electrochemical behavior of lithium nitrite in acetonitrile with quantum chemical methods. J. Am. Chem. Soc. 136, 3087–3096 (2014).
Kwak, W.-J. et al. Understanding the behavior of Li–oxygen cells containing LiI. J. Mater. Chem. A 3, 8855–8864 (2015).
Chase, G. V. et al. Soluble oxygen evolving catalysts for rechargeable metal–air batteries. US patent 20120028137 A1 (2012).
Feng, N., He, P. & Zhou, H. Enabling catalytic oxidation of Li2O2 at the liquid–solid interface: the evolution of an aprotic Li–O2 battery. ChemSusChem 8, 600–602 (2015).
Liang, Z. & Lu, Y. C. Critical role of redox mediator in suppressing charging instabilities of lithium–oxygen batteries. J. Am. Chem. Soc. 138, 7574–7583 (2016).
Zhu, Y. G., Wang, X., Jia, C., Yang, J. & Wang, Q. Redox-mediated ORR and OER reactions: redox flow lithium oxygen batteries enabled with a pair of soluble redox catalysts. ACS Catal. 6, 6191–6197 (2016).
Ottakam Thotiyl, M. M., Freunberger, S. A., Peng, Z. & Bruce, P. G. The carbon electrode in nonaqueous Li–O2 cells. J. Am. Chem. Soc. 135, 494–500 (2013).
Freunberger, S. A. et al. The lithium–oxygen battery with ether-based electrolytes. Angew. Chem. Int. Edn 50, 8609–8613 (2011).
Bergner, B. J. et al. Understanding the fundamentals of redox mediators in Li–O2 batteries: a case study on nitroxides. Phys. Chem. Chem. Phys. 17, 31769–31779 (2015).
Bergner, B. J. et al. How to improve capacity and cycling stability for next generation Li–O2 batteries: approach with a solid electrolyte and elevated redox mediator concentrations. ACS Appl. Mater. Interfaces 8, 7756–7765 (2016).
Grübl, D., Bergner, B., Schröder, D., Janek, J. & Bessler, W. G. Multistep reaction mechanisms in nonaqueous lithium–oxygen batteries with redox mediator: a model-based study. J. Phys. Chem. C 120, 24623–24636 (2016).
Hojberg, J. et al. An electrochemical impedance spectroscopy investigation of the overpotentials in Li–O2 batteries. ACS Appl. Mater. Interfaces 7, 4039–4047 (2015).
Freunberger, S. A. et al. Reactions in the rechargeable lithium–O2 battery with alkyl carbonate electrolytes. J. Am. Chem. Soc. 133, 8040–8047 (2011).
Adams, B. D. et al. Towards a stable organic electrolyte for the lithium oxygen battery. Adv. Energy Mater. 5, 1400867 (2015).
McCloskey, B. D. et al. Combining accurate O2 and Li2O2 assays to separate discharge and charge stability limitations in nonaqueous Li–O2 batteries. J. Phys. Chem. Lett. 4, 2989–2993 (2013).
Yang, S., He, P. & Zhou, H. Exploring the electrochemical reaction mechanism of carbonate oxidation in Li–air/CO2 battery through tracing missing oxygen. Energy Environ. Sci. 9, 1650–1654 (2016).
McCloskey, B. D., Garcia, J. M. & Luntz, A. C. Chemical and electrochemical differences in nonaqueous Li–O2 and Na–O2 batteries. J. Phys. Chem. Lett. 5, 1230–1235 (2014).
Lepoivre, F., Grimaud, A., Larcher, D. & Tarascon, J.-M. Long-time and reliable gas monitoring in Li–O2 batteries via a Swagelok derived electrochemical cell. J. Electrochem. Soc. 163, A923–A929 (2016).
Schwenke, K. U., Metzger, M., Restle, T., Piana, M. & Gasteiger, H. A. The influence of water and protons on Li2O2 crystal growth in aprotic Li–O2 cells. J. Electrochem. Soc. 162, A573–A584 (2015).
Hartmann, P. et al. A rechargeable room-temperature sodium superoxide (NaO2) battery. Nat. Mater. 12, 228–232 (2013).
Chen, Y., Freunberger, S. A., Peng, Z., Barde, F. & Bruce, P. G. Li–O2 battery with a dimethylformamide electrolyte. J. Am. Chem. Soc. 134, 7952–7957 (2012).
Bard, A. J. & Faulkner, L. R. Electrochemical Methods. Fundamentals and Applications 2 edn (Wiley, 2000).
Hartmann, P. et al. A comprehensive study on the cell chemistry of the sodium superoxide (NaO2) battery. Phys. Chem. Chem. Phys. 15, 11661–11672 (2013).
Bruce, P. G. et al. Raw Data used to Generate Fig. 4 and Supplementary Fig. 7 (Oxford Research Archive, 2017); http://dx.doi.org/10.5287/bodleian:JNGX5EGnM
Acknowledgements
P.G.B. is indebted to the EPSRC, including the SUPERGEN programme, for financial support.
Author information
Authors and Affiliations
Contributions
X.G. and Y.C. designed experiments and analysed the data. X.G. performed electrochemical performance tests and characterizations with contributions from Z.P.J. Y.C. performed the 13C-carbon experiments and analysed the data. P.G.B., X.G., Y.C., L.R.J. and Z.P.J. interpreted the data. P.G.B. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures 1–8 (PDF 1130 kb)
Rights and permissions
About this article
Cite this article
Gao, X., Chen, Y., Johnson, L. et al. A rechargeable lithium–oxygen battery with dual mediators stabilizing the carbon cathode. Nat Energy 2, 17118 (2017). https://doi.org/10.1038/nenergy.2017.118
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/nenergy.2017.118
This article is cited by
-
Why charging Li–air batteries with current low-voltage mediators is slow and singlet oxygen does not explain degradation
Nature Chemistry (2023)
-
Triarylmethyl cation redox mediators enhance Li–O2 battery discharge capacities
Nature Chemistry (2023)
-
Designing a photo-assisted Co-C3N4 cathode for high performance Li-O2 batteries
Nano Research (2023)
-
Addressing Transport Issues in Non-Aqueous Li–air Batteries to Achieving High Electrochemical Performance
Electrochemical Energy Reviews (2023)
-
Threshold potentials for fast kinetics during mediated redox catalysis of insulators in Li–O2 and Li–S batteries
Nature Catalysis (2022)