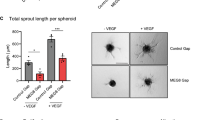
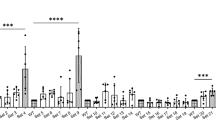
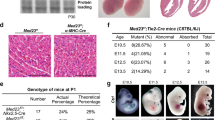
Abstract
We describe a technique to define gene function using antisense oligonucleotide (AS-ODN) inhibition of gene expression in mice. A single intravenous injection of an AS-ODN targeting vascular endothelial growth factor (VEGF) into pregnant mice between E7.5–8.5 resulted in a lack of primary angiogenesis. This enabled us to define the critical window required to inhibit VEGF expression and recapitulate the primary loss of function phenotype observed in VEGF (–/–) embryos. This phenotype was sequence-specific and time- and dose-dependent. Injection of an AS-ODN targeting a second gene, E-cadherin, into pregnant mice at E10 confirmed a hypothesized secondary phenotype. This is the first report of AS-ODN inhibition of gene expression in utero and provides a new strategy for target validation in functional genomics.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Christoffersen, R.E. Translating genomics information into therapeutics: a key role for oligonucleotides. Nat. Biotechnol. 15, 483– 484 (1997).
Juliano, R.L., Alahari, S., Yoo, H., Kole, R. & Cho, M. Antisense pharmacodynamics: critical issues in the transport and delivery of antisense oligonucleotides. Pharm. Res. 16, 494–502 (1999).
Nyce, J.W. Insight into adenosine receptor function using antisense and gene-knockout approaches. Trends. Pharmacol. Sci. 20, 79–83 (1999).
Jones, D.A. & Fitzpatrick, F.A. Genomics and the discovery of new drug targets. Curr. Opin. Chem. Biol. 3, 71–76 (1999).
Bramlage, B., Luzi, E. & Eckstein, F. Designing ribozymes for the inhibition of gene expression. Trends. Biotechnol. 16, 434– 438 (1998).
Dickinson, L.A. et al. Inhibition of RNA polymerase II transcription in human cells by synthetic DNA-binding ligands. Proc. Natl. Acad. Sci. USA. 95, 12890–12895 (1998).
Mei, H.Y. et al. Discovery of selective, small-molecule inhibitors of RNA complexes—I. The Tat protein/TAR RNA complexes required for HIV-1 transcription. Bioorg. Med. Chem. 5, 1173–1184 (1997).
Tatton, W.G., Chalmers-Redman, R.M., Ju, W.Y., Wadia, J. & Tatton, N.A. Apoptosis in neurodegenerative disorders: potential for therapy by modifying gene transcription. J. Neural. Transm. Suppl. 49, 245– 268 (1997).
Wodarz, A. & Nusse, R. Mechanisms of Wnt signaling in development. Annu. Rev. Cell Biol. 14, 59– 88 (1998).
Lai, L.W. & Lien, Y.H. Homologous recombination based gene therapy. Exp. Nephrol. 7, 11– 14 (1999).
Osterrieder, N. & Wolf, E. Lessons from gene knockouts. Rev. Sci. Tech. 17, 351– 364 (1998).
Sauer, B. Inducible gene targeting in mice using the Cre/lox system. Methods 14, 381–392 ( 1998).
Hynes, R.O. Targeted mutations in cell adhesion genes: what have we learned from them? Dev. Biol. 180, 402–412 (1996).
Hynes, R.O. & Bader, B.L. Targeted mutations in integrins and their ligands: their implications for vascular biology. Thromb. Haemostasis. 78, 83–87 ( 1997).
Carmeliet, P. et al. Abnormal blood vessel development and lethality in embryos lacking a single VEGF allele. Nature 380, 435– 439 (1996).
Ferrara, N. et al. Heterozygous embryonic lethality induced by targeted inactivation of the VEGF gene. Nature 380, 439– 442 (1996).
Robinson, G.S. et al. Oligodeoxynucleotides inhibit retinal neovascularization in a murine model of proliferative retinopathy. Proc. Natl. Acad. Sci. USA. 93, 4851–4856 ( 1996).
Smyth, A.P., Rook, S.L., Detmar, M. & Robinson, G.S. Antisense oligonucleotides inhibit vascular endothelial growth factor/vascular permeability factor expression in normal human epidermal keratinocytes. J. Invest. Dermatol. 108, 523–526 (1997).
Chen, B. & Hales, B.F. Antisense oligonucleotide down-regulation of E-cadherin in the yolk sac and cranial neural tube malformations. Biol. Reprod. 53, 1229–1238 (1995).
Gaudette, M.F., Hampikian, G., Metelev, V., Agrawal, S. & Crain, W.R. Effect on embryos of injection of phosphorothioate-modified oligonucleotides into pregnant mice. Antisense. Res. Dev. 3, 391–397 (1993).
Bourque, A.J. & Cohen, A.S. Quantitative analysis of phosphorothioate oligonucleotides in biological fluids using direct injection fast anion-exchange chromatography and capillary gel electrophoresis. J. Chromatogr. Biomed. Appl. 662, 343–349 (1994).
Zhao, Q. et al. Cellular distribution of phosphorothioate oligonucleotide following intravenous administration in mice. Antisense. Nucleic. Acid. Drug Dev. 8, 451–458 (1998).
Pacifici, G.M. & Nottoli, R. Placental transfer of drugs administered to the mother. Clin. Pharmacokinet. 28, 235–269 (1995).
Yu, D. et al. Hybrid oligonucleotides: synthesis, biophysical properties, stability studies, and biological activity. Bioorg. Med. Chem. 4, 1685–1692 (1996).
Guvakova, M.A., Yakubov, L.A., Vlodavsky, I., Tonkinson, J.L. & Stein, C. A. Phosphorothioate oligodeoxynucleotides bind to basic fibroblast growth factor, inhibit its binding to cell surface receptors, and remove it from low affinity binding sites on extracellular matrix. J. Biol. Chem. 270, 2620– 2627 (1995).
Rockwell, P. et al. Cell-surface perturbations of the epidermal growth factor and vascular endothelial growth factor receptors by phosphorothioate oligodeoxynucleotides. Proc. Natl. Acad. Sci. USA 94, 6523– 6528 (1997).
Takeichi, M. et al. Cadherins in brain patterning and neural network formation. Cold Spring Harb. Symp. Quant. Biol. 62, 505– 510 (1997).
Anderson, H. Adhesion molecules and animal development. Experientia 46, 2–13 (1990).
Senger, D.R. et al. Vascular permeability factor (VPF, VEGF) in tumor biology. Cancer Metastasis Rev. 12, 303–324 (1993).
Ferrara, N. Vascular endothelial growth factor: molecular and biological aspects. Curr. Top. Microbiol. Immunol. 237, 1– 30 (1999).
Aiello, L.P. et al. Vascular endothelial growth factor in ocular fluid of patients with diabetic retinopathy and other retinal disorders. N. Engl. J. Med. 331, 1480–1487 ( 1994).
Baumgartner, I. & Isner, J.M. Stimulation of peripheral angiogenesis by vascular endothelial growth factor (VEGF). Vasa. 27, 201–206 ( 1998).
Grunstein, J., Roberts, W.G., Mathieu-Costello, O., Hanahan, D. & Johnson, R.S. Tumor-derived expression of vascular endothelial growth factor is a critical factor in tumor expansion and vascular function. Cancer Res. 59, 1592– 1598 (1999).
Kitamoto, Y., Tokunaga, H. & Tomita, K. Vascular endothelial growth factor is an essential molecule for mouse kidney development: glomerulogenesis and nephrogenesis. J. Clin. Invest. 99, 2351–2357 (1997).
Chirgwin, J.M., Przybyla, A.E., MacDonald, R.J. & Rutter, W.J. Isolation of biologically active ribonucleic acid from sources enriched in ribonuclease. Biochemistry 18, 5294– 5299 (1979).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Driver, S., Robinson, G., Flanagan, J. et al. Oligonucleotide-based inhibition of embryonic gene expression. Nat Biotechnol 17, 1184–1187 (1999). https://doi.org/10.1038/70724
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/70724
This article is cited by
-
Reduction of liver Fas expression by an antisense oligonucleotide protects mice from fulminant hepatitis
Nature Biotechnology (2000)
-
Shortcuts from gene sequence to function
Nature Biotechnology (1999)