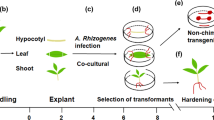
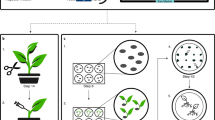
Abstract
The ability to regenerate plants from poplar cells cultured in vitro suggests that poplars may prove a valuable model system for the application of recombinant DNA technology to deciduous trees. We report here the transformation of a hybrid Populus trichocarpa × deltoides with two strains of Agrobacterium tumefaciens, as demonstrated by phytohormone autonomous growth, opine production, and Southern blot analysis.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
References
Nester, E.W., Gordon, M.P., Amasino, R.M., and Yanofsky, M.F. 1984. Crown gall: A molecular and physiological analysis. Ann. Rev. Plant Physiol. 35:387–413.
Hooykaas, P.J.J., and Schilperoort, R.A. 1984. The molecular genetics of crown gall tumorigenesis. Adv. Genet. 22:210–283.
Depicker, A., Van Montagu, M., and Schell, J. 1983. Plant cell transformation by Agrobacterium plasmids. p. 499–xxx. In: Genetic Engineering of Plants: An Agricultural Perspective. Kosuge, T., Meredith, C. P., and Hollaender, A. (eds). Plenum Press, New York.
Powell, A.L.T., and Gordon, M.P. in preparation. In: The Biochemistry of Plants. A Comprehensive Treatise. A. Marcus (ed.). 2nd Edn. Academic Press, N.Y.
Zambryski, P., Depicker, A., Kruger, D., and Goodman, H.M. 1982. Tumor induction by Agrobacterium tumefaciens: analysis of the boundaries of T-DNA. Mol. Gen. Genet. 1:361–370.
Yadav, N.S., Vanderleyden, J., Bennett, D.R., Barnes, W.M., and Chilton, M.-D. 1982. Short direct repeats flank the T-DNA on a nopaline Ti-plasmid. Proc. Natl. Acad. Sci. USA. 79:6322–6326.
Simpson, R.B., O'Hara, P.J., Kwok, J., Montoya, A.L., Lichenstein, C., Gordon, M.P., and Nester, E.W. 1982. DNA from the A6S/2 crown gall tumor contains scrambled Ti-plasmid sequences near its junction with plant DNA. Cell 29:1005–1014.
Akiyoshi, D.E., Klee, H., Amasino, R.M., Nester, E.W., and Gordon, M.P. 1984. T-DNA of Agrobacterium tumefaciens encodes an enzyme of cytokinin biosynthesis. Proc. Natl. Acad. Sci. USA. 81:5994–5998.
Barry, G.F., Rogers, S.G., Fraley, R.T., and Brand, L. 1984. Identification of a cloned cytokinin biosynthetic gene. Proc. Natl. Acad. Sci. USA. 81:4776–4780.
Inze, D., Follin, A., Van Ligsebettens, M., Simoens, C., Genetello, D., Van Montagu, M., and Schell, J. 1984. Genetic analysis of the individual T-DNA genes of Agrobacterium tumefaciens; further evidence that two genes are involved in indole-3-acetic acid synthesis. Mol. Gen. Genet. 194:265–274.
Joos, H., Inze, D., Caplan, A., Sormann, M., Van Montagu, M., and Schell, J. 1983. Genetic analysis of T-DNA transcripts in nopaline crown galls. Cell 32:1057–1067.
Schroder, G., Waffenschmidt, S., Weiler, E.W., and Schroder, J. 1983. The T-region of Ti-plasmids codes for an enzyme synthesizing indole-3-acetic acid. EMBO 2:403–409.
Thomashow, L., Reeves, S., and Thomashow, M.F. 1984. Crown gall oncogenesis: evidence that a T-DNA gene from the Agrobacterium Ti-plasmid pTiA6 encodes an enzyme that catalyzes synthesis of indolacetic acid. Proc. Natl. Acad. Sci. USA. 81:5071–5075.
Montoya, A.L., Chilton, M.-D., Gordon, M.P., Sciaky, D., and Nester, E.W. 1977. Octopine and nopaline metabolism in Agrobacterium tumefaciens and crown gall tumor cells: role of plasmid genes. J. Bacteriol. 129:101–107.
An, G., Watson, B.D., Stachel, S., Gordon, M.P., and Nester, E.W. 1985. New cloning vehicles for transformation of higher plants. EMBO 4:277–284.
Fraley, R.T., Rogers, S.G., Horsch, R.B., Eichholtz, D.A., Flick, J.S., Fink, C.L., Hoffman, N.L., and Sanders, P.R. 1985. The SEV system; a new disarmed Ti plasmid vector system for plant transformation. Bio/Technology 3:629–635.
Klee, H.J., Yanofsky, M.F., and Nester, E.W. 1985. Vectors for transformation of higher plants. Bio/Technology 3:637–642.
Klee, H.J., Gordon, M.P., and Nester, E.W. 1982. Complementation analysis of Agrobacterium tumefaciens Ti-plasmid mutations affecting oncogenicity. J. Bacteriol. 150:327–331.
Klee, H.S., White, F.F., Iyer, V.N., Gordon, M.P., and Nester, E.W. 1983. Mutational analysis of the virulence region of an Agrobacterium tumefaciens Ti-plasmid. J. Bacteriol. 153:878–883.
Hoekema, A., Hirsch, P.R., Hooykaas, R.J.J., and Schilperoort, R.A. 1983. A binary plant vector strategy based on separation of vir and T-region of the Agrobacterium tumefaciens Ti-plasmid. Nature 303:179–180.
DeFramond, A., Barton, K.A., and Chilton, M.-D. 1983. Mini-Ti; A new vector strategy for plant genetic engineering. Bio/Technology 1:262–272.
DeCleene, M., and DeLey, J. 1976. The host range of crown gall. Bot. Rev. 42:389–466.
Chalupa, V. 1974. Control of root and shoot formation and production of trees from poplar callus. Biologia Plantarum (Praha) 16:316–320.
Wolter, K.E. 1968. Root and shoot initiation in aspen callus cultures. Nature 219:509–510.
Douglas, D. 1982. Protoplast isolation from totipotent cell-cultures of Populus hybrid TT32. Proc. 5th Intl. Cong. Plant Tissue and Cell Culture, p. 605–606.
Dhillon, S.S., Miksche, J.P., and Cecich, R.A. 1984. DNA changes in senescing leaves of Populus deltoides. Plant Physiol. (Suppl.)75:120.
International Poplar Commission. 1979. Poplars and willows in wood production and land use. FAO Forestry Series, No. 10. Rome.
Fege, A.S. 1983. The practice and physiological basis of collection, storing and planting Populus hardwood cuttings. Proc. of Workshop of Intensive Plantation Culture. USDA Forest Service General Technical Report NC-91, p. 1–11.
Heilman, P.E., and Stettler, R.F. 1985. Genetic variation and productivity of Populus trichocarpa T. & G. and its hybrids. II. Biomass production in a 4-year plantation. Canadian Journal of Forest Research 15:384–388.
Stettler, R.F., and Heilman, P.E. 1984. Short-rotation poplar culture in the Pacific Northwest: components of a genetic program. In: Ecology and Management of Forest Biomass Production Systems. Perttu, K. (ed.). Dept. Ecol. & Environ. Res., Swed. Univ. Agric. Sci. Rep. 15:217–232.
Sciaky, D., Montoya, A.L., and Chilton, M.-D. 1978. Fingerprints of Agrobacterium Ti-plasmids. Plasmid. 1:238–253.
Hood, E.E., Jen, G., Kayes, L., Kramer, J., Fraley, R.T., and Chilton, M.-D. 1984. Restriction endonuclease map of pTiBo542, a potential Ti plasmid vector for genetic engineering of plants. Bio/Technology 2:702–709.
Horsch, R.B., Fry, J.E., Hoffman, N.L., Eichholtz, D., Rogers, S.G., Fraley, R.T. 1985. A simple and general method for transferring genes into plants. Science 227:1229–1231.
Kwok, W. 1983. Ph.D. thesis, University of Washington.
Thomashow, M.F., Nutter, R., Montoya, A.L., Gordon, M.P., Nester, E.W. 1980. Integration and organization of Ti-plasmid sequences in crown gall tumors. Cell 19:729–739.
Murashige, T., and Skoog, F. 1962. A revised medium for rapid growth and bioassays with tobacco tissue cultures. Physiol. Plant. 15:473–497.
Murray, M.G., and Thompson, W.F. 1980. Rapid isolation of high molecular weight plant DNA. Nucl. Acids Res. 8:4321–4325.
Maniatis, T., Fritsch, E.F., Sambrook, J. 1982. Molecular Cloning. A laboratory manual. Cold Spring Harbor Laboratory, NY.
Southern, E.M. 1975. Detection of specific sequences among DNA fragments separated by gel electrophoresis. J. Mol. Biol. 98:503–517.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Parsons, T., Sinkar, V., Stettler, R. et al. Transformation of Poplar by Agrobacterium tumefaciens. Nat Biotechnol 4, 533–536 (1986). https://doi.org/10.1038/nbt0686-533
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0686-533
This article is cited by
-
Construction of genetic transformation system of Salix mongolica: in vitro leaf-based callus induction, adventitious buds differentiation, and plant regeneration
Plant Cell, Tissue and Organ Culture (PCTOC) (2018)
-
The cloning and characterization of a poplar stomatal density gene
Genes & Genomics (2014)