Abstract
Twinned growth behaviour in the rapidly emerging area of two-dimensional nanomaterials still remains unexplored although it could be exploited to fabricate heterostructure and superlattice materials. Here we demonstrate how one can utilize the twinned growth relationship between two two-dimensional materials to construct vertically stacked heterostructures. As a demonstration, we achieve 100% overlap of the two transition metal dichalcogenide layers constituting a ReS2/WS2 vertical heterostructure. Moreover, the crystal size of the stacked structure is an order of magnitude larger than previous reports. Such twinned transition metal dichalcogenides vertical heterostructures exhibit great potential for use in optical, electronic and catalytic applications. The simplicity of the twinned growth can be utilized to expand the fabrication of other heterostructures or two-dimensional material superlattice and this strategy can be considered as an enabling technology for research in the emerging field of two-dimensional van der Waals heterostructures.
Similar content being viewed by others
Introduction
The rational stacking of two different transition metal dichalcogenides (TMDCs, two-dimensional atomic crystalline materials with tunable electronic structure1,2,3,4,5 and great potential in optoelectronic devices6,7,8,9,10), has drawn significant attention as it endows TMDCs with great opportunities to expand their pristine properties11,12,13,14,15 and broaden their applications16,17,18,19,20. Initially, vertically stacked heterostructures comprising multi-layered TMDCs were obtained by sequential mechanical exfoliation. These studies triggered subsequent studies focused on the top-down creation of TMDCs heterostructures with varied chemical composition, interlayer spacing, and angular alignment11. However, due to the limited size and randomly generated locations of the exfoliated TMDCs flakes, this stacking procedure is neither controllable nor scalable. Later, Ajayan et al. presented an approach for the chemical vapour deposition (CVD) synthesis of stacked WS2/MoS2 heterostructures, which showed the potential for larger scale production21. However, due to the random nucleation process and poor control over the growth rate, it is extremely difficult to significantly increase the stacking area or to precisely control the growth behaviour of vertically stacked TMDCs heterostructures. Thus far, the stacking of two TMDCs constituents in a strictly controlled manner (especially in terms of control over the overlap percentage) has yet to be achieved, even though this is highly desirable.
Herein, we demonstrate the twinned growth of two 2D (two-dimensional) nanomaterials, namely ReS2 and WS2. We achieve 100% overlap for each of the stacked TMDCs structures, with crystal sizes of the heterostructures one order of magnitude larger than previous reports12,13,14,16,18,20,21,22,23,24,25,26. For the twinned growth of ReS2/WS2 vertical heterostructures, Au is chosen as the substrate and W–Re alloy foil is used as the Re and W sources, which lower the barrier energies for this special twinned growth process. We believe that the developed approach will promote and accelerate ongoing research efforts of 2D crystalline van der Waals heterostructures.
Results
The morphology and spectral characterization of ReS2/WS2
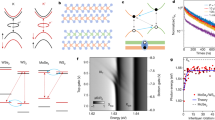
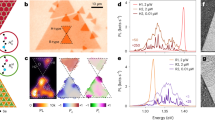
Figure 1 schematically shows the strategy for the twinned growth of ReS2 and WS2 towards vertically stacked heterostructures. From scanning electron microscopy (SEM) images (for example, Fig. 2a) taken from the Au surface after the CVD reaction, one can observe large-area uniformity and a high yield of triangular vertically stacked ReS2/WS2 crystals. One can also see that the domain size of the smaller stacked ReS2/WS2 structures is typically >10 μm, while the bigger crystal reaches a large size of 600 μm2 (Fig. 2a). This is about 1 order of magnitude larger than other reports of vertically stacked TMDCs heterostructures synthesized by traditional CVD technique or by a sequential exfoliation process (Fig. 2b). Moreover, from Fig. 2b, we can also conclude that a 100% overlap of two TMDCs is obtained, and this is also much higher than previously reported findings. To further characterize the structure of the twinned ReS2/WS2 crystals, the samples were transferred onto 300 nm Si/SiO2 substrates. After transfer, typical optical image of the triangular crystal is shown as Fig. 2c, which reveals that the stacked ReS2/WS2 structure exhibits a uniform light-purple triangle consistent with a high uniformity in agreement with the SEM data (representative image can be seen in Fig. 2a). In addition, atomic force microscope images of the transferred crystals show a uniform height of ∼1.6 nm over the entire structure (Supplementary Fig. 1), which confirms the bilayered, 100% overlapped structure and twinned growth nature of the ReS2/WS2 heterostructures.
(a) SEM image of the triangular ReS2/WS2 vertical crystalline heterostructures over Au. A ReS2/WS2 crystal with grain size up to 600 μm2 is indicated. (b) Crystal size plotted as a function of overlap percentage of TMDCs heterostacks obtained in this work and those reported in other literature. (c) Optical image of a ReS2/WS2 twinned vertical heterostructure crystal after transfer onto a Si/SiO2 substrate. (d) Comparison of the Raman spectra of ReS2, WS2, and ReS2/WS2, in which four distinct peaks are observed from the twinned ReS2/WS2 crystal (e,f) Raman mappings of peak intensity at 160.5 and 351.3 cm−1 respectively, corresponding to the E2g mode of ReS2 and WS2 respectively. Scale bar, 40 μm (a); 5 μm (c,e,f).
Figure 2d shows the comparison between the Raman spectra of pure ReS2, pure WS2 and the twinned ReS2/WS2 heterostructure. Within the Raman spectrum of heterostructure, one can observe peaks from both ReS2 (E2g≈160.5 cm−1 and A1g≈211.3 cm−1)27 and WS2 (E2g≈351.3 cm−1 and A1g≈417.8 cm−1)28. This shows that the structures are ReS2/WS2 vertically stacked heterostructures and not an alloyed RexW1−xS2 crystal, since the Raman spectrum of a RexW1−xS2 alloy would show two main peaks with their positions between the E2g and A1g peaks of pure ReS2 or WS2. To further confirm the 100% overlapped structure of the vertical heterostructures, Raman mapping was used. Typical examples are provided in Fig. 2e,f, in which Raman intensity maps using the ReS2 E2g mode at 160.5 cm−1 and the WS2 E2g mode at 351.3 cm−1 demonstrate a uniform response for both ReS2 and WS2 over the entire crystal. This rules out the possibility of alloying. In addition, Raman spectra were collected at fixed spatial positions at the center and corners of 11 randomly selected triangle ReS2/WS2 crystals. Each verified the distinct signatures corresponding to ReS2 and WS2 (Supplementary Fig. 2), so that the 100% overlapped structure and the large-scale twinned growth of the ReS2/WS2 vertical heterostructures were further confirmed.
Optoelectronic characterization of ReS2/WS2
To explore the electronic structure of our ReS2/WS2 twinned heterostructures, X-ray photoelectron spectroscopy (XPS) was implemented (see Fig. 3a and Supplementary Fig. 3). From the XPS data, the (Re and W):S atomic ratio is 1:2.0, indicating that the twinned ReS2/WS2 is stoichiometric. As shown in Fig. 3b, comparison of the W 4f core level doublet from WS2 and ReS2/WS2 shows an up-shift of 300 meV, corresponding to a positive net charge on the WS2 bottom layer29. Similarly, comparison of the Re 4f core level doublet from ReS2 and ReS2/WS2 shows a down-shift of 475 meV, corresponding to a negative net charge on the ReS2 top layer30. Our results therefore indicate that the ReS2 layer has a negative net charge, while the WS2 layer has a positive net charge as a result of a contact potential. Hence the ReS2/WS2 heterostructures serve as an atomically thin capacitor with a potential up to 775 meV, which originates from the work function difference induced charge transfer between the constituent ReS2 and WS2 layers and is twice that of previously reported TMDCs vertical heterostructures20.
(a) Sketch of the X-ray photoelectron analyses of ReS2/WS2 heterostack. (b) XPS core level shift analyses of ReS2/WS2 heterostructures. (c) Photoluminescence spectrum of the ReS2/WS2 heterostack, in which the three peaks are attributed to the exciton relaxations within ReS2 (green), WS2 (blue) and between ReS2/WS2 layers (grey). (d) Band diagram of ReS2/WS2 heterostructures under photo excitation, depicting (1) absorption and exciton generation in WS2 and ReS2, (2) relaxation of excitons at the ReS2/WS2 interface driven by the band offset, and (3) radiative recombination of spatially indirect excitons. (e) Low-magnification TEM image of the ReS2/WS2 twinned vertical heterostructures, where a triangle crystal of ReS2/WS2 hetero-bilayer is marked. (f) High-resolution TEM image of ReS2/WS2 vertical heterostructures showing the resulting Moiré pattern. (g) Fast Fourier transform (FFT) of the heterostructures in f. The inset shows the two patterns of ReS2 (green) and WS2 (purple) with a rotation angle to be about 5.6°. (h) Tentative orientation model of rotating the upper ReS2 by an angle of 5.6° with respect to the ground WS2. Insert is the atomistic illustration of the heterostructure of ReS2/WS2 with their respective lattice constants and a misalignment angle ϕ. Scale bar, 1 μm (e); 4 nm (f); 4 nm−1 (g).
Photoluminescence (PL) spectrometry was used to investigate the interaction between ReS2/WS2 heterostack layers. As shown in Fig. 3c, three prominent peaks are observed at exciton transition energies of 1.89, 1.63 and 1.26 eV, corresponding to wavelengths of 657, 759 and 987 nm, respectively. The two strong exciton peaks at 1.63 eV (759 nm) and 1.89 eV (657 nm) are consistent with the PL intensities for ReS2 and WS2, and are in good agreement with previous works29,30. The weak peak at 1.26 eV (987 nm) can be attributed to an indirect exciton transition between the ReS2 and WS2 layers. This interlayer PL exciton transition is a type II band alignment, which highlights the twinned growth nature of the ReS2/WS2 layers forming the heterostructure. Figure 3d shows the band diagram for ReS2/WS2 heterostructures under photo excitation. Owing to energy lost to the band offset, the PL exciton peak energy (1.26 eV) is lower than the excitonic band gaps for either of the TMDCs constituents (1.63 eV for ReS2 or 1.89 eV for WS2). Moreover, the strong luminescence signal at energies corresponding to the excitonic band gaps of ReS2 (1.63 eV) and WS2 (1.89 eV) suggests that a minority of the photoexcited carriers are relaxed at the interface leading to a low luminescence signal from the spatially indirect recombination process. Although the intensity of the indirect excitonic peak is weak, it is still an indication that the interface of the twinned ReS2/WS2 is clean and contamination-free, which confirms the advantages of our strategy for the fabrication of high-quality TMDCs heterostructures.
High-resolution characterization of ReS2/WS2
To further reveal the crystalline structure and the stack orientation of the twinned ReS2/WS2 heterostructures, high-resolution transmission electron microscopy (HRTEM) was utilized to characterize the samples after transferring them to a TEM grid or onto a Si/SiO2 substrate. Low-magnification TEM image of a ReS2/WS2 twinned vertical heterostructure is presented in Fig. 3e, in which the crystal edge is marked. The structural continuity within the ReS2/WS2 domain suggests that our twinned vertical heterostructure is of high quality. Clear Moiré pattern, with periodicity measured to be about 3.55 nm, is observed in the high-resolution TEM image of the stacked structures as shown in Fig. 3f and the corresponding result of Fast Fourier transform is exhibited in Fig. 3g. Inspection of the Fast Fourier transform pattern reveals that in this particular sample the two hexagonal reciprocal lattices are rotated by ϕ=5.6° with respect to each other and there is negligible strain in the two constituent layers. Further simulation of the atomic structure by rotating the upper ReS2 by the angle of ϕ with respect to the ground WS2 layer (Fig. 3h) has shown similar Moiré pattern to that observed in Fig. 3f. To further confirm the stacked nature of the heterostructure, cross-section TEM samples were prepared by focused ion beam (FIB). The cross-sectional TEM images of ReS2/WS2 heterostructure demonstrate the clean interface as well as the bilayer stacked structure of our twinned ReS2/WS2 heterostacks (Supplementary Fig. 4).
Discussion
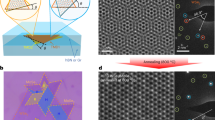
To have a better understanding of the twinned growth behaviour between the ReS2 and WS2 layers on Au, we simulated the growth process of ReS2 and WS2 based on the density functional theory (DFT) calculations (as shown in Fig. 4). Before these first-principles calculations, however, X-ray diffraction investigations were conducted. The data confirmed that the Au substrate is crystalline (Supplementary Fig. 5). When referenced with JCPDS data (JCPDS 04–0784), a clear match for Au (111) is obtained. Moreover, this was found for the entire surface, which confirms the single crystalline nature of our Au substrates.
Although the growth process of ReS2 and WS2 is complex, it can be simplified as follows31. The Re and W precursors are partially reduced by H2S gas to form a subsulfide species of ReSx and WSx (x=1∼3), which are then further sulfurized into ReS2 or WS2 over the substrate (as shown in Fig. 4a). To confirm the proposed mechanism, the adsorption energies for Re, W atoms over Au (111) and for Re atoms over WS2 (001) were calculated using first-principles calculations within DFT. The adsorption results for these atoms on the different substrates are provided in Fig. 4b–d. From the data it is clear that the adsorption energies for W atoms over Au(111) and Re atoms on WS2(001) are very close to each other (EadsW, Au(111)=2.77 eV,  ). However, the adsorption energy for Re atoms over Au(111) (EadsRe, Au(111)=1.52 eV) is far weaker. At the implemented high growth temperature of 900 °C this weak energy would easily be overcome, resulting in the facile desorption of Re species making the nucleation of ReS2 highly unlikely. However, the strong adsorption energy (2.77 eV) is sufficiently accessible for WS2 to nucleate and grow over Au (111). Once WS2 forms on the substrate, the high adsorption energy of Re atom on WS2(001) would induce the subsequent adsorption, nucleation and growth of ReS2 on the newly formed WS2 surface. This beautifully highlights the twinned growth of ReS2/WS2 crystals.
). However, the adsorption energy for Re atoms over Au(111) (EadsRe, Au(111)=1.52 eV) is far weaker. At the implemented high growth temperature of 900 °C this weak energy would easily be overcome, resulting in the facile desorption of Re species making the nucleation of ReS2 highly unlikely. However, the strong adsorption energy (2.77 eV) is sufficiently accessible for WS2 to nucleate and grow over Au (111). Once WS2 forms on the substrate, the high adsorption energy of Re atom on WS2(001) would induce the subsequent adsorption, nucleation and growth of ReS2 on the newly formed WS2 surface. This beautifully highlights the twinned growth of ReS2/WS2 crystals.
To further confirm the twinned growth behaviour, different support substrates, namely, W foil, Re foil and W–Re alloy foil were investigated at a growth temperature of 900 °C. Figure 5 shows the scheme for the different support substrates and corresponding growth results. When W foil is used, WS2 can easily form on the surface of Au(111) (see Fig. 5a and Supplementary Fig. 6), confirming the high adsorption energy of W atoms on Au(111). This allows the nucleation and growth of WS2 on Au(111) at 900 °C. However, when Re foil is used as the support substrate, no ReS2 Raman response is found on the Au(111) surface (as shown in Fig. 5b). This indicates that ReS2 is not obtained at 900 °C due to the facile desorption of Re atoms from Au(111) because of their low adsorption energy. However, when the W–Re alloy was used as the support substrate, four distinct peaks arising from the E2g, A1g modes of both ReS2 and WS2 are observed (Fig. 5c). This tells us that stacked ReS2/WS2 heterostructures have formed and this is attributed to the preferential growth of WS2 on Au and then the subsequent growth of ReS2 on WS2(001) as discussed above. The data confirms that Re atoms are adsorbed on the WS2(001) surface enabling nucleation and growth of ReS2. In addition, it should be noted that the reaction temperature of 900 °C is very crucial for the dominance of the twinned growth behaviour in the process (Supplementary Fig. 7).
Finally, we demonstrate the hydrogen evolution reaction activity for our ReS2/WS2 twinned vertically stacked heterostructures (Supplementary Fig. 8). The data show that our twinned heterostructures exhibit superior hydrogen evolution reaction activity as compared to pure WS2 and own great potential as a catalytic material. Besides, our ReS2/WS2 twinned vertical heterostructures also show better transfer performance than individual ReS2 (Supplementary Fig. 9). Furthermore, the strategy developed here is versatile as a general method for fabricating other TMDCs heterostructures. For example, MoS2/WS2 vertical heterostructures could be fabricated using Mo-W alloy foils in place of the W–Re alloy foils during the growth process (Supplementary Fig. 10).
In summary, the use of a W–Re alloy foil as a supply source of Re and W atoms, along with the difference in the adsorption energies of Re and W atoms on Au(111) surfaces allows us to demonstrate the possibility of twinned growth between 2D nanomaterials. Importantly, the presented strategy is not limited to ReS2 and WS2, which also allows the fabrication of other TMDCs vertical heterostructures such as MoS2/WS2. Besides, the strategy can be extended to any 2D material whose precursor possesses an appropriate adsorption energy at the required surface. A key factor is the correct choice of the reaction temperature to enable the selective growth of one nanomaterial over another. This type of 2D twinned vertically stacked heterostructures has a 100% overlap and their crystal sizes are one order of magnitude larger than previous reports. The simplicity of the process may be expanded to construct other vertically stacked or in-plane 2D heterostructures, thus, advancing research in the emerging field of 2D van der Waals heterostructures as well as the fundamental understanding of the nucleation and growth of TMDCs.
Methods
The growth of ReS2/WS2 twinned vertical heterostructures
To achieve the one-step growth of ReS2/WS2 twinned vertically stacked heterostructures, we utilized a re-solidified Au substrate in which Re and W atoms had been dissolved in. To prepare the special substrate, a piece of Au wire was placed on W–Re alloy foil. Under the protection of an Ar/H2 atmosphere, the Au spread evenly over the entire foil by annealing at 1,100 °C for ∼10 min which also allows Re and W atoms diffuse into the Au lattice. After that, the temperature was decreased to 900 °C for the CVD growth reaction. The simultaneous growth of ReS2/WS2 layers forming a vertically stacked heterostructure starts upon the introduction of H2S into the system for 10 min. This simple, scalable synthesis process is a direct CVD growth approach with no intermediate transfer steps.
Characterization
Scanning electron microscope images were taken by Zeiss Sigma. Optical images were taken with an optical microscope (Olympus DX51). Raman spectroscopy and photoluminescence with an excitation wavelength of 532 nm were carried out using a Renishaw inVia. The atomic force microscope images were collected on an NT-MDT Ntegra Spectra. The ReS2/WS2 was transferred onto the 300 nm SiO2/Si for such measurements. X-ray photoelectron spectroscopy was performed on a Thermo Scientific, ESCALAB 250Xi. The measuring spot size was 500 μm and the binding energies were calibrated by referencing the C 1 s peak (284.8 eV). The TEM images were taken with an aberration corrected, high-resolution TEM (AC-HRTEM, FEI Titan3) operating at 80 kV. X-ray diffraction measurements were performed using a Rigaku MiniFlex600 with Cu–Ka radiation over the range of 2θ=10∼80°.
DFT calculations
The spin polarized density functional theory calculations were performed using the DMol3 (ref. 32) module in the Materials Studio software (Bio Accelrys). Exchange-correlation functional was based on Perdew-Burke-Ernzerh (PBE) within the generalized gradient approximation. The core electrons were treated with DFT semi-core pseudopotentials. The optimized lattice constant of Au is 4.180 Å, in good agreement with the experimental value of 4.078 Å. The Au(111) facet was modelled by an unreconstructed 4 × 4 surface slab, which contains four atomic layers with a vacuum region of ∼20 Å. The self-consistent-field (SCF) convergence criterion was set to be <10−5 Hartree and the force convergence was set to be lower than 0.002 Hartree per Å for all the optimizations of the adsorptions. In particular, for the optimization of the adsorption of Re atoms and W atoms on Au(111) facets, the bottom two layers of Au atoms were fixed at their bulk positions. A 5 × 5 × 1 k-point mesh was used to sample the Brillouin zone of the supercells. The adsorption energy of a X (X=Re or W) atom on the surfaces of M (M=Au(111) or WS2(001)) substrate is defined as EadsX, M=E(M)+E(X)−E(M−X).
Data availability
The data that support the findings of this study are available from the corresponding author upon request.
Additional information
How to cite this article: Zhang, T. et al. Twinned growth behaviour of two-dimensional materials. Nat. Commun. 7, 13911 doi: 10.1038/ncomms13911 (2016).
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
References
Xu, M., Liang, T., Shi, M. & Chen, H. Graphene-like two-dimensional materials. Chem. Rev. 113, 3766–3798 (2013).
Splendiani, A. et al. Emerging photoluminescence in monolayer MoS2 . Nano Lett. 10, 1271–1275 (2010).
Wang, X. S., Feng, H. B., Wu, Y. M. & Jiao, L. Y. Controlled synthesis of highly crystalline MoS2 flakes by chemical vapor deposition. J. Am. Chem. Soc. 135, 5304–5307 (2013).
Baugher, B. W. H., Churchill, H. O. H., Yang, Y. F. & Herrero, P. J. Intrinsic electronic transport properties of high-quality monolayer and bilayer MoS2 . Nano Lett. 13, 4212–4216 (2013).
He, K., Poole, C., Mak, K. F. & Shan, J. Experimental demonstration of continuous electronic structure tuning via strain in atomically thin MoS2 . Nano Lett. 13, 2931–2936 (2013).
Wang, Q. H., Kalantar-Zadeh, K., Kis, A., Coleman, J. N. & Strano, M. S. Electronics and optoelectronics of two-dimensional transition metal dichalcogenides. Nat. Nanotechnol. 7, 699–712 (2012).
Zeng, H. L., Dai, J. F., Yao, W., Xiao, D. & Cui, X. D. Valley polarization in MoS2 monolayers by optical pumping. Nat. Nanotechnol. 7, 490–493 (2012).
Baugher, B. W. H., Churchill, H. O. H., Yang, Y. & Jarillo-Herrero, P. Optoelectronic devices based on electrically tunable p-n diodes in a monolayer dichalcogenide. Nat. Nanotechnol. 9, 262–267 (2014).
Cheng, R. et al. Few-layer molybdenum disulfide transistors and circuits for high-speed flexible electronics. Nat. Commun. 5, 5143 (2014).
Jariwala, D., Sangwan, V. K., Lauhon, L. J., Marks, T. J. & Hersam, M. C. Emerging device applications for semiconducting two-dimensional transition metal dichalcogenides. ACS Nano 8, 1102–1120 (2014).
Geim, A. K. & Grigorieva, I. V. Van der waals heterostructures. Nature 499, 419–425 (2013).
Hong, X. P. et al. Ultrafast charge transfer in atomically thin MoS2/WS2 heterostructures. Nat. Nanotechnol. 9, 682–686 (2014).
Chiu, M. H. et al. Spectroscopic signatures for interlayer coupling in MoS2-WSe2 van der Waals stacking. ACS Nano 8, 9649–9656 (2014).
Rivera, P. et al. Observation of long-lived interlayer excitons in monolayer MoSe2-WSe2 heterostructures. Nat. Commun. 6, 6242 (2015).
Rigosi, A. F., Hill, H. M., Li, Y., Chernikov, A. & Heinz, T. F. Probing interlayer interactions in transition metal dichalcogenide heterostructures by optical spectroscopy: MoS2/WS2 and MoSe2/WSe2 . Nano Lett. 15, 5033–5038 (2015).
Li, M. Y. et al. Epitaxial growth of a monolayer WSe2-MoS2 lateral p-n junction with an atomically sharp interface. Science 349, 524–528 (2015).
Bellus, M. Z., Ceballos, F., Chiu, H. Y. & Zhao, H. Tightly bound trions in transition metal dichalcogenide heterostructures. ACS Nano 9, 6459–6464 (2015).
Yu, Y. et al. Equally efficient inter layer exciton relaxation and improved absorption in epitaxial and nonepitaxial MoS2/WS2 heterostructures. Nano Lett. 15, 486–491 (2015).
Roy, T. et al. Dual-gated MoS2/WSe2 van der Waals tunnel diodes and transistors. ACS Nano 9, 2071–2079 (2015).
Fang, H. et al. Strong interlayer coupling in van der Waals heterostructures built from single-layer chalcogenides. Proc. Natl Acad. Sci. USA 111, 6198–6202 (2014).
Gong, Y. J. et al. Vertical and in-plane heterostructures from WS2/MoS2 monolayers. Nat. Mater. 13, 1135–1142 (2014).
Tongay, S. et al. Tuning interlayer coupling in large-area heterostructures with CVD grown MoS2 and WS2 monolayers. Nano Lett. 14, 3185–3190 (2014).
Chen, K. et al. Electronic properties of MoS2-WS2 heterostructures synthesized with two-step lateral epitaxial strategy. ACS Nano 9, 9868–9876 (2015).
Yoo, Y., Degregorio, Z. P. & Johns, J. E. Seed crystal homogeneity controls lateral and vertical heteroepitaxy of monolayer MoS2 and WS2 . J. Am. Chem. Soc. 137, 14281–14287 (2015).
Chen, K. et al. Lateral built-in potential of monolayer MoS2-WS2 in-plane heterostructures by a shortcut growth strategy. Adv. Mater. 27, 6431–6437 (2015).
Chiu, M. H. et al. Determination of band alignment in the single-layer MoS2/WSe2 heterojunction. Nat. Commun. 6, 7666 (2015).
Tongay, S. et al. Monolayer behaviour in bulk ReS2 due to electronic and vibrational decoupling. Nat. Commun. 5, 3252 (2014).
Berkdemir, A. et al. Identification of individual and few layers of WS2 using Raman spectroscopy. Sci. Rep. 3, 1755 (2013).
Gao, Y. et al. Large-area synthesis of high-quality and uniform monolayer WS2 on reusable Au foils. Nat. Commun. 6, 8569 (2015).
Keyshar, K. et al. Chemical vapor deposition of monolayer rhenium disulfide (ReS2). Adv. Mater. 27, 4640–4648 (2015).
Shi, J. P. et al. Substrate facet effect on the growth of monolayer MoS2 on Au foils. ACS Nano 9, 4017–4025 (2015).
Delley, B. From molecules to solids with the DMol3 approach. J. Chem. Phys. 113, 7756–7764 (2000).
Acknowledgements
The research was supported by the Natural Science Foundation of China (Grants 21673161, 21473124), the Sino-German Center for Research Promotion (Grant GZ 871). We thank Prof Hongxing Xu for the PL characterizations and Prof Hexiang Deng who assists in the X-ray diffraction characterization. We thank Prof Lei Liao for the FET fabrication. And we thank Dina Bieberstein in Thomas Gemming’s group for the preparation of the cross-sectional TEM samples.
Author information
Authors and Affiliations
Contributions
L.F. developed the concept and conceived the experiments. T.Z. and B.J. carried out the experiments. L.F. and T.Z. wrote the manuscript. L.F., T.Z., B.J., Z.X., R.G.M., Y.X., L.F.C., L.W.F., T.G., S.L.C., M.H.R. contributed to the data analysis and scientific discussion.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures. (PDF 1303 kb)
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Zhang, T., Jiang, B., Xu, Z. et al. Twinned growth behaviour of two-dimensional materials. Nat Commun 7, 13911 (2016). https://doi.org/10.1038/ncomms13911
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/ncomms13911
This article is cited by
-
Anisotropic monolayer of ReX2 on Au foils for exploring abnormal growth behavior and electronic properties
Nano Research (2023)
-
WS2 moiré superlattices derived from mechanical flexibility for hydrogen evolution reaction
Nature Communications (2021)
-
Recent progress about 2D metal dichalcogenides: Synthesis and application in photodetectors
Nano Research (2021)
-
The coupling effect characterization for van der Waals structures based on transition metal dichalcogenides
Nano Research (2021)
-
Two-dimensional Janus van der Waals heterojunctions: A review of recent research progresses
Frontiers of Physics (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.