Abstract
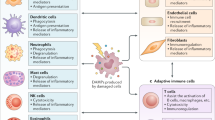
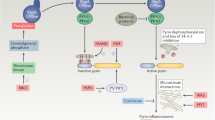
Innate immunity achieves our primary host defense by recognizing invading microorganisms through pathogen-associated molecular patterns (PAMPs) and by reacting to tissue damage signals called damage-associated molecular patterns (DAMPs). DAMP molecules, including high mobility group box 1 protein (HMGB-1), heat-shock proteins (HSPs), uric acid, altered matrix proteins, and S100 proteins, represent important danger signals that mediate inflammatory responses through the receptor for advanced glycation end-products (RAGE, also known as AGER) and Toll-like receptors, after release from activated or necrotic cells. The terms 'alarmins' and 'endokines' have also been proposed for DAMP molecules. A prototypic DAMP molecule, the nuclear protein HMGB-1, is either passively released by necrotic cells or actively secreted with delay by activated cells. S100A8, S100A9, and S100A12 are calcium-binding proteins expressed in the cytoplasm of phagocytes. They are rapidly secreted by activated monocytes or neutrophils, which are abundant in inflamed synovial tissue. HSPs are involved in the crosstalk between innate and adaptive immune systems, and primarily mediate immune regulatory functions. Multiple positive feedback loops between DAMPs and PAMPs and their overlapping receptors temporally and spatially drive these processes and may represent the molecular basis for the observation that infections, as well as nonspecific stress factors, can trigger flares in rheumatic diseases.
Key Points
-
The innate immune system, as well as the adaptive immune system, has a key role in initiating and perpetuating synovial inflammation by responding to groups of macromolecules that represent a recognition pattern for potential threats to the host
-
Owing to their similarities to cytokines and their release by activated or damaged cells under conditions of cell stress, the terms 'endokines', 'alarmins', or 'danger signals' have been used as synonyms for proinflammatory damage-associated molecular pattern molecules (DAMPs)
-
DAMPs include high mobility group box 1 protein (HMGB-1), heat-shock proteins, uric acid, altered matrix proteins, S100 proteins, and a growing number of molecules associated with cell stress and necrosis
-
As opposed to pathogen-associated molecular patterns (PAMPs), comprised of pathogen-derived factors, DAMPs represent endogenous molecules that are also ligands for pattern recognition receptors (PRRs)
-
The multiligand receptor for advanced glycation end-products (RAGE) and Toll-like receptors are PRRs, which are involved in transducing signals after binding DAMP molecules
-
The overlapping receptor systems of DAMPs and PAMPs may represent the molecular basis for the well-known link between nonspecific inflammatory triggers like infections and disease flares in autoimmune disorders
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Butler DM et al. (1997) DBA/1 mice expressing the human TNF-alpha transgene develop a severe, erosive arthritis: characterization of the cytokine cascade and cellular composition. J Immunol 159: 2867–2876
Bresnihan B et al. (1998) Treatment of rheumatoid arthritis with recombinant human interleukin-1 receptor antagonist. Arthritis Rheum 41: 2196–2204
Feldmann M and Maini RN (2001) Anti-TNF alpha therapy of rheumatoid arthritis: what have we learned? Annu Rev Immunol 19: 163–196
Ulfgren AK et al. (1995) Detection of cytokine producing cells in the synovial membrane from patients with rheumatoid arthritis. Ann Rheum Dis 54: 654–661
Youssef PP et al. (1998) Quantitative microscopic analysis of inflammation in rheumatoid arthritis synovial membrane samples selected at arthroscopy compared with samples obtained blindly by needle biopsy. Arthritis Rheum 41: 663–669
Lotze MT and Tracey KJ (2005) High-mobility group box 1 protein (HMGB1): nuclear weapon in the immune arsenal. Nat Rev Immunol 5: 331–342
Bianchi ME (2006) DAMPs, PAMPs and alarmins: all we need to know about danger. J Leukoc Biol
Oppenheim JJ and Yang D (2005) Alarmins: chemotactic activators of immune responses. Curr Opin Immunol 17: 359–365
Foell D et al. (2006) S100 proteins expressed in phagocytes: a novel group of damage-associated molecular pattern molecules. J Leukoc Biol 81: 1–5
Muller-Ladner U et al. (2005) Mechanisms of Disease: the molecular and cellular basis of joint destruction in rheumatoid arthritis. Nat Clin Pract Rheumatol 1: 102–110
Falgarone G et al. (2005) Role for innate immunity in rheumatoid arthritis. Joint Bone Spine 72: 17–25
Medzhitov R et al. (1997) A human homologue of the Drosophila Toll protein signals activation of adaptive immunity. Nature 388: 394–397
Bierhaus A et al. (2005) Understanding RAGE, the receptor for advanced glycation end products. J Mol Med 83: 876–886
Bierhaus A et al. (2006) RAGE in inflammation: a new therapeutic target? Curr Opin Investig Drugs 7: 985–991
Jiang W and Pisetsky DS (2007) Mechanisms of Disease: the role of high-mobility group protein 1 in the pathogenesis of inflammatory arthritis. Nat Clin Pract Rheumatol 3: 52–58
Sunahori K et al. (2006) Increased expression of receptor for advanced glycation end products by synovial tissue macrophages in rheumatoid arthritis. Arthritis Rheum 54: 97–104
Radstake TR et al. (2004) Expression of toll-like receptors 2 and 4 in rheumatoid synovial tissue and regulation by proinflammatory cytokines interleukin-12 and interleukin-18 via interferon-gamma. Arthritis Rheum 50: 3856–3865
Cecil DL et al. (2005) Inflammation-induced chondrocyte hypertrophy is driven by receptor for advanced glycation end products. J Immunol 175: 8296–8302
van der Heijden IM et al. (2000) Presence of bacterial DNA and bacterial peptidoglycans in joints of patients with rheumatoid arthritis and other arthritides. Arthritis Rheum 43: 593–598
Schumacher HR Jr et al. (1999) Chlamydia trachomatis nucleic acids can be found in the synovium of some asymptomatic subjects. Arthritis Rheum 42: 1281–1284
Seong SY and Matzinger P (2004) Hydrophobicity: an ancient damage-associated molecular pattern that initiates innate immune responses. Nat Rev Immunol 4: 469–478
Wang H et al. (1999) HMG-1 as a late mediator of endotoxin lethality in mice. Science 285: 248–251
Kamphuis S et al. (2006) Heat-shock protein 60 as a tool for novel therapeutic strategies that target the induction of regulatory T cells in human arthritis. Expert Opin Biol Ther 6: 579–589
Leadbetter EA et al. (2002) Chromatin-IgG complexes activate B cells by dual engagement of IgM and Toll-like receptors. Nature 416: 603–607
Foell D and Roth J (2004) Proinflammatory S100 proteins in arthritis and autoimmune disease. Arthritis Rheum 50: 3762–3771
Scaffidi P et al. (2002) Release of chromatin protein HMGB1 by necrotic cells triggers inflammation. Nature 418: 191–195
Kokkola R et al. (2003) Successful treatment of collagen-induced arthritis in mice and rats by targeting extracellular high mobility group box chromosomal protein 1 activity. Arthritis Rheum 48: 2052–2058
Taniguchi N et al. (2003) High mobility group box chromosomal protein 1 plays a role in the pathogenesis of rheumatoid arthritis as a novel cytokine. Arthritis Rheum 48: 971–981
Rammes A et al. (1997) Myeloid-related protein (MRP) 8 and MRP14, calcium-binding proteins of the S100 family, are secreted by activated monocytes via a novel, tubulin-dependent pathway. J Biol Chem 272: 9496–9502
Viemann D et al. (2005) Myeloid-related proteins 8 and 14 induce a specific inflammatory response in human microvascular endothelial cells. Blood 105: 2955–2962
Hofmann MA et al. (1999) RAGE mediates a novel proinflammatory axis: a central cell surface receptor for S100/calgranulin polypeptides. Cell 97: 889–901
Robinson MJ et al. (2001) The S100 family heterodimer, MRP-8/14, binds with high affinity to heparin and heparan sulphate glycosaminoglycans on endothelial cells. J Biol Chem 277: 3658–3665
Srikrishna G et al. (2001) Two proteins modulating transendothelial migration of leukocytes recognize novel carboxylated glycans on endothelial cells. J Immunol 166: 4678–4688
Imamichi T et al. (1993) Expression and cloning of migration inhibitory factor-related protein (MRP)8 and MRP14 in arthritis-susceptible rats. Biochem Biophys Res Commun 194: 819–825
Nabbe KC et al. (2003) Coordinate expression of activating Fc gamma receptors I and III and inhibiting Fc gamma receptor type II in the determination of joint inflammation and cartilage destruction during immune complex-mediated arthritis. Arthritis Rheum 48: 255–265
van Lent P et al. (2003) The inhibitory receptor FcgammaRII reduces joint inflammation and destruction in experimental immune complex-mediated arthritides not only by inhibition of FcgammaRI/III but also by efficient clearance and endocytosis of immune complexes. Am J Pathol 163: 1839–1848
Hofmann MA et al. (2002) RAGE and arthritis: the G82S polymorphism amplifies the inflammatory response. Genes Immun 3: 123–135
Foell D et al. (2003) Expression of the pro-inflammatory protein S100A12 (EN-RAGE) in rheumatoid and psoriatic arthritis. Rheumatology (Oxford) 42: 1383–1389
Yang Z et al. (2001) Proinflammatory properties of the human S100 protein S100A12. J Leukoc Biol 69: 986–994
Frosch M et al. (2000) Myeloid-related proteins 8 and 14 are specifically secreted during interaction of phagocytes and activated endothelium and are useful markers for monitoring disease activity in pauciarticular-onset juvenile rheumatoid arthritis. Arthritis Rheum 43: 628–637
Srikrishna G et al. (2005) Carboxylated glycans mediate colitis through activation of NF-kappa B. J Immunol 175: 5412–5422
van Eden W et al. (2005) Heat-shock proteins induce T-cell regulation of chronic inflammation. Nat Rev Immunol 5: 318–330
Srivastava P (2002) Roles of heat-shock proteins in innate and adaptive immunity. Nat Rev Immunol 2: 185–194
de Kleer IM et al. (2004) CD4+CD25bright regulatory T cells actively regulate inflammation in the joints of patients with the remitting form of juvenile idiopathic arthritis. J Immunol 172: 6435–6443
Kamphuis S et al. (2005) Tolerogenic immune responses to novel T-cell epitopes from heat-shock protein 60 in juvenile idiopathic arthritis. Lancet 366: 50–56
Prakken BJ et al. (2002) Inhibition of adjuvant-induced arthritis by interleukin-10-driven regulatory cells induced via nasal administration of a peptide analog of an arthritis-related heat-shock protein 60 T cell epitope. Arthritis Rheum 46: 1937–1946
Prakken BJ et al. (2004) Epitope-specific immunotherapy induces immune deviation of proinflammatory T cells in rheumatoid arthritis. Proc Natl Acad Sci USA 101: 4228–4233
Okamura Y et al. (2001) The extra domain A of fibronectin activates Toll-like receptor 4. J Biol Chem 276: 10229–10233
Su SL et al. (2005) Expression and regulation of Toll-like receptor 2 by IL-1beta and fibronectin fragments in human articular chondrocytes. Osteoarthritis Cartilage 13: 879–886
Di Virgilio F et al. (2005) Leukocyte P2 receptors: a novel target for anti-inflammatory and anti-tumor therapy. Curr Drug Targets Cardiovasc Haematol Disord 5: 85–99
Martinon F et al. (2006) Gout-associated uric acid crystals activate the NALP3 inflammasome. Nature 440: 237–241
Shi Y et al. (2003) Molecular identification of a danger signal that alerts the immune system to dying cells. Nature 425: 516–521
Travis S et al. (2005) RDP58 is a novel and potentially effective oral therapy for ulcerative colitis. Inflamm Bowel Dis 11: 713–719
Liacini A et al. (2002) Inhibition of interleukin-1-stimulated MAP kinases, activating protein-1 (AP-1) and nuclear factor kappa B (NF-kappa B) transcription factors down-regulates matrix metalloproteinase gene expression in articular chondrocytes. Matrix Biol 21: 251–262
Powell JD and Horton MR (2005) Threat matrix: low-molecular-weight hyaluronan (HA) as a danger signal. Immunol Res 31: 207–218
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Foell, D., Wittkowski, H. & Roth, J. Mechanisms of Disease: a 'DAMP' view of inflammatory arthritis. Nat Rev Rheumatol 3, 382–390 (2007). https://doi.org/10.1038/ncprheum0531
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncprheum0531
This article is cited by
-
Ghost messages: cell death signals spread
Cell Communication and Signaling (2023)
-
Myeloid-related protein 8/14 in plasma and serum in patients with new-onset juvenile idiopathic arthritis in real-world setting in a single center
Pediatric Rheumatology (2022)
-
Physiological responses and adaptations to high methane production in Japanese Black cattle
Scientific Reports (2022)
-
Cold-inducible RNA-binding protein (CIRP) potentiates uric acid-induced IL-1β production
Arthritis Research & Therapy (2021)
-
Interactions between tumor-derived proteins and Toll-like receptors
Experimental & Molecular Medicine (2020)