Abstract
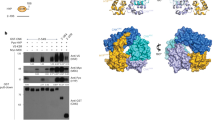
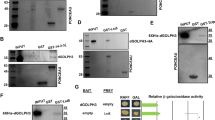
Class I phosphoinositide 3-kinases (PI(3)Ks) are activated through associated adaptor molecules in response to G protein-coupled and tyrosine kinase receptor signalling1. They contain Ras-binding domains (RBDs) and can also be activated through direct association with active GTP-bound Ras2,3,4,5,6,7,8,9,10. The ability of Ras to activate PI(3)K has been established in vitro and by overexpression analysis, but its relevance for normal PI(3)K function in vivo is unknown. The Drosophila class I PI(3)K, Dp110, is activated by nutrient-responsive insulin signalling and modulates growth, oogenesis and metabolism11,12,13,14,15. To investigate the importance of Ras-mediated PI(3)K activation for normal PI(3)K function, we replaced Dp110 with Dp110RBD, which is unable to bind to Ras but otherwise biochemically normal. We found that Ras-mediated Dp110 regulation is dispensable for viability. However, egg production, which requires large amounts of growth, is dramatically lowered in Dp110RBD flies. Furthermore, insulin cannot maximally activate PI(3)K signalling in Dp110RBD imaginal discs and Dp110RBD flies are small. Thus, Dp110 integrates inputs from its phosphotyrosine-binding adaptor and Ras to achieve maximal PI(3)K signalling in specific biological situations.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Vanhaesebroeck, B. et al. Synthesis and function of 3-phosphorylated inositol lipids. Annu. Rev. Biochem. 70, 535–602 (2001).
Marte, B. M., Rodriguez-Viciana, P., Wennstrom, S., Warne, P. H. & Downward, J. R-Ras can activate the phosphoinositide 3-kinase but not the MAP kinase arm of the Ras effector pathways. Curr. Biol. 7, 63–70 (1997).
Pacold, M. E. et al. Crystal structure and functional analysis of Ras binding to its effector phosphoinositide 3-kinase γ. Cell 103, 931–943 (2000).
Rodriguez-Viciana, P., Sabatier, C. & McCormick, F. Signaling specificity by Ras family GTPases is determined by the full spectrum of effectors they regulate. Mol. Cell Biol. 24, 4943–4954 (2004).
Rodriguez-Viciana, P. et al. Phosphatidylinositol-3-OH kinase as a direct target of Ras. Nature 370, 527–532 (1994).
Rodriguez-Viciana, P. et al. Role of phosphoinositide 3-OH kinase in cell transformation and control of the actin cytoskeleton by Ras. Cell 89, 457–467 (1997).
Rodriguez-Viciana, P., Warne, P. H., Vanhaesebroeck, B., Waterfield, M. D. & Downward, J. Activation of phosphoinositide 3-kinase by interaction with Ras and by point mutation. EMBO J. 15, 2442–2451 (1996).
Rubio, I., Rodriguez-Viciana, P., Downward, J. & Wetzker, R. Interaction of Ras with phosphoinositide 3-kinase γ. Biochem. J. 326, 891–895 (1997).
Suire, S., Hawkins, P. & Stephens, L. Activation of phosphoinositide 3-kinase γ by Ras. Curr. Biol. 12, 1068–1075 (2002).
Vanhaesebroeck, B. et al. P110δ, a novel phosphoinositide 3-kinase in leukocytes. Proc. Natl Acad. Sci. USA 94, 4330–4335 (1997).
Drummond-Barbosa, D. & Spradling, A. C. Stem cells and their progeny respond to nutritional changes during Drosophila oogenesis. Dev. Biol. 231, 265–278 (2001).
LaFever, L. & Drummond-Barbosa, D. Direct control of germline stem cell division and cyst growth by neural insulin in Drosophila. Science 309, 1071–1073 (2005).
Leevers, S. J. & Hafen, E. Growth regulation by insulin and TOR signaling in Drosophila. in Cell Growth: Control of Cell Size (eds. Hall, M. N., Raff, M. & Thomas, G.) 167–192 (Cold Spring Harbour Laboratory Press, Cold Spring Harbour, 2004).
Leevers, S. J., Weinkove, D., MacDougall, L. K., Hafen, E. & Waterfield, M. D. The Drosophila phosphoinositide 3-kinase Dp110 promotes cell growth. EMBO J. 15, 6584–6594 (1996).
Wu, Q. & Brown, M. R. Signaling and function of insulin-like peptides in insects. Annu. Rev. Entomol. 51, 1–24 (2006).
Lizcano, J. M. et al. Insulin-induced Drosophila S6 kinase activation requires phosphoinositide 3-kinase and protein kinase B. Biochem. J. 374, 297–306 (2003).
Sarbassov, D. D., Guertin, D. A., Ali, S. M. & Sabatini, D. M. Phosphorylation and regulation of Akt/PKB by the rictor-mTOR complex. Science 307, 1098–1101 (2005).
Weinkove, D., Leevers, S. J., MacDougall, L. K. & Waterfield, M. D. p60 is an adaptor for the Drosophila phosphoinositide 3-kinase, Dp110. J. Biol. Chem. 272, 14606–14610 (1997).
Weinkove, D., Neufeld, T. P., Twardzik, T., Waterfield, M. D. & Leevers, S. J. Regulation of imaginal disc cell size, cell number and organ size by Drosophila class I(A) phosphoinositide 3-kinase and its adaptor. Curr. Biol. 9, 1019–1029 (1999).
Colicelli, J. Human RAS superfamily proteins and related GTPases. Sci. STKE re13 (2004).
Karim, F. D. & Rubin, G. M. Ectopic expression of activated Ras1 induces hyperplastic growth and increased cell death in Drosophila imaginal tissues. Development 125, 1–9 (1998).
Prober, D. A. & Edgar, B. A. Ras1 promotes cellular growth in the Drosophila wing. Cell 100, 435–446 (2000).
Prober, D. A. & Edgar, B. A. Interactions between Ras1, dMyc, and dPI3K signaling in the developing Drosophila wing. Genes Dev. 16, 2286–2299 (2002).
Diaz-Benjumea, F. J. & Hafen, E. The sevenless signalling cassette mediates Drosophila EGF receptor function during epidermal development. Development 120, 569–578 (1994).
Strutt, H. & Strutt, D. EGF signaling and ommatidial rotation in the Drosophila eye. Curr. Biol. 13, 1451–1457 (2003).
Kypta, R. M., Su, H. & Reichardt, L. F. Association between a transmembrane protein tyrosine phosphatase and the cadherin-catenin complex. J. Cell Biol. 134, 1519–1529 (1996).
Acknowledgements
We thank R. Williams for advice on mutation of the RBD, M. White for pGEX–Ras1, H. Stocker and E. Hafen for hypomorphic Dp110 mutants, and L. Foukas, B. Vanhaesebroeck, B. Baum, N. Tapon and M. Giannakou for advice, reagents and helpful discussions. We thank the Cancer Research UK London Research Institute (CRUK LRI) Equipment Park and Fly Facility for technical support and M. Cully, J. Downward, C. Marshall, S. Marygold, N. Tapon and B. Vanhaesebroeck for advice on manuscript preparation. This work was supported by Cancer Research UK, The Ludwig Institute for Cancer Research and the BBSRC.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
Supplementary Figures S1, S2, S3 and S4 (PDF 514 kb)
Rights and permissions
About this article
Cite this article
Orme, M., Alrubaie, S., Bradley, G. et al. Input from Ras is required for maximal PI(3)K signalling in Drosophila. Nat Cell Biol 8, 1298–1302 (2006). https://doi.org/10.1038/ncb1493
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1493
This article is cited by
-
GPCR activation of Ras and PI3Kγ in neutrophils depends on PLCβ2/β3 and the RasGEF RasGRP4
The EMBO Journal (2012)
-
Synergism between altered cortical polarity and the PI3K/TOR pathway in the suppression of tumour growth
EMBO reports (2012)
-
Differential roles for the p101 and p84 regulatory subunits of PI3Kγ in tumor growth and metastasis
Oncogene (2012)
-
The emerging mechanisms of isoform-specific PI3K signalling
Nature Reviews Molecular Cell Biology (2010)
-
Oncogenic signaling of class I PI3K isoforms
Oncogene (2008)