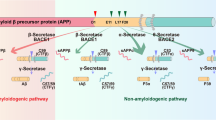
Abstract
Amyloidosis is a clinical disorder caused by extracellular deposition of proteins that are normally soluble as insoluble, abnormal fibrils that impair organ function. More than 20 unrelated proteins can form amyloid fibrils in vivo. All fibrils share cross-β core structure and pathognomonic red–green birefringence when viewed under cross-polarized light after staining with Congo red. Amyloidosis can be acquired or hereditary, localized or systemic, and is classified according to the fibril precursor protein. Local amyloid deposition occurs in the brain in Alzheimer's disease and in the pancreas in maturity-onset diabetes, but a direct role in the pathogenesis of these diseases remains unproven. Systemic amyloidosis, with amyloid deposits in the viscera, blood vessel walls and connective tissues, is usually fatal and is the cause of about one death per thousand in developed countries. Recent elucidation of fundamental aspects of the pathogenesis of amyloidosis, and developments in diagnosis and monitoring of this disorder have greatly improved outcome for patients. Several exciting novel therapeutic strategies, reviewed in this article, are in development. These include interference with different stages of fibrillogenesis and enhancement of clearance of amyloid deposits.
Key Points
-
Amyloidosis—extracellular deposition of insoluble fibrils composed of misfolded proteins—can be localized or systemic
-
Current treatments (e.g. anti-inflammatory drugs, chemotherapy and transplantation) aim to reduce the supply of amyloid fibril precursor proteins
-
Recent advances in our understanding of the pathogenesis of amyloidosis have spurred attempts to develop novel treatments
-
New therapeutic strategies aim to perturb fibrillogenesis by stabilizing precursor proteins (e.g. Fibrillex®) or promote clearance of amyloid (e.g. immunotherapy, CPHPC)
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Pepys MB (2006) Amyloidosis. Annu Rev Med 57: 223–241
Booth DR et al. (1997) Instability, unfolding and aggregation of human lysozyme variants underlying amyloid fibrillogenesis. Nature 385: 787–793
Sunde M et al. (1997) Common core structure of amyloid fibrils by synchrotron X-ray diffraction. J Mol Biol 273: 729–739
Kisilevsky R and Fraser P (1996) Proteoglycans and amyloid fibrillogenesis. Ciba Found Symp 199: 58–67
Li JP et al. (2005) In vivo fragmentation of heparan sulfate by heparanase overexpression renders mice resistant to amyloid protein A amyloidosis. Proc Natl Acad Sci USA 102: 6473–6477
Tennent GA et al. (1995) Serum amyloid P component prevents proteolysis of the amyloid fibrils of Alzheimer's disease and systemic amyloidosis. Proc Natl Acad Sci USA 92: 4299–4303
Pepys MB et al. (2002) Targeted pharmacological depletion of serum amyloid P component for treatment of human amyloidosis. Nature 417: 254–259
Hawkins PN et al. (1988) Diagnostic radionuclide imaging of amyloid: biological targeting by circulating human serum amyloid P component. Lancet 1: 1413–1418
Hawkins PN et al. (1990) Evaluation of systemic amyloidosis by scintigraphy with 123I-labeled serum amyloid P component. N Engl J Med 323: 508–513
Hawkins PN et al. (1993) Serum amyloid P component scintigraphy and turnover studies for diagnosis and quantitative monitoring of AA amyloidosis in juvenile rheumatoid arthritis. Arthritis Rheum 36: 842–851
Gillmore JD et al. (2001) Amyloid load and clinical outcome in AA amyloidosis in relation to circulating concentration of serum amyloid A protein. Lancet 358: 24–29
Gillmore JD et al. (1999) Serum amyloid P component scintigraphy in AL amyloidosis. In Amyloid and Amyloidosis 1998, 148–150 (Eds Kyle RA and Gertz MA) New York: Parthenon Publishing
Tan SY et al. (1996) Long term effect of renal transplantation on dialysis-related amyloid deposits and symptomatology. Kidney Int 50: 282–289
Rydh A et al. (1998) Serum amyloid P component scintigraphy in familial amyloid polyneuropathy: regression of visceral amyloid following liver transplantation. Eur J Nucl Med 25: 709–713
Gillmore JD et al. (1999) Curative hepatorenal transplantation for systemic amyloidosis associated with fibrinogen a-chain Glu526Val in an English family. In Amyloid and Amyloidosis 1998, 336–338 (Eds Kyle RA and Gertz MA) New York: Parthenon Publishing
Moreau P et al. (1998) Prognostic factors for survival and response after high-dose therapy and autologous stem cell transplantation in systemic AL amyloidosis: a report on 21 patients. Br J Haematol 101: 766–769
Kyle RA et al. (1997) A trial of three regimens for primary amyloidosis: colchicine alone, melphalan and prednisone, and melphalan, prednisone, and colchicine. N Engl J Med 336: 1202–1207
Dominguez DI and De Strooper B (2002) Novel therapeutic strategies provide the real test for the amyloid hypothesis of Alzheimer's disease. Trends Pharmacol Sci 23: 324–330
Ledue TB et al. (1998) Analytical evaluation of particle-enhanced immunonephelometric assays for C-reactive protein, serum amyloid A and mannose-binding protein in human serum. Ann Clin Biochem 35: 745–753
Bradwell AR et al. (2001) Highly sensitive, automated immunoassay for immunoglobulin free light chains in serum and urine. Clin Chem 47: 673–680
Drayson M et al. (2001) Serum free light-chain measurements for identifying and monitoring patients with nonsecretory multiple myeloma. Blood 97: 2900–2902
Lachmann HJ et al. (2003) Outcome in systemic AL amyloidosis in relation to changes in concentration of circulating free immunoglobulin light chains following chemotherapy. Br J Haematol 122: 78–84
Buxbaum J (1986) Aberrant immunoglobulin synthesis in light chain amyloidosis: free light chain and light chain fragment production by human bone marrow cells in short-term tissue culture. J Clin Invest 78: 798–806
Kyle RA and Gertz MA (1995) Primary systemic amyloidosis: clinical and laboratory features in 474 cases. Semin Hematol 32: 45–59
Sacchettini JC and Kelly JW (2002) Therapeutic strategies for human amyloid diseases. Nat Rev Drug Discov 1: 267–275
Almeida MR et al. (2004) Selective binding to transthyretin and tetramer stabilization in serum from patients with familial amyloidotic polyneuropathy by an iodinated diflunisal derivative. Biochem J 381: 351–356
Kisilevsky R et al. (1995) Arresting amyloidosis in vivo using small molecule anionic sulphonates or sulphates: implications for Alzheimer's disease. Nature Med 1: 143–148
Soto C et al. (1998) β-sheet breaker peptides inhibit fibrillogenesis in a rat brain model of amyloidosis: implications for Alzheimer's therapy. Nat Med 4: 822–826
Sigurdsson EM et al. (2000) In vivo reversal of amyloid-beta lesions in rat brain. J Neuropathol Exp Neurol 59: 11–17
Soto C et al. (2000) Reversion of prion protein conformational changes by synthetic beta-sheet breaker peptides. Lancet 355: 192–197
O'Nuallain B and Wetzel R (2002) Conformational Abs recognizing a generic amyloid fibril epitope. Proc Natl Acad Sci USA 99: 1485–1490
Hrncic R et al. (2000) Antibody-mediated resolution of light chain-associated amyloid deposits. Am J Pathol 157: 1239–1246
Solomon A et al. (2003) Therapeutic potential of chimeric amyloid-reactive monoclonal antibody 11-1F4. Clin Cancer Res 9: 3831S–3838S
Solomon A et al. (2003) Immunotherapy in systemic primary (AL) amyloidosis using amyloid-reactive monoclonal antibodies. Cancer Biother Radiopharm 18: 853–860
Schenk D et al. (1999) Immunization with amyloid-β attenuates Alzheimer-disease-like pathology in the PDAPP mouse. Nature 400: 173–177
Sigurdsson EM et al. (2001) Immunization with a nontoxic/nonfibrillar amyloid-beta homologous peptide reduces Alzheimer's disease-associated pathology in transgenic mice. Am J Pathol 159: 439–447
Bard F et al. (2000) Peripherally administered antibodies against amyloid β-peptide enter the central nervous system and reduce pathology in a mouse model of Alzheimer disease. Nature Med 6: 916–919
DeMattos RB et al. (2001) Peripheral anti-Aβ antibody alters CNS and plasma A beta clearance and decreases brain A beta burden in a mouse model of Alzheimer's disease. Proc Natl Acad Sci USA 98: 8850–8855
Janus C et al. (2000) A beta peptide immunization reduces behavioural impairment and plaques in a model of Alzheimer's disease. Nature 408: 979–982
Morgan D et al. (2000) A beta peptide vaccination prevents memory loss in an animal model of Alzheimer's disease. Nature 408: 982–985
Senior K (2002) Dosing in phase II trial of Alzheimer's vaccine suspended. Lancet Neurol 1: 3
Hock C et al. (2003) Antibodies against beta-amyloid slow cognitive decline in Alzheimer's disease. Neuron 38: 547–554
Pepys MB et al. (1997) Amyloid P component: a critical review. Amyloid: Int J Exp Clin Invest 4: 274–295
Pepys MB et al. (1994) Human serum amyloid P component is an invariant constituent of amyloid deposits and has a uniquely homogeneous glycostructure. Proc Natl Acad Sci USA 91: 5602–5606
Botto M et al. (1997) Amyloid deposition is delayed in mice with targeted deletion of the serum amyloid P component gene. Nature Med 3: 855–859
Gillmore JD et al. (2000) Curative hepatorenal transplantation in systemic amyloidosis caused by the Glu526Val fibrinogen α-chain variant in an English family. QJ Med 93: 269–275
Gillmore JD et al. (1998) Allogeneic bone marrow transplantation for systemic AL amyloidosis. Brit J Haematol 100: 226–228
Acknowledgements
The work of the Centre for Amyloidosis and Acute Phase Proteins is supported by grants from the Medical Research Council (UK), The Wellcome Trust, the Wolfson Foundation, and NHS Research and Development Funds.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
Work conducted within Roche was funded entirely by F Hoffmann-La Roche Ltd, which also provided equipment and support to the Centre for Amyloidosis and Acute Phase Proteins.
The patent on CPHPC is held by Roche. Roche retains all rights to commercial exploitation of CPHPC for treatment of Alzheimer's disease and diabetes, but has granted an exclusive licence to UCL Biomedica, the commercial arm of the University College Medical School, for development of CPHPC for other indications.
Rights and permissions
About this article
Cite this article
Gillmore, J., Hawkins, P. Drug Insight: emerging therapies for amyloidosis. Nat Rev Nephrol 2, 263–270 (2006). https://doi.org/10.1038/ncpneph0169
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/ncpneph0169
This article is cited by
-
Pharmacokinetics and Pharmacodynamics of Patisiran in Patients with hATTR Amyloidosis and with Polyneuropathy After Liver Transplantation
Clinical Pharmacokinetics (2023)
-
Sopping up SAP
Science-Business eXchange (2010)
-
Amyloidosis in a patient with autosomal dominant polycystic kidney disease and tuberculosis: a case report
International Urology and Nephrology (2007)