Abstract
The tumour vascular microenvironment supports tumorigenesis not only by supplying oxygen and diffusible nutrients but also by secreting soluble factors that promote tumorigenesis. Here we identify a feedforward mechanism in which endothelial cells (ECs), in response to tumour-derived mediators, release angiocrines driving aberrant vascularization and glioblastoma multiforme (GBM) progression through a hypoxia-independent induction of hypoxia-inducible factor (HIF)-1α. Phosphorylation of profilin-1 (Pfn-1) at Tyr 129 in ECs induces binding to the tumour suppressor protein von Hippel–Lindau (VHL), and prevents VHL-mediated degradation of prolyl-hydroxylated HIF-1α, culminating in HIF-1α accumulation even in normoxia. Elevated HIF-1α induces expression of multiple angiogenic factors, leading to vascular abnormality and tumour progression. In a genetic model of GBM, mice with an EC-specific defect in Pfn-1 phosphorylation exhibit reduced tumour angiogenesis, normalized vasculature and improved survival. Moreover, EC-specific Pfn-1 phosphorylation is associated with tumour aggressiveness in human glioma. These findings suggest that targeting Pfn-1 phosphorylation may offer a selective strategy for therapeutic intervention of malignant solid tumours.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Hanahan, D. & Weinberg, R. A. Hallmarks of cancer: the next generation. Cell 144, 646–674 (2011).
Vaupel, P., Kallinowski, F. & Okunieff, P. Blood flow, oxygen and nutrient supply, and metabolic microenvironment of human tumors: a review. Cancer Res. 49, 6449–6465 (1989).
Joyce, J. A. & Pollard, J. W. Microenvironmental regulation of metastasis. Nat. Rev. Cancer 9, 239–252 (2009).
Folkman, J. Tumor angiogenesis: therapeutic implications. N. Engl. J. Med. 285, 1182–1186 (1971).
Jain, R. K. Normalizing tumor vasculature with anti-angiogenic therapy: a new paradigm for combination therapy. Nat. Med. 7, 987–989 (2001).
Carmeliet, P. & Jain, R. K. Principles and mechanisms of vessel normalization for cancer and other angiogenic diseases. Nat. Rev. Drug Discov. 10, 417–427 (2011).
Weis, S. M. & Cheresh, D. A. Tumor angiogenesis: molecular pathways and therapeutic targets. Nat. Med. 17, 1359–1370 (2011).
Burger, P. C., Vogel, F. S., Green, S. B. & Strike, T. A. Glioblastoma multiforme and anaplastic astrocytoma. Pathologic criteria and prognostic implications. Cancer 56, 1106–1111 (1985).
Johnson, D. R. & O’Neill, B. P. Glioblastoma survival in the United States before and during the temozolomide era. J. Neurooncol. 107, 359–364 (2012).
Huse, J. T. & Holland, E. C. Targeting brain cancer: advances in the molecular pathology of malignant glioma and medulloblastoma. Nat. Rev. Cancer 10, 319–331 (2010).
Kim, K. J. et al. Inhibition of vascular endothelial growth factor-induced angiogenesis suppresses tumour growth in vivo. Nature 362, 841–844 (1993).
Batchelor, T. T. et al. AZD2171, a pan-VEGF receptor tyrosine kinase inhibitor, normalizes tumor vasculature and alleviates edema in glioblastoma patients. Cancer Cell 11, 83–95 (2007).
Friedman, H. S. et al. Bevacizumab alone and in combination with irinotecan in recurrent glioblastoma. J. Clin. Oncol. 27, 4733–4740 (2009).
Witke, W. The role of profilin complexes in cell motility and other cellular processes. Trends Cell Biol. 14, 461–469 (2004).
Fan, Y. et al. Stimulus-dependent phosphorylation of profilin-1 in angiogenesis. Nat. Cell Biol. 14, 1046–1056 (2012).
Charles, N. et al. Perivascular nitric oxide activates notch signaling and promotes stem-like character in PDGF-induced glioma cells. Cell Stem Cell 6, 141–152 (2010).
Galban, S. et al. DW-MRI as a biomarker to compare therapeutic outcomes in radiotherapy regimens incorporating temozolomide or gemcitabine in glioblastoma. PLoS ONE 7, e35857 (2012).
McConville, P. et al. Magnetic resonance imaging determination of tumor grade and early response to temozolomide in a genetically engineered mouse model of glioma. Clin. Cancer Res. 13, 2897–2904 (2007).
Jain, R. K. Molecular regulation of vessel maturation. Nat. Med. 9, 685–693 (2003).
Forsythe, J. A. et al. Activation of vascular endothelial growth factor gene transcription by hypoxia-inducible factor 1. Mol. Cell Biol. 16, 4604–4613 (1996).
Calvani, M., Rapisarda, A., Uranchimeg, B., Shoemaker, R. H. & Melillo, G. Hypoxic induction of an HIF-1α-dependent bFGF autocrine loop drives angiogenesis in Human endothelial cells. Blood 107, 2705–2712 (2006).
Schofield, C. J. & Ratcliffe, P. J. Oxygen sensing by HIF hydroxylases. Nat. Rev. Mol. Cell Biol. 5, 343–354 (2004).
Min, J.H. et al. Structure of an HIF-1α-pVHL complex: Hydroxyproline recognition in signaling. Science 296, 1886–1889 (2002).
Keith, B. & Simon, M. C. Hypoxia-inducible factors, stem cells, and cancer. Cell 129, 465–472 (2007).
Semenza, G. L. Intratumoral hypoxia, radiation resistance, and HIF-1. Cancer Cell 5, 405–406 (2004).
Plate, K. H., Breier, G., Weich, H. A. & Risau, W. Vascular endothelial growth factor is a potential tumour angiogenesis factor in human gliomas in vivo. Nature 359, 845–848 (1992).
Dunn, I. F., Heese, O. & Black, P. M. Growth factors in glioma angiogenesis: FGFs, PDGF, EGF, and TGFs. J. Neurooncol. 50, 121–137 (2000).
Nyberg, P., Salo, T. & Kalluri, R. Tumor microenvironment and angiogenesis. Front. Biosci. 13, 6537–6553 (2008).
Park, J. H. et al. Gastric epithelial reactive oxygen species prevent normoxic degradation of hypoxia-inducible factor-1alphaup in gastric cancer cells. Clin. Cancer Res. 9, 433–440 (2003).
Sumbayev, V. V. LPS-induced Toll-like receptor 4 signalling triggers cross-talk of apoptosis signal-regulating kinase 1 (ASK1) and HIF-1alphaup protein. FEBS Lett. 582, 319–326 (2008).
Selak, M. A. et al. Succinate links TCA cycle dysfunction to oncogenesis by inhibiting HIF-α prolyl hydroxylase. Cancer Cell 7, 77–85 (2005).
Tannahill, G. M. et al. Succinate is an inflammatory signal that induces IL-1β through HIF-1α. Nature 496, 238–242 (2013).
Laughner, E., Taghavi, P., Chiles, K., Mahon, P. C. & Semenza, G. L. HER2 (neu) signaling increases the rate of hypoxia-inducible factor 1alphaup (HIF-1α) synthesis: Novel mechanism for HIF-1-mediated vascular endothelial growth factor expression. Mol. Cell Biol. 21, 3995–4004 (2001).
Li, Y. M. et al. A hypoxia-independent hypoxia-inducible factor-1 activation pathway induced by phosphatidylinositol-3 kinase/Akt in HER2 overexpressing cells. Cancer Res. 65, 3257–3263 (2005).
Koka, V. et al. Role of Her-2/neu overexpression and clinical determinants of early mortality in glioblastoma multiforme. Am. J. Clin. Oncol. 26, 332–335 (2003).
Potti, A. et al. Determination of HER-2/neu overexpression and clinical predictors of survival in a cohort of 347 patients with primary malignant brain tumors. Cancer Invest. 22, 537–544 (2004).
Haynik, D. M., Roma, A. A. & Prayson, R. A. HER-2/neu expression in glioblastoma multiforme. Appl. Immunohistochem. Mol. Morphol. 15, 56–58 (2007).
Ding, B. S. et al. Inductive angiocrine signals from sinusoidal endothelium are required for liver regeneration. Nature 468, 310–315 (2010).
Ding, B. S. et al. Endothelial-derived angiocrine signals induce and sustain regenerative lung alveolarization. Cell 147, 539–553 (2011).
Muramatsu, R. et al. Angiogenesis induced by CNS inflammation promotes neuronal remodeling through vessel-derived prostacyclin. Nat. Med. 18, 1658–1664 (2012).
Lu, J. et al. Endothelial cells promote the colorectal cancer stem cell phenotype through a soluble form of jagged-1. Cancer Cell 23, 171–185 (2013).
Lee, S. et al. Autocrine VEGF signaling is required for vascular homeostasis. Cell 130, 691–703 (2007).
Seghezzi, G. et al. Fibroblast growth factor-2 (FGF-2) induces vascular endothelial growth factor (VEGF) expression in the endothelial cells of forming capillaries: an autocrine mechanism contributing to angiogenesis. J. Cell Biol. 141, 1659–1673 (1998).
Butler, J. M., Kobayashi, H. & Rafii, S. Instructive role of the vascular niche in promoting tumour growth and tissue repair by angiocrine factors. Nat. Rev. Cancer 10, 138–146 (2010).
Kreisl, T. N. et al. Phase II trial of single-agent bevacizumab followed by bevacizumab plus irinotecan at tumor progression in recurrent glioblastoma. J. Clin. Oncol. 27, 740–745 (2009).
Fan, Y., Gong, Y., Ghosh, P. K., Graham, L. M. & Fox, P. L. Spatial coordination of actin polymerization and ILK-Akt2 activity during endothelial cell migration. Dev. Cell 16, 661–674 (2009).
Janke, J. et al. Suppression of tumorigenicity in breast cancer cells by the microfilament protein profilin 1. J. Exp. Med. 191, 1675–1686 (2000).
Ding, Z., Gau, D., Deasy, B., Wells, A. & Roy, P. Both actin and polyproline interactions of profilin-1 are required for migration, invasion and capillary morphogenesis of vascular endothelial cells. Exp. Cell Res. 315, 2963–2973 (2009).
Venneri, M. A. et al. Identification of proangiogenic TIE2-expressing monocytes (TEMs) in human peripheral blood and cancer. Blood 109, 5276–5285 (2007).
De Palma, M. et al. Tie2 identifies a hematopoietic lineage of proangiogenic monocytes required for tumor vessel formation and a mesenchymal population of pericyte progenitors. Cancer Cell 8, 211–226 (2005).
Pyonteck, S. M. et al. CSF-1R inhibition alters macrophage polarization and blocks glioma progression. Nat. Med. 19, 1264–1272 (2013).
Liu, Y. et al. Somatic cell type specific gene transfer reveals a tumor-promoting function for p21(Waf1/Cip1). EMBO J. 26, 4683–4693 (2007).
Ciznadija, D., Liu, Y., Pyonteck, S. M., Holland, E. C. & Koff, A. Cyclin D1 and cdk4 mediate development of neurologically destructive oligodendroglioma. Cancer Res. 71, 6174–6183 (2011).
Liberzon, A. et al. Molecular signatures database (MSigDB) 30. Bioinformatics 27, 1739–1740 (2011).
Benita, Y. et al. An integrative genomics approach identifies Hypoxia Inducible Factor-1 (HIF-1)-target genes that form the core response to hypoxia. Nucleic Acids Res. 37, 4587–4602 (2009).
Acknowledgements
We are grateful to A. Horowitz for helpful suggestions, E. Ritchie for technical assistance, and J. Drazba for image analysis. This work was supported in part by National Institutes of Health grants K99 HL103792 (to Y.F.), and P01 HL029582, P01 HL076491 and R21 HL094841 (to P.L.F.).
Author information
Authors and Affiliations
Contributions
Y.F. designed, performed and analysed experiments, produced figures, and wrote the initial draft of the paper. A.A.P. performed the gene array analysis and generated Supplementary Fig. 3. Y.G. contributed to the immunohistochemistry analysis in Fig. 5. S.D. and D.H. performed mouse GBM experiments. J.D.L., S.M.E. and J.N.R. helped write and edit the late drafts of the manuscript. P.L.F designed, supervised and analysed experiments and wrote the final draft of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
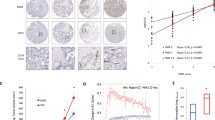
Supplementary Figure 1 Multiplex analysis of Pfn-1 phosphorylation at Tyr129 in human tumours.
Human tissue sections retrieved from biopsy specimens from subjects with tumours and normal tissue controls were stained with anti-P-Pfn-1-Tyr129 antibody. Fluorescence intensity of P-Pfn-1-Tyr129 was quantified by immunofluorescence (mean ± s.e.m, total n = 368 patient specimens, two-tailed unpaired t test).
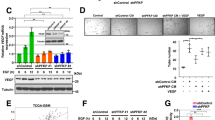
Supplementary Figure 2 Pfn-1 phosphorylation regulates expression of multiple angiogenic factors in EC.
Mouse aortic EC were isolated from Pfn1flox/flox:Y129F (Pfn-1WT) and Tie2–Cre;Pfn1flox/flox:Y129F (Pfn-1Y129F) mice, and incubated with glioma cells-conditioned medium (GCM) for 8 h. Cell lysates incubated with antibody-captured membranes of Protome Profiler Array, and immunoblotted with streptavidin-HRP. (a) The list of analysed angiogenic factors. (b) Signal densitometry was analysed by using NIH ImageJ software (mean, two replicates in one experiment). The fold-change in EC expressing Pfn-1Y129F compared with EC expressing Pfn-1WT is indicated.
Supplementary Figure 3 Pfn-1 phosphorylation is critical for global expression of HIF-1α-inducible genes in primary GBM microvascular EC.
GBM was induced by orthotopic injection of GBM tumour cells in Pfn1flox/flox:Y129F (Pfn1WT) and Tie2–Cre;Pfn1flox/flox:Y129F (Pfn1Y129F) mice (n =3 for each). Tumours were excised and digested to generate single-cell suspension. CD105+ EC were isolated by magnetic-activated cell sorting (MACS), and treated with glioma-conditioned medium. mRNA was isolated and subjected to array analysis (Affymetrix Mouse Gene 2.0 ST). Heat map represents the genes in the microarray regulated by HIF-1 (list compiled from literature, see methods). The color-coded scale for the normalized expression value is shown at the top of the figure. The genes are arranged in order of increasing fold changes (ratio of Pfn1Y129F to Pfn1WT expression level). The insets on the right at the top and bottom indicate the downregulated (P < 0.05, fold change < 0.66) and upregulated (P < 0.05, fold change >1.5) genes in the Pfn1Y129F mice respectively. The 42 downregulated genes are shown on the left.
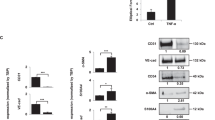
Supplementary Figure 4 Phospho-Pfn-1 interacts with VHL.Purified Pfn-1-His were in vitro phosphorylated with or without purified Src kinase and re-purified with Ni+-beads.
Pfn-1 interaction with chip-immobilized VHL was determined by SPR.
Supplementary Figure 5 VHL competes with G-actin for Pfn-1 binding.
Purified Pfn-1-His were in vitro phosphorylated with lysate from GCM-treated EC and re-purified with Ni+-beads. Pfn-1 interaction with chip-immobilized VHL in the presence or absence of G-actin was determined by SPR.
Supplementary Figure 6 HIF-1α is critical for GCM-induced Pfn-1 phosphorylation.
Human microvascular EC were transfected with control or HIF-1α siRNA, and treated with glioma-conditioned medium (GCM) for 24 h. Cell lysate was resolved by SDS-PAGE and subjected to immunoblot analysis.
Supplementary Figure 7 Pfn1 mRNA is elevated in glioblastoma and its level correlates with glioma patient survival.
(a) Analysis of Pfn1 mRNA expression in normal brain and glioblastoma tissue. Databases of oncomine (http://www.oncomine.com) were analysed from three independent studies including the cancer genome atlas (TCGA) of National Cancer Institute and the studies by Murat (Murat et al, J Clin Oncol, 2008; 26: 3015–3024) and Sun (Sun et al, Cancer Cell 2006; 9: 287–300). Results are shown as box plots representing median, 25th and 75th percentiles as boxes, 10th and 90th percentiles as bars, and the range of data as dots. Total n = 799 patients. Statistical significance was determined by students t-test (b, c) Rembrandts database of the National Cancer Institute (http://caintegrator.nci.nih.gov/rembrandt) was analysed. (b) mRNA expression analysis of actin-binding proteins in normal brain and glioblastoma tissue. Results are shown as box plots representing median, 25th and 75th percentiles as boxes, and the range of data as bars. Total n = 256 patients. Statistical significance was determined by students t-test. (c) Analysis of survival rate in glioma patients with intermediate and upregulated Pfn1 mRNA expression. Total n = 343 patients.
Supplementary information
Supplementary Information
Supplementary Information (PDF 937 kb)
Rights and permissions
About this article
Cite this article
Fan, Y., Potdar, A., Gong, Y. et al. Profilin-1 phosphorylation directs angiocrine expression and glioblastoma progression through HIF-1α accumulation. Nat Cell Biol 16, 445–456 (2014). https://doi.org/10.1038/ncb2954
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb2954
This article is cited by
-
Profilin 1 deficiency drives mitotic defects and reduces genome stability
Communications Biology (2023)
-
Targeting PAK4 to reprogram the vascular microenvironment and improve CAR-T immunotherapy for glioblastoma
Nature Cancer (2020)
-
NF-kappa B interacting long noncoding RNA enhances the Warburg effect and angiogenesis and is associated with decreased survival of patients with gliomas
Cell Death & Disease (2020)
-
Profilin: many facets of a small protein
Biophysical Reviews (2020)
-
Self-seeding circulating tumor cells promote the proliferation and metastasis of human osteosarcoma by upregulating interleukin-8
Cell Death & Disease (2019)