Abstract
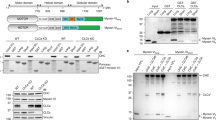
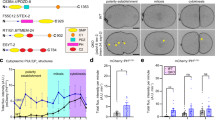
The budding of clathrin-coated vesicles is essential for protein transport. After budding, clathrin must be uncoated before the vesicles can fuse with other membranous structures. In vitro, the molecular chaperone Hsc70 uncoats clathrin-coated vesicles in an ATP-dependent process that requires a specific J-domain protein such as auxilin. However, there is little evidence that either Hsc70 or auxilin is essential in vivo. Here we show that C. elegans has a single auxilin homologue that is identical to mammalian auxilin in its in vitro activity. When RNA-mediated interference (RNAi) is used to inhibit auxilin expression in C. elegans, oocytes show markedly reduced receptor-mediated endocytosis of yolk protein tagged with green fluorescent protein (GFP). In addition, most of these worms arrest during larval development, exhibit defective distribution of GFP–clathrin in many cell types, and show a marked change in clathrin dynamics, as determined by fluorescence recovery after photobleaching (FRAP). We conclude that auxilin is required for in vivo clathrin-mediated endocytosis and development in C. elegans.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Kelley, W. L. Trends Biochem. Sci. 23, 222–227 (1998).
Prasad, K., Barouch, W., Greene, L. & Eisenberg, E. J. Biol. Chem. 268, 23758–23761, (1993).
Ungewickell, E. et al. Nature 378, 632–635 (1995).
Greener, T., Zhao, X., Nojima, H., Eisenberg, E. & Greene, L. E. J. Biol. Chem. 275, 1365–1370 (2000).
Umeda A., Meyerholz A., & Ungewickell, E. Eur. J. Cell Biol. 79, 336–342 (2000).
Schroder, S. et al. Eur. J. Biochem. 228, 297–304 (1995).
Kanaoka. Y., Kimura, S. H., Okazaki, I., Ikeda M. & Nojima, H. FEBS Lett. 402, 73–80 (1997).
Grant, B. & Hirsh, D. Mol. Biol. Cell 10, 4311–4326 (1999).
Fire, A, Xu, S., Montgomery, M. K., Kostas, S. A., Driver, S. E. & Mello, C. C. Nature 391, 806–811 (1998).
Montgomery, M. K., Xu, S. & Fire, A. Proc. Natl Acad. Sci. USA 95, 15502–15507 (1998).
Yochem, J., Tuck S., Greenwald, I. & Han, M. Development 126, 597–606 (1999).
Thacker, C., Peters, K. Srayko, M. & Rose, A. M. Genes Dev. 9, 956–971 (2000).
Tavernarakis N., Wang S. L., Dorovkov, M., Ryazanov, A. & Driscoll, M. Nature Genet. 2, 180–183 (2000).
Kelly, W. G., Xu S., Montgomery, M. K. & Fire, A. Genetics 146, 227–238 (1997).
Che S., Weil, M. M., Etkin, L. D., Epstein, H. F. & Kuang, J. Biochim. Biophys. Acta 1354, 231–240 (1997).
Gaidorov, I., Santini, F., Warren, R. A. & Keen, J. H. Nature Cell Biol. 1, 1–7 (1999).
Jiang, R. Gao, B., Prasad, K., Greene, L. E. & Eisenberg, E. J. Biol. Chem. 275, 8439–8447 (2000).
Brodin L., Low, P. & Shupliakov, O. Curr. Opin. Neurobiol. 10, 312–320 (2000).
Greene, L. E. & Eisenberg, E. J. Biol. Chem. 265, 6682–6687 (1990).
Prasad, K., Heuser, J., Eisenberg, E. & Greene, L. J. Biol. Chem. 269, 6931–6939 (1994).
Ye, W. & Lafer, E. M. J. Biol. Chem. 270, 10933–10939 (1995).
Barouch, W., Prasad, K., Greene, L. E. & Eisenberg, E. J. Biol. Chem. 269, 28563–28568 (1994).
Holstein, S. E., Ungewickell, H. & Ungewickell, E. J. Cell Biol. 135,925–937 (1996).
Brenner, S. Genetics 77, 71–94 (1974).
Fire, A. EMBO J. 5, 2673–2680 (1986).
Mello, C. C., Kramer J. M., Stinchcomb, D. & Ambros, V. EMBO J. 10, 3959–3970 (1991).
Acknowledgements
We thank M. Krause for valuable assistance, Y. Xu for electron micrographs of the baskets, A. Fire for GFP plasmids, Y. Kohara for cDNA clones of C. elegans auxilin, and W. Przylecki for technical support. This work was supported in part by March of Dimes grant FY99-583 (to D.H.).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Greener, T., Grant, B., Zhang, Y. et al. Caenorhabditis elegans auxilin: a J-domain protein essential for clathrin-mediated endocytosis in vivo. Nat Cell Biol 3, 215–219 (2001). https://doi.org/10.1038/35055137
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/35055137
This article is cited by
-
Molecular mechanism and physiological functions of clathrin-mediated endocytosis
Nature Reviews Molecular Cell Biology (2011)
-
Disruption of zebrafish cyclin G-associated kinase (GAK) function impairs the expression of Notch-dependent genes during neurogenesis and causes defects in neuronal development
BMC Developmental Biology (2010)
-
Regulation of endosomal clathrin and retromer-mediated endosome to Golgi retrograde transport by the J-domain protein RME-8
The EMBO Journal (2009)
-
The clathrin-binding motif and the J-domain of Drosophila Auxilin are essential for facilitating Notch ligand endocytosis
BMC Developmental Biology (2008)
-
Caenorhabditis elegans RME-6 is a novel regulator of RAB-5 at the clathrin-coated pit
Nature Cell Biology (2005)