Abstract
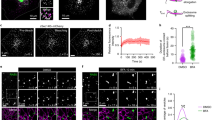
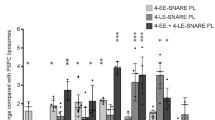
The tyrosine phosphatase PTP-MEG2 is targeted by its amino-terminal Sec14p homology domain to the membrane of secretory vesicles. There it regulates vesicle size by promoting homotypic vesicle fusion by a mechanism that requires its catalytic activity. Here, we identify N-ethylmaleimide-sensitive factor (NSF), a key regulator of vesicle fusion, as a substrate for PTP-MEG2. PTP-MEG2 reduced the phosphotyrosine content of NSF and co-localized with NSF and syntaxin 6 in intact cells. Furthermore, endogenous PTP-MEG2 co-immunoprecipitated with endogenous NSF. Phosphorylation of NSF at Tyr 83, as well as an acidic substitution at the same site, increased its ATPase activity and prevented αSNAP binding. Conversely, expression of a Y83F mutant of NSF caused spontaneous fusion events. Our results suggest that the molecular mechanism by which PTP-MEG2 promotes secretory vesicle fusion involves the local release of NSF from a tyrosine-phosphorylated, inactive state. This represents a novel mechanism for localized regulation of NSF and the first demonstrated role for a protein tyrosine phosphatase in the regulated secretory pathway.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hunter T. Protein kinases and phosphatases: the yin and yang of protein phosphorylation and signaling. Cell 80, 225–236 (1995).
Mustelin, T. Src Family Tyrosine Kinases in Leukocytes. 1–155 (R.G. Landes Co., Austin, 1994).
Tonks, N.K. & Neel, B.G. From form to function: Signaling by protein tyrosine phosphatases. Cell 87, 365–368 (1996).
Mustelin, T. et al. Protein tyrosine phosphatases in T cell physiology. Mol. Immunol. 41, 687–700 (2004).
Mustelin, T. et al. Protein tyrosine phosphatases. Front. Biosci. 7, 85–142 (2002).
Alonso, A. et al. Protein tyrosine phosphatases in the human genome. Cell 117, 1–13 (2004).
Manning, G., Whyte, D.B., Martinez, R., Hunter, T. & Sudarsanam, S. The protein kinase complement of the human genome. Science 298, 1912–1934 (2002).
Wang, X. et al. Enlargement of secretory vesicles by protein tyrosine phosphatase PTP-MEG2 in rat basophilic leukemia mast cells and Jurkat T cells. J. Immunol. 168, 4612–4619 (2002).
Kruger, J.M. et al. Protein-tyrosine phosphatase MEG2 is expressed by human neutrophils. Localization to the phagosome and activation by polyphosphoinositides. J. Biol. Chem. 277, 2620–2628 (2002).
Gu, M., Warshawsky, I. & Majerus, P.W. Cloning and expression of a cytosolic megakaryocyte protein-tyrosine-phosphatase with sequence homology to retinaldehyde-binding protein and yeast SEC14p. Proc. Natl Acad. Sci. USA 89, 2980–2984 (1992).
Huynh, H. et al. Homotypic secretory vesicle fusion induced by the protein tyrosine phosphatase MEG2 depends on polyphosphoinositides in T cells. J. Immunol. 171, 6661–6671 (2003).
Krugmann, S. et al. Identification of ARAP3, a novel PI3K effector regulating both Arf and Rho GTPases, by selective capture on phosphoinositide affinity matrices. Mol. Cell 9, 95–108 (2002).
Flint, A.J., Tiganis, T., Barford, D. & Tonks, N.K. Development of “substrate-trapping” mutants to identify physiological substrates of protein tyrosine phosphatases. Proc. Natl Acad. Sci. USA 94, 1680–1685 (1997).
Jahn, R. & Südhof, T.C. Membrane fusion and exocytosis. Annu. Rev. Biochem. 68, 863–911 (1999).
Hay, J.C. & Scheller, R.H. SNAREs and NSF in targeted membrane fusion. Curr. Opin. Cell Biol. 9, 505–512 (1997).
Wendler, F., Pagem L., Urbé, S. & Tooze, S.A. Homotypic fusion of immature secretory granules during maturation requires syntaxin 6. Mol. Biol. Cell 12, 1699–1709 (2001).
May, A.P., Misura, K.M.S., Whiteheart, S.W. & Weis, W.I. Crystal structure of the amino-terminal domain of N-ethylmaleimide-sensitive fusion protein. Nature Cell Biol. 1, 174–182 (1999).
Yu, R.C., Jahn, R. & Brunger, A.T. NSF N-terminal domain crystal structure: models of NSF function. Mol. Cell 4, 97–107 (1999).
Paumet, F. et al. Soluble NSF attachment protein receptors (SNAREs) in RBL-2H3 mast cells: functional role of syntaxin 4 in exocytosis and identification of a vesicle-associated membrane protein 8-containing secretory compartment. J. Immunol. 164, 5850–5857 (2000).
Lavoie, C. et al. Tyrosine phosphorylation of p97 regulates transitional endoplasmic reticulum assembly in vitro. Proc. Natl Acad. Sci. USA 97, 13637–13642 (2000).
Volinia, S. et al. A human phosphatidylinositol 3-kinase complex related to the yeast Vps34p–Vps15p protein sorting system, EMBO J. 14, 3339–3348 (1995).
Schu, P.V. et al. Phosphatidylinositol 3-kinase encoded by yeast VPS34 gene essential for protein sorting. Science 260, 88–91 (1993).
Corvera, S., D'Arrigo, A. & Stenmark, H. Phosphoinositides in membrane traffic. Curr. Opin. Cell Biol. 11, 460–465 (1999).
Austin, C.D. & Shields, D. Formation of nascent secretory vesicles from the trans-Golgi network of endocrine cells is inhibited by tyrosine kinase and phosphatase inhibitors. J. Cell Biol. 135, 1471–1483 (1996).
von Willebrand, M. et al. Inhibition of phosphatidylinositol 3-kinase blocks TCR/CD3-induced activation of the mitogen-activated kinase Erk2. Eur. J. Biochem. 235, 828–835 (1996).
Saxena, M., Williams, S., Brockdorff, J., Gilman, J. & Mustelin, T. Inhibition of T cell signaling by MAP kinase-targeted hematopoietic tyrosine phosphatase (HePTP). J. Biol. Chem. 274, 11693–11700 (1999).
Saxena, M., Williams, S., Taskén, K. & Mustelin, T. Crosstalk between cAMP-dependent kinase and MAP kinase through hematopoietic protein tyrosine phosphatase (HePTP). Nature Cell Biol. 1, 305–310 (1999).
Bottini, N. et al. A functional variant of lymphoid tyrosine phosphatase is associated with type I diabetes. Nature Genet. 36, 337–338 (2004).
Alonso, A. et al. Tyrosine phosphorylation of VHR phosphatase by ZAP-70. Nature Immunol. 4, 44–48 (2003).
Gjörloff-Wingren, A. et al. Subcellular localization of intracellular protein tyrosine phosphatases in T cells. Eur. J. Immunol. 30, 2412–2421 (2000).
Reynolds, E.S. The use of lead citrate at high pH as an electronopaque stain in electron microscopy. J. Cell Biol. 17, 208–216 (1963).
Acknowledgements
We are grateful to G. Reed and S. Whiteheart for anti-NSF antibodies and cDNA, to S. Tooze for anti-syntaxin-6 antibodies and to T. Tsuji for advice on ATPase assays. This work was supported by fellowships from the American Italian Cancer Foundation, the Litta Foundation and by grants AI55741, AI35603, AI48032, AI53585, CA96949 (to T.M.) and AG00252 (to H.H.) from the National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information, Figures
Fig. S1 and Fig. S2 (PDF 1154 kb)
Rights and permissions
About this article
Cite this article
Huynh, H., Bottini, N., Williams, S. et al. Control of vesicle fusion by a tyrosine phosphatase. Nat Cell Biol 6, 831–839 (2004). https://doi.org/10.1038/ncb1164
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ncb1164
This article is cited by
-
Effect of L3MBTL3/PTPN9 polymorphisms on risk to alcohol-induced ONFH in Chinese Han population
Neurological Sciences (2022)
-
Synthesis of lipid membranes for artificial cells
Nature Reviews Chemistry (2021)
-
Heterozygous Meg2 Ablation Causes Intraocular Pressure Elevation and Progressive Glaucomatous Neurodegeneration
Molecular Neurobiology (2019)
-
MEG2 is regulated by miR-181a-5p and functions as a tumour suppressor gene to suppress the proliferation and migration of gastric cancer cells
Molecular Cancer (2017)
-
LRRK2 phosphorylates pre-synaptic N-ethylmaleimide sensitive fusion (NSF) protein enhancing its ATPase activity and SNARE complex disassembling rate
Molecular Neurodegeneration (2016)