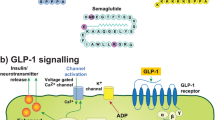
Abstract
The finding that activation of peroxisome proliferator-activated receptor-γ (PPARγ) suppresses inflammation in peripheral macrophages and in models of human autoimmune disease instigated the evaluation of this salutary action for the treatment of CNS disorders with an inflammatory component. The fact that NSAIDs delay the onset of and reduce the risk of developing Alzheimer's disease (AD), while also binding to and activating PPARγ, led to the hypothesis that one dimension of NSAID protection in AD is mediated by PPARγ. Several lines of evidence from experiments using AD-related transgenic cellular and animal models have supported this hypothesis. The capacity of PPARγ agonists to elicit anti-inflammatory, anti-amyloidogenic and insulin-sensitizing effects might account for their observed protective effects. Several clinical trials employing PPARγ agonists have yielded promising results, and further trials are in preparation. Positive outcomes following PPARγ administration have been obtained in animal models of other neurodegenerative diseases, including Parkinson's disease and amyotrophic lateral sclerosis, both of which are associated with a considerable degree of neuroinflammation. Finally, activation of PPARγ has been found to be protective in several models of multiple sclerosis. The verification of these findings in human cells prompted the initiation of clinical studies evaluating PPARγ activation in patients with multiple sclerosis.
Key Points
-
The evaluation of peroxisome proliferator-activated receptor gamma (PPARγ)-mediated actions for CNS disorders was prompted by experimental findings that showed that PPARγ activation suppresses inflammation in peripheral macrophages and in models of human autoimmune disease
-
Synthetic PPARγ ligands have been approved by the FDA and are currently in clinical use as oral treatments for non-insulin-dependent type 2 diabetes
-
Early clinical trials indicate that patients with Alzheimer's disease might benefit from the anti-amyloidogenic, anti-inflammatory and insulin-sensitizing effects of PPARγ activation
-
Positive outcomes in animal models of amyotrophic lateral sclerosis and Parkinson's disease indicate that patients with these conditions might benefit from PPARγ-activating drugs
-
Consistent with data from animal models, observations in a single patient and from a phase I study indicate a clinical benefit of PPARγ agonists in patients with multiple sclerosis
-
Studies in rodent models of cerebral ischemia showed neuroprotection in response to synthetic PPARγ activators, and a large clinical trial recently reported that a synthetic PPARγ agonist reduces the combined risk of heart attack, stroke and death in high-risk patients with type 2 diabetes
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Desvergne B and Wahli W (1999) Peroxisome proliferator-activated receptors: nuclear control of metabolism. Endocr Rev 20: 649–688
Daynes RA and Jones DC (2002) Emerging roles of PPARs in inflammation and immunity. Nat Rev Immunol 2: 748–759
Michalik L et al. (2006) International Union of Pharmacology. LXI: peroxisome proliferator-activated receptors. Pharmacol Rev 58: 726–741
Willson TM et al. (2001) Peroxisome proliferator-activated receptor gamma and metabolic disease. Annu Rev Biochem 70: 341–367
in t'Veld BA et al. (2001) Nonsteroidal antiinflammatory drugs and the risk of Alzheimer's disease. N Engl J Med 345: 1515–1521
Heneka MT et al. (2001) Role for peroxisome proliferator-activated receptor-gamma in Alzheimer's disease. Ann Neurol 49: 276
Heneka MT and O'Banion MK (2007) Inflammatory processes in Alzheimer's disease. J Neuroimmunol 184: 69–91
Wyss-Coray T (2006) Inflammation in Alzheimer disease: driving force, bystander or beneficial response? Nat Med 12: 1005–1015
Hüll M et al. (2002) Pathways of inflammatory activation in Alzheimer's disease: potential targets for disease modifying drugs. Curr Med Chem 9: 83–88
Vodovotz Y et al. (1996) Inducible nitric oxide synthase in tangle-bearing neurons of patients with Alzheimer's disease. J Exp Med 184: 1425–1433
Heneka MT et al. (1999) Peroxisome proliferator-activated receptor gamma agonists protect cerebellar granule cells from cytokine-induced apoptotic cell death by inhibition of inducible nitric oxide synthase. J Neuroimmunol 100: 156–168
Heneka MT et al. (2000) Peroxisome proliferator-activated receptor-γ ligands reduce neuronal inducible nitric oxide synthase expression and cell death in vivo. J Neurosci 20: 6862–6867
Combs CK et al. (2000) Inflammatory mechanisms in Alzheimer's disease: inhibition of β-amyloid-stimulated proinflammatory responses and neurotoxicity by PPARγ agonists. J Neurosci 20: 558–567
Luna-Medina R et al. (2005) Regulation of inflammatory response in neural cells in vitro by thiadiazolidinones derivatives through peroxisome proliferator-activated receptor gamma activation. J Biol Chem 280: 21453–21462
Heneka MT et al. (2003) Noradrenergic depletion increases inflammatory responses in brain: effects on Ikappa B and HSP70 expression. J Neurochem 85: 387–398
Maeshiba Y et al. (1997) Disposition of the new antidiabetic agent pioglitazone in rats, dogs, and monkeys. Drug Res 47: 29–35
Yan Q et al. (2003) Anti-inflammatory drug therapy alters β-amyloid processing and deposition in an animal model of Alzheimer's disease. J Neurosci 23: 7504–7509
Heneka MT et al. (2005) Acute treatment with the PPARγ agonist pioglitazone and ibuprofen reduces glial inflammation and Aβ1–42 levels in APPV717I transgenic mice. Brain 128: 1442–1453
Sastre M et al. (2006) Nonsteroidal anti-inflammatory drugs repress β-secretase gene promoter activity by the activation of PPARγ. Proc Nat Acad Sci USA 103: 443–448
Sastre M et al. (2003) Nonsteroidal anti-inflammatory drugs and peroxisome proliferator-activated receptor-γ agonists modulate immunostimulated processing of amyloid precursor protein through regulation of β-secretase. J Neurosci 23: 9796–9804
d'Abramo C et al. (2005) Role of peroxisome proliferator-activated receptor γ in amyloid precursor protein processing and amyloid β-mediated cell death. Biochem J 391: 693–698
Camacho IE et al. (2004) Peroxisome proliferator-activated receptor γ induces a clearance mechanism for the amyloid-β peptide. J Neurosci 24: 10908–10917
Pedersen WA et al. (2006) Rosiglitazone attenuates learning and memory deficits in Tg2576 Alzheimer mice. Exp Neurol 199: 265–273
Watson GS et al. (2005) Preserved cognition in patients with early Alzheimer disease and amnestic mild cognitive impairment during treatment with rosiglitazone: a preliminary study. Am J Geriatr Psychiatry 13: 950–958
Risner ME et al. (2006) Efficacy of rosiglitazone in a genetically defined population with mild-to-moderate Alzheimer's disease. Pharmacogenomics J 6: 246–254
Luchsinger JA et al. (2001) Diabetes mellitus and risk of Alzheimer's disease and dementia with stroke in a multiethnic cohort. Am J Epidemiol 154: 635–641
Geldmacher D and Landreth G (2004) Pioglitazone in Alzheimer's disease: rationale and clinical trial design. In Proceedings of the 9th International Conference on Alzheimer's Disease and Related Disorders: 2004 17–22 July, Philadelphia. P1: 397
Pedersen WA and Flynn ER (2004) Insulin resistance contributes to aberrant stress responses in the Tg2576 mouse model of Alzheimer's disease. Neurobiol Dis 17: 500–506
Kuusisto J et al. (1997) Association between features of the insulin resistance syndrome and Alzheimer's disease independently of apolipoprotein E4 phenotype: cross sectional population based study. Br Med J 315: 1045–1049
Liberatore GT et al. (1999) Inducible nitric oxide synthase stimulates dopaminergic neurodegeneration in the MPTP model of Parkinson disease. Nat Med 5: 1403–1409
Breidert T et al. (2002) Protective action of the peroxisome proliferator-activated receptor-gamma agonist pioglitazone in a mouse model of Parkinson's disease. J Neurochem 82: 615–624
Dehmer T et al. (2004) Protection by pioglitazone in the MPTP model of Parkinson's disease correlates with I kappa B alpha induction and block of NF kappa B and iNOS activation. J Neurochem 88: 494–501
Hunter RL et al. (2007) Inflammation induces mitochondrial dysfunction and dopaminergic neurodegeneration in the nigrostriatal system. J Neurochem 100: 1375–1386
Weydt P and Moller T (2005) Neuroinflammation in the pathogenesis of amyotrophic lateral sclerosis. Neuroreport 16: 527–531
Kiaei M et al. (2005) Peroxisome proliferator-activated receptor-gamma agonist extends survival in transgenic mouse model of amyotrophic lateral sclerosis. Exp Neurol 191: 331–336
Schutz B et al. (2005) The oral antidiabetic pioglitazone protects from neurodegeneration and amyotrophic lateral sclerosis-like symptoms in superoxide dismutase-G93A transgenic mice. J Neurosci 25: 7805–7812
Park SW et al. (2007) Thiazolidinedione class of peroxisome proliferator-activated receptor gamma agonists prevents neuronal damage, motor dysfunction, myelin loss, neuropathic pain, and inflammation after spinal cord injury in adult rats. J Pharmacol Exp Ther 320: 1002–1012
Martin R et al. (1992) Immunological aspects of demyelinating diseases. Ann Rev Immunol 10: 153–187
Pershadsingh HA (2004) Peroxisome proliferator-activated receptor-γ: therapeutic target for diseases beyond diabetes: quo vadis? Expert Opin Investig Drugs 13: 215–228
Niino M et al. (2001) Amelioration of experimental autoimmune encephalomyelitis in C57BL/6 mice by an agonist of peroxisome proliferator-activated receptor-γ. J Neuroimmunol 116: 40–48
Diab A et al. (2002) Peroxisome proliferator-activated receptor-gamma agonist 15-deoxy-Δ12,14-prostaglandin J2 ameliorates experimental autoimmune encephalomyelitis. J Immunol 168: 2508–2515
Rossi A et al. (2000) Anti-inflammatory cyclopentenone prostaglandins are direct inhibitors of IκB kinase. Nature 403: 103–108
Feinstein DL et al. (2002) Peroxisome proliferator-activated receptor-γ agonists prevent experimental autoimmune encephalomyelitis. Ann Neurol 51: 694–702
Charo IF and Ransohoff RM (2006) Mechanisms of disease—the many roles of chemokines and chemokine receptors in inflammation. N Engl J Med 354: 610–621
Bright JJ et al. (2003) Peroxisome proliferator-activated receptor-gamma-deficient heterozygous mice develop an exacerbated neural antigen-induced Th1 response and experimental allergic encephalomyelitis. J Immunol 171: 5743–5750
Raikwar HP et al. (2006) PPARγ antagonists reverse the inhibition of neural antigen-specific Th-1 response and experimental allergic encephalomyelitis by Ciglitazone and 15-deoxy-Δ12,14-prostaglandin J2 . J Neuroimmunol 178: 76–86
Rieckmann P et al. (1995) Tumor necrosis factor-alpha messenger RNA expression in patients with relapsing–remitting multiple sclerosis is associated with disease activity. Ann Neurol 37: 82–88
Schmidt S et al. (2004) Anti-inflammatory and antiproliferative actions of PPAR-γ agonists on T lymphocytes derived from MS patients. J Leukoc Biol 75: 478–485
Klotz L et al. (2005) Proinflammatory stimulation and pioglitazone treatment regulate peroxisome proliferator-activated receptor γ levels in peripheral blood mononuclear cells from healthy controls and multiple sclerosis patients. J Immunol 175: 4948–4955
Waite KJ et al. (2001) Interferon-γ-induced regulation of peroxisome proliferator-activated receptor γ and STATs in adipocytes. J Biol Chem 276: 7062–7068
Zheng Z and Yenari MA (2004) Post-ischemic inflammation: molecular mechanisms and therapeutic implications. Neurol Res 26: 884–892
Perera MN et al. (2006) Inflammation following stroke. J Clin Neurosci 13: 1–8
Sundararajan S et al. (2005) Peroxisome proliferator-activated receptor-γ ligands reduce inflammation and infarction size in transient focal ischemia. Neuroscience 130: 685–696
Victor NA et al. (2006) Altered PPARγ expression and activation after transient focal ischemia in rats. Eur J Neurosci 24: 1653–1663
Chen SD et al. (2006) Effects of rosiglitazone on global ischemia-induced hippocampal injury and expression of mitochondrial uncoupling protein 2. Biochem Biophys Res Commun 351: 198–203
Pereira MP et al. (2005) The nonthiazolidinedione PPARγ agonist L-796,449 is neuroprotective in experimental stroke. J Neuropathol Exp Neurol 64: 797–805
Zhao Y et al. (2005) The intracerebral application of the PPARγ-ligand pioglitazone confers neuroprotection against focal ischaemia in the rat brain. Eur J Neurosci 22: 278–282
Luo Y et al. (2006) Neuroprotection against focal ischemic brain injury by the peroxisome proliferator-activated receptor-γ agonist rosiglitazone. J Neurochem 97: 435–448
Pereira MP et al. (2006) Rosiglitazone and 15-deoxy-Δ12,14-prostaglandin J2 cause potent neuroprotection after experimental stroke through noncompletely overlapping mechanisms. J Cereb Blood Flow Metab 26: 218–229
Culman J et al. (2007) PPARγ: therapeutic target for ischemic stroke. Trends Pharmacol Sci 28: 244–249
Tureyen K et al. (2007) Peroxisome proliferator-activated receptor-γ agonists induce neuroprotection following transient focal ischemia in normotensive, normoglycemic as well as hypertensive and type-2 diabetic rodents. J Neurochem 101: 41–56
Dormandy JA et al. (2005) Secondary prevention of macrovascular events in patients with type 2 diabetes in the PROactive Study (PROspective pioglitAzone Clinical Trial In macroVascular Events): a randomised controlled trial. Lancet 366: 1279–1289
Lee J and Reding M (2007) Effects of thiazolidinediones on stroke recovery: a case-matched controlled study. Neurochem Res 32: 635–638
Lee BC et al. (2006) Peroxisome proliferator-activated receptor-γ2 Pro12Ala polymorphism is associated with reduced risk for ischemic stroke with type 2 diabetes. Neurosci Lett 410: 141–145
Acknowledgements
MT Heneka was supported by a grant from the Deutsche Forschungsgemeinschaft (DFG, HE 3350/6-1). GE Landreth was supported by grants from the NIH (AG16740) and the Blanchett Hooker Rockefeller Foundation. This publication is part of the German Research Network on Dementia and was funded by the German Federal Ministry for Education and Research (grant 01 G1 0420).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Heneka, M., Landreth, G. & Hüll, M. Drug Insight: effects mediated by peroxisome proliferator-activated receptor-γ in CNS disorders. Nat Rev Neurol 3, 496–504 (2007). https://doi.org/10.1038/ncpneuro0586
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneuro0586
This article is cited by
-
Meta-analysis of mouse transcriptomic studies supports a context-dependent astrocyte reaction in acute CNS injury versus neurodegeneration
Journal of Neuroinflammation (2020)
-
Pioglitazone ameliorates the phenotype of a novel Parkinson’s disease mouse model by reducing neuroinflammation
Molecular Neurodegeneration (2016)
-
Rosiglitazone Synergizes the Neuroprotective Effects of Valproic Acid Against Quinolinic Acid-Induced Neurotoxicity in Rats: Targeting PPARγ and HDAC Pathways
Neurotoxicity Research (2014)
-
Modulating Microglia Activity with PPAR-γ Agonists: A Promising Therapy for Parkinson’s Disease?
Neurotoxicity Research (2013)
-
The PPAR-gamma agonist pioglitazone protects cortical neurons from inflammatory mediators via improvement in peroxisomal function
Journal of Neuroinflammation (2012)