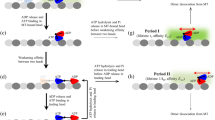
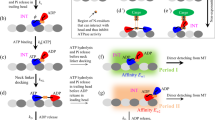
Abstract
Kinesin is a stepping motor that successively produces forward and backward 8-nm steps along microtubules. Under physiological conditions, the steps powering kinesin's motility are biased in one direction and drive various biological motile processes. The physical mechanism underlying the unidirectional bias of the kinesin steps is not fully understood. Here we explored the mechanical kinetics and thermodynamics of forward and backward kinesin steps by analyzing their temperature and load dependence. Results show that the frequency asymmetry between forward and backward steps is produced by entropy. Furthermore, the magnitude of the entropic asymmetry is 6 kBT, more than three times greater than expected from a current model, in which a mechanical conformational change within the kinesin molecular structure directly biases the kinesin steps forward. We propose that the stepping direction of kinesin is preferably caused by an entropy asymmetry resulting from the compatibility between the kinesin and microtubule interaction based on their polar structures.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Vale, R.D. & Milligan, R.A. The way things move: looking under the hood of molecular motor proteins. Science 288, 88–95 (2000).
Howard, J. Molecular motors: structural adaptations to cellular functions. Nature 389, 561–567 (1997).
Cross, R.A. The kinetic mechanism of kinesin. Trends Biochem. Sci. 29, 301–309 (2004).
Endow, S. Kinesin motors as molecular machines. Bioessays 25, 1212–1219 (2003).
Svoboda, K., Schmidt, C.F., Schnapp, B.J. & Block, S.M. Direct observation of kinesin stepping by optical trapping interferometry. Nature 365, 721–727 (1993).
Carter, N.J. & Cross, R.A. Mechanics of the kinesin step. Nature 435, 308–312 (2005).
Hua, W., Young, E.C., Fleming, M.L. & Gelles, J. Coupling of kinesin steps to ATP hydrolysis. Nature 388, 390–393 (1997).
Schnitzer, M.J. & Block, S.M. Kinesin hydrolyses one ATP per 8-nm step. Nature 388, 386–390 (1997).
Nishiyama, M., Higuchi, H. & Yanagida, T. Chemomechanical coupling of the forward and backward steps of single kinesin molecules. Nat. Cell Biol. 4, 790–797 (2002).
Hackney, D.D. Evidence for alternating head catalysis by kinesin during microtubule-stimulated ATP hydrolysis. Proc. Natl. Acad. Sci. USA 91, 6865–6869 (1994).
Kaseda, K., Higuchi, H. & Hirose, K. Alternate fast and slow stepping of a heterodimeric kinesin molecule. Nat. Cell Biol. 5, 1079–1082 (2003).
Yildiz, A., Tomishige, M., Vale, R.D. & Selvin, P.R. Kinesin walks hand-over-hand. Science 303, 676–678 (2004).
Asbury, C.L., Fehr, A.N. & Block, S.M. Kinesin moves by an asymmetric hand-over-hand mechanism. Science 302, 2130–2134 (2003).
Case, R.B., Pierce, D.W., Hom-Booher, N., Hart, C.L. & Vale, R.D. The directional preference of kinesin motors is specified by an element outside of the motor catalytic domain. Cell 90, 959–966 (1997).
Rice, S. et al. A structural change in the kinesin motor protein that drives motility. Nature 402, 778–784 (1999).
Rice, S. et al. Thermodynamic properties of the kinesin neck-region docking to the catalytic core. Biophys. J. 84, 1844–1854 (2003).
Schnitzer, M.J., Visscher, K. & Block, S.M. Force production by single kinesin motors. Nat. Cell Biol. 2, 718–723 (2000).
Block, S.M., Asbury, C.L., Shaevitz, J.W. & Lang, M.J. Probing the kinesin reaction cycle with a 2D optical force clamp. Proc. Natl. Acad. Sci. USA 100, 2351–2356 (2003).
Wang, M. et al. Force and velocity measured for single molecules of RNA polymerase. Science 282, 902–907 (1998).
Mazumdar, M. & Cross, R.A. Engineering a lever into the kinesin neck. J. Biol. Chem. 273, 29352–29359 (1998).
Eyring, H. The activated complex in chemical reactions. J. Chem. Phys. 3, 107–115 (1935).
Atkins, P.W. Physical Chemistry 3rd edn. Ch. 30, 737–761 (Oxford University Press, Oxford, UK, 1987).
Yang, W.Y. & Gruebele, M. Folding at the speed limit. Nature 423, 193–197 (2003).
Kojima, H., Muto, E., Higuchi, H. & Yanagida, T. Mechanics of single kinesin molecules measured by optical trapping nanometry. Biophys. J. 73, 2012–2022 (1997).
Nishiyama, M. et al. Substeps within the 8-nm step of the ATPase cycle of single kinesin molecules. Nat. Cell Biol. 3, 425–428 (2001).
Kawaguchi, K. & Ishiwata, S. Temperature dependence of force, velocity, and processivity of single kinesin molecules. Biochem. Biophys. Res. Commun. 272, 895–899 (2000).
Nishiyama, M., Higuchi, H., Ishii, Y., Taniguchi, Y. & Yanagida, T. Single molecule processes on the stepwise movement of ATP-driven molecular motors. Biosystems 71, 145–156 (2003).
Böhm, K. et al. Effect of temperature on kinesin-driven microtubule gliding and kinesin ATPase activity. FEBS Lett. 466, 59–62 (2000).
Kawaguchi, K. & Ishiwata, S. Thermal activation of single kinesin molecules with temperature pulse microscopy. Cell Motil. Cytoskeleton 49, 41–47 (2001).
Fersht, A.R. Structure and Mechanism in Protein Science (W.H. Freeman, New York, 1999).
Hirose, K., Lockhart, A., Cross, R.A. & Amos, L.A. Three-dimensional cytoelectron microscopy of dimeric kinesin and ncd motor domains on microtubules. Proc. Natl. Acad. Sci. USA 93, 9539–9544 (1996).
Hirose, K., Löwe, J., Alonso, M., Cross, R.A. & Amos, L.A. Congruent docking of dimeric kinesin and ncd into three-dimensional electron cryomicroscopy maps of microtubule-motor ADP complexes. Mol. Biol. Cell 10, 2063–2074 (1999).
Mandelkow, E. & Hoenger, A. Structures of kinesin and kinesin-microtubule interactions. Curr. Opin. Cell Biol. 11, 34–44 (1999).
Uemura, S. & Ishiwata, S. Loading direction regulates the affinity of ADP for kinesin. Nat. Struct. Biol. 10, 308–311 (2003).
Kawaguchi, K., Uemura, S. & Ishiwata, S. Equilibrium and transition between single- and double-headed binding of kinesin as revealed by single-molecule mechanics. Biophys. J. 84, 1103–1113 (2003).
Ali, M.Y. et al. Unconstrained steps of myosin VI appear longest among known molecular motors. Biophys. J. 86, 3804–3810 (2004).
Kitamura, K., Tokunaga, M., Esaki, S., Iwane, A.H. & Yanagida, T. Mechanism of muscle contraction based on stochastic properties of single actomyosin motors observed in vitro. Biophysics 1, 1–19 (2005).
Svoboda, K. & Block, S.M. Force and velocity measured for single kinesin molecules. Cell 77, 773–784 (1994).
William, H.P., Saul, A.T., William, T.V. & Brian, P.F. Numerical Recipes in C 2nd edn. (Cambridge University Press, New York, 1992).
Rief, M. et al. Myosin-V stepping kinetics: a molecular model for processivity. Proc. Natl. Acad. Sci. USA 97, 9482–9486 (2000).
Altman, D., Sweeney, H.L. & Spudich, J.A. The mechanism of myosin VI translocation and its load-induced anchoring. Cell 116, 737–749 (2004).
Howard, J. Mechanics of Motor Proteins and the Cytoskeleton (Sinauer Associates, Sunderland, Massachusetts, 2001).
Kull, F.J. et al. Crystal structure of the kinesin motor domain reveals a structural similarity to myosin. Nature 380, 550–555 (1996).
Tadakuma, H., Yamaguchi, J., Ishihara, Y. & Funatsu, T. Imaging of single fluorescent molecules using video-rate confocal microscopy. Biochem. Biophys. Res. Commun. 287, 323–327 (2001).
Acknowledgements
We thank F. Oosawa, T. Kodama, K. Sekimoto, H. Higuchi, A. Ishijima, H. Kojima, T. Ariga and colleagues at Osaka University and Japan Science and Technology Agency for valuable discussions and P. Karagiannis and J. West for carefully revising the manuscript.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Fig. 1
Step size and direction of kinesin steps. (PDF 38 kb)
Supplementary Fig. 2
Distribution of dwell times of the forward (blue) and backward (red) steps. T = 35 °C. (PDF 31 kb)
Rights and permissions
About this article
Cite this article
Taniguchi, Y., Nishiyama, M., Ishii, Y. et al. Entropy rectifies the Brownian steps of kinesin. Nat Chem Biol 1, 342–347 (2005). https://doi.org/10.1038/nchembio741
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nchembio741
This article is cited by
-
Experimental and theoretical energetics of walking molecular motors under fluctuating environments
Biophysical Reviews (2020)
-
Reconceptualizing kinesin’s working cycle as separate chemical and mechanical processes
Frontiers of Physics (2018)
-
A mechanical model for the role of the neck linker during kinesin stepping and gating
Science China Physics, Mechanics and Astronomy (2011)
-
Impact of Slip Cycles on the Operation Modes and Efficiency of Molecular Motors
Journal of Statistical Physics (2010)
-
Direct observation of the binding state of the kinesin head to the microtubule
Nature (2009)