Abstract
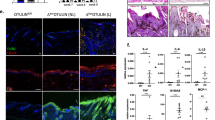
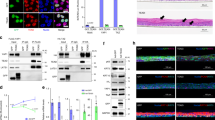
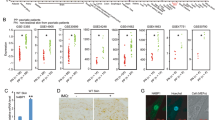
Mice that lack JunB in epidermal cells are born with normal skin; however, keratinocytes hyperproliferate in vitro and on TPA treatment in vivo. Loss of JunB expression in the epidermis of adult mice affects the skin, the proliferation of haematopoietic cells and bone formation. G-CSF is a direct transcriptional target of JunB and mutant epidermis releases large amounts of G-CSF that reach high systemic levels and cause skin ulcerations, myeloproliferative disease and low bone mass. The absence of G-CSF significantly improves hyperkeratosis and prevents the development of myeloproliferative disease, but does not affect bone loss. This study describes a mechanism by which the absence of JunB in epithelial cells causes multi-organ disease, suggesting that the epidermis can act as an endocrine-like organ.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Eferl, R. & Wagner, E. F. AP-1: a double-edged sword in tumorigenesis. Nature Rev. Cancer 3, 859–868 (2003).
Schorpp, M. et al. The human ubiquitin C promoter directs high ubiquitous expression of transgenes in mice. Nucleic Acids Res. 24, 1787–1788 (1996).
Li, B., Tournier, C., Davis, R. J. & Flavell, R. A. Regulation of IL-4 expression by the transcription factor JunB during T helper cell differentiation. EMBO J. 18, 420–432 (1999).
Schorpp-Kistner, M., Wang, Z. Q., Angel, P. & Wagner, E. F. JunB is essential for mammalian placentation. EMBO J. 18, 934–948 (1999).
Passegué, E., Jochum, W., Schorpp-Kistner, M., Mohle-Steinlein, U. & Wagner, E. F. Chronic myeloid leukemia with increased granulocyte progenitors in mice lacking junB expression in the myeloid lineage. Cell 104, 21–32 (2001).
Kenner, L. et al. Mice lacking JunB are osteopenic due to cell-autonomous osteoblast and osteoclast defects. J. Cell Biol. 164, 613–623 (2004).
Hess, J. et al. Defective endochondral ossification in mice with strongly compromised expression of JunB. J Cell Sci. 116, 4587–4596 (2003).
Passegué, E. & Wagner, E. F. JunB suppresses cell proliferation by transcriptional activation of p16(INK4a) expression. EMBO J. 19, 2969–2979 (2000).
Passegué, E., Wagner, E. F. & Weissman, I. L. JunB deficiency leads to a myeloproliferative disorder arising from hematopoietic stem cells. Cell 119, 431–443 (2004).
Licht, A. H. et al. JunB is required for endothelial cell morphogenesis by regulating core-binding factor beta. J. Cell Biol. 175, 981–991 (2006).
Staber, P. B. et al. The oncoprotein NPM–ALK of anaplastic large-cell lymphoma induces JUNB transcription via ERK1/2 and JunB translation via mTOR signaling. Blood 110, 3374–3383 (2007).
Szabowski, A. et al. c-Jun and JunB antagonistically control cytokine-regulated mesenchymal-epidermal interaction in skin. Cell 103, 745–755 (2000).
Angel, P., Szabowski, A. & Schorpp-Kistner, M. Function and regulation of AP-1 subunits in skin physiology and pathology. Oncogene 20, 2413–2423 (2001).
Zenz, R. et al. Activator protein 1 (Fos/Jun) functions in inflammatory bone and skin disease. Arthritis Res Ther. 10, 201 (2008).
Zenz, R. et al. Psoriasis-like skin disease and arthritis caused by inducible epidermal deletion of Jun proteins. Nature 437, 369–375 (2005).
Tarutani, M. et al. Tissue-specific knockout of the mouse Pig-a gene reveals important roles for GPI-anchored proteins in skin development. Proc. Natl Acad. Sci. USA 94, 7400–7405 (1997).
Byrne, C. & Fuchs, E. Probing keratinocyte and differentiation specificity of the human K5 promoter in vitro and in transgenic mice. Mol. Cell Biol. 13, 3176–3190 (1993).
Schmidt, D. et al. Critical role for NF-kappaB-induced JunB in VEGF regulation and tumor angiogenesis. EMBO J. 26, 710–719 (2007).
Lieschke, G. J. et al. Mice lacking granulocyte colony-stimulating factor have chronic neutropenia, granulocyte and macrophage progenitor cell deficiency, and impaired neutrophil mobilization. Blood 84, 1737–1746 (1994).
Kopf, M. et al. Impaired immune and acute-phase responses in interleukin-6-deficient mice. Nature 368, 339–342 (1994).
Kawada, A. et al. Granulocyte and macrophage colony-stimulating factors stimulate proliferation of human keratinocytes. Arch. Dermatol. Res. 289, 600–602 (1997).
Mueller, M. M. & Fusenig, N. E. Constitutive expression of G-CSF and GM-CSF in human skin carcinoma cells with functional consequence for tumor progression. Int. J. Cancer 83, 780–789 (1999).
Kawakami, T. et al. Elevated serum granulocyte colony-stimulating factor levels in patients with active phase of sweet syndrome and patients with active behcet disease: implication in neutrophil apoptosis dysfunction. Arch. Dermatol. 140, 570–574 (2004).
Liu, D., Seiter, K., Mathews, T., Madahar, C. J. & Ahmed, T. Sweet's syndrome with CML cell infiltration of the skin in a patient with chronic-phase CML while taking Imatinib Mesylate. Leuk. Res. 28, S61–S63 (2004).
Kokai, Y. et al. Overexpression of granulocyte colony-stimulating factor induces severe osteopenia in developing mice that is partially prevented by a diet containing vitamin K2 (menatetrenone). Bone 30, 880–885 (2002).
Lee, M. Y., Fukunaga, R., Lee, T. J., Lottsfeldt, J. L. & Nagata, S. Bone modulation in sustained hematopoietic stimulation in mice. Blood 77, 2135–2141 (1991).
Semerad, C. L. et al. G-CSF potently inhibits osteoblast activity and CXCL12 mRNA expression in the bone marrow. Blood 106, 3020–3027 (2005).
Clowes, J. A., Riggs, B. L. & Khosla, S. The role of the immune system in the pathophysiology of osteoporosis. Immunol. Rev. 208, 207–227 (2005).
Lee, N. K. et al. Endocrine regulation of energy metabolism by the skeleton. Cell 130, 456–469 (2007).
Borregaard, N., Theilgaard-Monch, K., Cowland, J. B., Stahle, M. & Sorensen, O. E. Neutrophils and keratinocytes in innate immunity--cooperative actions to provide antimicrobial defense at the right time and place. J. Leukoc. Biol. 77, 439–443 (2005).
Bonizzi, G. & Karin, M. The two NF-kappaB activation pathways and their role in innate and adaptive immunity. Trends Immunol. 25, 280–288 (2004).
Brocard, J. et al. Spatio-temporally controlled site-specific somatic mutagenesis in the mouse. Proc. Natl Acad. Sci. USA 94, 14559–14563 (1997).
Zenz, R. et al. c-Jun regulates eyelid closure and skin tumor development through EGFR signaling. Dev. Cell 4, 879–889 (2003).
Acknowledgements
We are very grateful to P. Angel, R. Eferl, D. Maurer, E. Passegué, A. J. Pospisilik, M. Sibilia and J. Guinea Viniegra for critical comments and suggestions to the manuscript; A. Bozec and V. Komnenovic for invaluable support for bone histology, U. Möhle-Steinlein for Southern blots and H. Tkadletz for help in preparing the illustrations. The IMP is funded by Boehringer Ingelheim and this work was supported by grants P14680-Gen and P18478 from the Austrian Research Foundation, by the Research Training Network (RTN) Program of the European Community and part of this work was supported by an FWF Network grant NFN S94-SP11. IMBA is founded by the Ministry of Science and the Austrian Academy of Sciences. JMP is also supported by grants from the Austrian Research Foundation (SFB).
Author information
Authors and Affiliations
Contributions
A.M., R.Z., H.B.S. and E.F.W. planned and designed the experiments; A.M., R.Z., H.B.S., L.K. and H.S. performed the experiments; A.M., R.Z., H.B.S., L.K., J.M.P. and E.F.W. analysed and interpreted the data; J.M.P. contributed reagents; A.M., R.Z., H.B.S. and E.F.W. wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplemenatary Figures S1, S2, S3, S4, S5, S6, S7 and Supplementary Information (PDF 1175 kb)
Rights and permissions
About this article
Cite this article
Meixner, A., Zenz, R., Schonthaler, H. et al. Epidermal JunB represses G-CSF transcription and affects haematopoiesis and bone formation. Nat Cell Biol 10, 1003–1011 (2008). https://doi.org/10.1038/ncb1761
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1761
This article is cited by
-
The AP-1 transcription factors c-Jun and JunB are essential for CD8α conventional dendritic cell identity
Cell Death & Differentiation (2021)
-
JunB defines functional and structural integrity of the epidermo-pilosebaceous unit in the skin
Nature Communications (2018)
-
RNA-Seq and ChIP-Seq Reveal SQSTM1/p62 as a Key Mediator of JunB Suppression of NF-κB-Dependent Inflammation
Journal of Investigative Dermatology (2015)
-
Pancreatic β-cells activate a JunB/ATF3-dependent survival pathway during inflammation
Oncogene (2012)
-
Epidermal phospholipase Cδ1 regulates granulocyte counts and systemic interleukin-17 levels in mice
Nature Communications (2012)