Abstract
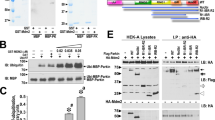
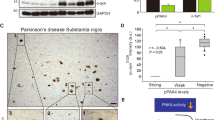
Mutations of the ubiquitin ligase parkin account for most autosomal recessive forms of juvenile Parkinson's disease (AR-JP). Several studies have suggested that parkin possesses DNA-binding and transcriptional activity. We report here that parkin is a p53 transcriptional repressor. First, parkin prevented 6-hydroxydopamine-induced caspase-3 activation in a p53-dependent manner. Concomitantly, parkin reduced p53 expression and activity, an effect abrogated by familial parkin mutations known to either abolish or preserve its ligase activity. ChIP experiments indicate that overexpressed and endogenous parkin interact physically with the p53 promoter and that pathogenic mutations abolish DNA binding to and promoter transactivation of p53. Parkin lowered p53 mRNA levels and repressed p53 promoter transactivation through its Ring1 domain. Conversely, parkin depletion enhanced p53 expression and mRNA levels in fibroblasts and mouse brains, and increased cellular p53 activity and promoter transactivation in cells. Finally, familial parkin missense and deletion mutations enhanced p53 expression in human brains affected by AR-JP. This study reveals a ubiquitin ligase-independent function of parkin in the control of transcription and a functional link between parkin and p53 that is altered by AR-JP mutations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Kitada, T. et al. Mutations in the parkin gene cause autosomal recessive juvenile parkinsonism. Nature 392, 605–608 (1998).
Shimura, H. et al. Familial Parkinson disease gene product, parkin, is a ubiquitin-protein ligase. Nature Genet. 25, 302–305 (2000).
Jiang, H., Ren, Y., Zhao, J. & Feng, J. Parkin protects human dopaminergic neuroblastoma cells against dopamine-induced apoptosis. Hum. Mol. Genet. 13, 1745–1754 (2004).
Pesah, Y. et al. Drosophila parkin mutants have decreased mass and cell size and increased sensitivity to oxygen radical stress. Development 131, 2183–2194 (2004).
Cookson, M. R. et al. RING finger 1 mutations in Parkin produce altered localization of the protein. Hum. Mol. Genet. 12, 2957–2965 (2003).
Horowitz, J. M., Myers, J., Vernace, V. A., Stachowiak, M. K. & Torres, G. Spatial distribution, cellular integration and stage development of Parkin protein in Xenopus brain. Brain Res. Dev. Brain Res. 126, 31–41 (2001).
Morett, E. & Bork, P. A novel transactivation domain in parkin. Trends in Biochem. Sci. 24, 229–231 (1999).
Unschuld, P. G. et al. Parkin modulates gene expression in control and ceramide-treated PC12 cells. Mol. Biol. Rep. 33, 13–32 (2006).
Kamijo, T. et al. Tumor suppression at the mouse INK4a locus mediated by the alternative reading frame product p19ARF. Cell 91, 649–659 (1997).
Sauer, H. & Ortel, W. H. Progressive degeneration of nigrostriatal dopamine neurons following intrastriatal terminal lesions with 6-hydroxydopamine: a combined retrograde tracing and immunocytochemical study in the rat. Neuroscience 59, 401–415 (1994).
Henn, I. H., Gostner, J. M., Lackner, P., Tatzelt, J. & Winklhofer, K. F. Pathogenic mutations inactivate parkin by distinct mechanisms. J. Neurochem. 92, 114–122 (2005).
Datta, S. R., Brunet, A. & Greenberg, M. E. Cellular survival: a play in three Akts. Genes Dev. 13, 2905–2927 (1999).
Jeong, S. J., Pise-Masison, C. A., Radonovich, M. F., Park, H. U. & Brady, J. N. Activated AKT regulates NF-κB activation, p53 inhibition and cell survival in HTLV-1-transformed cells. Oncogene 24, 6719–6728 (2005).
Tatematsu, K. et al. Transcriptional activity of RBCK1 protein (RBCC protein interacting with PKC 1): requirement of RING-finger and B-Box motifs and regulation by protein kinases. Biochem. Biophys. Res. Comm. 247, 392–396 (1998).
Grossman, S. R. et al. Polyubiquitination of p53 by a ubiquitin ligase activity of p300. Science 300, 342–344 (2003).
Esser, C., Scheffner, M. & Hohfeld, J. The chaperone-associated ubiquitin ligase CHIP is able to target p53 for proteasomal degradation. J. Biol. Chem. 280, 27443–27448 (2005).
Leng, R. P. et al. Pirh2, a p53-induced ubiquitin-protein ligase, promotes p53 degradation. Cell 112, 779–791 (2003).
Dornan, D. et al. The ubiquitin ligase COP1 is a critical negative regulator of p53. Nature 429, 86–92 (2004).
Haupt, Y., Maya, R., Kazaz, A. & Oren, M. Mdm2 promotes the rapid degradation of p53. Nature 387, 296–299 (1997).
Xirodimas, D. P., Saville, M. K., Bourdon, J. C., Hay, R. T. & Lane, D. P. Mdm2-mediated NEDD8 conjugation of p53 inhibits its transcriptional activity. Cell 118, 83–97 (2004).
Hattori, N. et al. Point mutations (Thr240Arg and Gln311Stop) [correction of Thr240Arg and Ala311Stop] in the Parkin gene. Biochem. Biophys. Res. Comm. 249, 754–758 (1998).
Sriram, S. R. et al. Familial-associated mutations differentially disrupt the solubility, localization, binding and ubiquitination properties of parkin. Hum. Mol. Genet. 14, 2571–2586 (2005).
Wang, C. et al. Alterations in the solubility and intracellular localization of parkin by several familial Parkinson's disease-linked point mutations. J. Neurochem. 93, 422–431 (2005).
Von Coelln, R. et al. Loss of locus coeruleus neurons and reduced startle in parkin null mice. Proc. Natl Acad. Sci. USA 101, 10744–10749 (2004).
Alves da Costa, C., Ancolio, K. & Checler, F. Wild-type but not Parkinson's disease-related Ala53Thr-α-synuclein protect neuronal cells from apoptotic stimuli. J. Biol. Chem 275, 24065–24069 (2000).
Qin, C. et al. Estrogen up-regulation of p53 gene expression in MCF-7 breast cancer cells is mediated by calmodulin kinase IV-dependent activation of a nuclear factor kappaB/CCAAT-binding transcription factor-1 complex. Mol. Endocrinol. 16, 1793–1809 (2002).
Chung, K. K. et al. Parkin ubiquitinates the alpha-synuclein-interacting protein, synphilin-1: implications for Lewy-body formation in Parkinson disease. Nature Med. 7, 1144–1150 (2001).
Alves da Costa, C. et al. Presenilin-dependent gamma-secretase-mediated control of p53-associated cell death in Alzheimer's disease. J. Neurosci. 26, 6377–6385 (2006).
Goldberg, M. S. et al. Parkin-deficient mice exhibit nigrostriatal deficits but not loss of dopaminergic neurons. J. Biol. Chem. 278, 43628–43635 (2003).
Abmayr, S. M., Yao, T., Parmely, T. & Workman, J. L. Preparation of nuclear and cytoplasmic extracts from mammalian cells. Curr. Protoc. Mol. Biol. Ch 12, Unit 12.1 (2006).
Coleman, J. E. et al. Efficient large-scale production and concentration of HIV-1-based lentiviral vectors for use in vivo. Physiol. Genomics 12, 221–228 (2003).
Acknowledgements
We would like to thank Amanda Patel for helpful discussion concerning gel shift analyses. We wish to thank M. Oren, B. Vogelstein and T. Dawson for providing us with the p53 promoter, PG13 and parkin-deleted constructs. M. Roussel and T. Dawson are thanked for providing the p19Arf–1−/− and p19Arf–1−/−p53−/− and parkin knockout cells, and parkin-deletion constructs, respectively. We thank J. C. Bourdon for providing polyclonal p53-directed antibodies. We wish to thank F. Brau for help in confocal analysis and F. Aguila for artwork. This work was supported by the Fondation pour la Recherche Médicale.
Author information
Authors and Affiliations
Contributions
S.C. and E.G. performed a subset of experiments and contributed equally to the work; A.W. performed lentiviral experiments; O.C. and A.B. provided parkin cDNA; N.W.W. and P.M.A.S. provided 'English' human brain samples; H.T. provided 'Japanese' human brain samples; M.S.G. and J.S. provided parkin-null fibroblasts and brains; S.S. provided p53 promoter constructs; C.A.C. and F.C. are co-senior contributors; C.A.C. designed the study, performed most of the experiments, discussed data and wrote the manuscript; F.C. discussed data and contributed to the writing of the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Information (PDF 1090 kb)
Rights and permissions
About this article
Cite this article
da Costa, C., Sunyach, C., Giaime, E. et al. Transcriptional repression of p53 by parkin and impairment by mutations associated with autosomal recessive juvenile Parkinson's disease. Nat Cell Biol 11, 1370–1375 (2009). https://doi.org/10.1038/ncb1981
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1981
This article is cited by
-
Proteomic Analysis of Protective Effects of Dl-3-n-Butylphthalide against mpp + -Induced Toxicity via downregulating P53 pathway in N2A Cells
Proteome Science (2023)
-
The Role of Ubiquitin–Proteasome System and Mitophagy in the Pathogenesis of Parkinson's Disease
NeuroMolecular Medicine (2023)
-
CBX3 accelerates the malignant progression of glioblastoma multiforme by stabilizing EGFR expression
Oncogene (2022)
-
Involvement of heat shock proteins and parkin/α-synuclein axis in Parkinson’s disease
Molecular Biology Reports (2022)
-
Parkin facilitates proteasome inhibitor-induced apoptosis via suppression of NF-κB activity in hepatocellular carcinoma
Cell Death & Disease (2019)