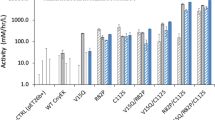
Abstract
We have investigated the effect of cysteine to serine substitutions in human basic fibroblast growth factor (bFGF) on the formation of inclusion bodies in Escherichia coli. Using a temperature–sensitive expression system, about 30% of human bFGF, which contains four cysteines at positions 26, 70, 88, and 93, is deposited into inclusion bodies. A single mutation at position 88 and a double mutation at positions 70 and 88 do not greatly alter the partition of bFGF into soluble and insoluble cell fractions. However, a single substitution of cysteine 70 by serine decreases the fraction of soluble bFGF significantly. When cysteines 26 and 93 (conserved among related growth factors) are replaced by serines, no soluble bFGF is formed in E. coli. Cysteine to serine substitutions also affect proteolytic susceptibility of bFGF during in vitro refolding from crude inclusion bodies. About 60% of human bFGF is lost to proteolytic degradation during in vitro refolding. Replacement of cysteines by serines increases the total recovery of bFGF, although more aggregates are formed during refolding. Ser–88–bFGF was expressed at the highest level, gave the highest soluble fraction in vivo, and exhibited the greatest fractional recovery and was recovered with the largest insoluble fraction after in vitro refolding. Thermal stability experiments at 42°C and 70°C revealed that cysteine to serine substitutions did not cause aggregation of the folded protein in vitro.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others

References
Williams, D.C., Van Frank, R.M., Muth, W.L. and Burnett, J.P. 1982. Cytoplasmic inclusion bodies in Escherichia coli producing bio synthetic chuman insulin proteins. Science 215: 687–689.
Cheng, Y.-S.E. 1983. Increased cell buoyant densities of protein overproducing Escherichia coli cells. Biochem. Biophys. Res. Commun. 111: 104–111.
Georgiou, G., Telford, J.N., Shuler, M.L. and Wilson, D.B. 1986. Localization of inclusion bodies in Escherichia coli overproducing β-lactamase or alkaline phosphatase. Appl. Environ. Microbiol. 52: 1157–1161.
Hart, R.A., Rinas, U. and Bailey, J.E. 1990. Protein composition of Vitreoscilla hemoglobin inclusion bodies produced in Escherichia coli. J. Biol .Chem. 265: 12728–12733.
Schein, C.H. 1989. Production of soluble recombinant proteins in bacteria. Bio/Technology 7: 1141–1149.
Mitraki, A. and King, J. 1989. Protein folding intermediates and inclusion body formation. Bio/Technology 7: 690–697.
Kiefhaber, T., Rudolph, R., Kohler, H.H. and Buchner, J. 1991. Protein aggregation in vivo and in vitro: A quantitative model of the kinetic competition between folding and aggregation. BiolTechnology 9: 825–829.
Kopetzki, E., Schumacher, G. and Buckel, P. 1989. Control of formation of active soluble or inactive insoluble baker's yeast α-glucosidase P1 in Escherichia coli by induction and growth conditions. Mol. Gen. Genet. 216: 149–155.
Chalmers, J.J., Kim, E., Telford, J.N., Wong, E.Y., Tacon, W.C., Shuler, M.L. and Wilson, D.B. 1990. Effects of temperature on Escherichia coli overproducing β-lactamase or human epidermal growth factor. Appl. Environ. Microbiol. 56: 104–111.
Schein, C.H. and Noteborn, M.H.M. 1988. Formation of soluble recombinant proteins in Escherichia coli is favored by lower growth temperature. Bio/Technology 6: 291–294.
Piatak, M., Lane, J.A., Laird, W., Bjorn, M.J., Wang, A. and Williams, M. 1988. Expression of soluble and fully functional ricin A chain in Escherichia coli is temperature-sensitive. J. Biol. Chem. 263: 4837–4843.
Mizukami, T., Komatsu, Y., Hosoi, N., Itoh, S. and Oka, T. 1986. Production of active human interferon-β in E coli. I. Preferential production by lower culture temperature. Biotechnol. Lett. 8: 605–610.
Gross, M., Sweet, R.W., Sathe, G., Yokoyama, S., Fasano, O., Goldfarb, M., Wigler, M. and Rosenberg, M. 1985. Purification and charaterization of human H-ras proteins expressed in Escherichia coli. Mol. Cell. Biol. 5: 1015–1024
Goldenberg, D.P., Smith, D.H. and King, J. 1983. Genetic analysis of the folding pathway for the tailspike protein of phage P22. Proc. Natl. Acad. Sci. USA 80: 7060–7064
Haase-Pettingell, C.A. and King, J. 1988. Formation of aggregates from a thermolabile in vivo folding intermediate in P22 tailspike maturation. A model for inclusion body formation. J. Biol. Chem. 263: 4977–4983
King, J., Fane, B., Haase-Pettingell, C., Mitraki, A., Villafane, R. and Yu, M.-H. 1990. Identification of amino acid sequences influencing intracellular folding pathways using temperature-sensitive folding mutations, In: .Protein Folding: Deciphering the Second Half of the Genetic Code. Gierasch, L. M. and King, J. (Eds.). American Association for the Advancement of Science, Washington, D.C. 225–240.
Mitraki, A., Fane, B., Haase-Pettingell, C., Sturtevant, J. and King, J. 1991. Global suppression of protein folding defects and inclusion body formation. Science 253: 54–58.
Krueger, J.K., Stock, A.M., Schutt, C.E. and Stock, J.B. 1990. Inclusion bodies from proteins produced at high levels in Escherichia coli, p. 136–141. In: Protein Folding: Deciphering the Second Half of the Genetic Code. Gierasch, L. M. and King, J. Eds. American Association for the Advancement of Science, Washington, D.C.
Wetzel, R., Perry, L.J. and Veilleux, C. 1991. Mutations in human interferon gamma affecting inclusion body formation identified by a general immunochemical screen. Bio/Technology 9: 731–737.
Strandberg, L. and Enfors, S.-O. 1991. Factors influencing inclusion body formation in the production of a fused protein in Escherichia coli. Appl. Environ. Microbiol. 57: 1669–1674.
Iwane, M., Kurokawa, T., Sasada, R., Seno, M., Nakagawa, S. and Igarashi, K. 1987. Expression of cDNA encoding human basic fibroblast growth factor in E. coli. Biochem. Biophys. Res. Commun. 146: 470–477.
Seno, M., Sasada, R., Iwane, M., Sudo, K., Kurokawa, T., Ito, K. and Igarashi, K. 1988. Stabilizing basic fibroblast growth factor using protein engineering. Biochem. Biophys. Res. Commun. 151: 701–708.
Squires, C.H., Childs, J., Eisenberg, S.P., Polverini, P.J. and Sommer, A. 1988. Production and characterization of human basic fibroblast growth factor from Escherichia coli. J. Biol. Chem. 263: 16297–16302.
Fox, G.M., Schiffer, S.G., Rohde, M.F., Tsai, L.B., Banks, A.R. and Arakawa, T., 1988. Production, biological activity, and structure of recombinant basic fibroblast growth factor and an analog with cysteine replaced by serine. J. Biol. Chem. 263: 18452–18458.
Abraham, J.A., Whang, J.L., Tumolo, A., Mergia, A., Friedman, J., Gospodarowicz, D. and Fiddes, J.C. 1986. Human basic fibroblast growth factor: nucleotide sequence and genomic organization. EMBO J. 5: 2523–2528.
Kurokawa, T., Sasada, R., Iwane, M. and Igarashi, K. 1987. Cloning and expression of cDNA encoding human basic fibroblast growth factor. FEBS Lett. 213: 189–194.
Zhu, X., Komiya, H., Chirino, A., Faham, S., Fox, G.M., Arakawa, T., Hsu, B.T. and Rees, D.C. 1991. Three-dimensional structures of acidic and basic fibroblast growth factors. Science 251: 90–93.
Eriksson, A.E., Cousens, L.S., Weaver, L.H. and Matthews, B.W. 1991. Three-dimensional structure of human basic fibroblast growth factor. Proc. Natl. Acad. Sci. USA 88: 3441–3445.
Zhang, J., Cousens, L.S., Barr, P.J. and Sprang, S.R. 1991. Three-dimensional structure of human basic fibroblast growth factor, a structural homolog of interleukin 1β. Proc. Natl. Acad. Sci. USA 88: 3446–3450.
Yoshida, T., Miyagawa, K., Odagiri, H., Sakamoto, H., Little, P.F.R., Terada, M. and Sugimura, T. 1987. Genomic sequence of hst, a transforming gene encoding a protein homologous to fibroblast growth factors and the int-2-encoded protein. Proc. Natl. Acad. Sci. USA 84: 7305–7309.
Arakawa, T., Hsu, Y.-R., Schiffer, S.G., Tsai, L.B., Curless, C. and Fox, G.M. 1989. Characterization of a cysteine-free analog of recombinant human basic fibroblast growth factor. Biochem. Biophys. Res. Commun. 161: 335–341.
Babbitt, P.C., West, B.L., Buechter, D.D., Kuntz, I.D. and Kenyon, G.L. 1990. Removal of a proteolytic activity associated with aggregates formed from expression of creatine kinase in Escherichia coli leads to improved recovery of active enzyme. Bio/Technology 8: 945–949.
Zettlmeissl, G., Rudolph, R. and Jaenicke, R. 1979. Reconstitution of lactic dehydrogenase. Noncovalent aggregation vs. reactivation. 1. Physical properties and kinetics of aggregation. Biochemistry 18: 5567–5575.
Rothman, J.E. 1989. Polypeptide chain binding proteins: catalysts of protein folding and related processes in cells. Cell 59: 591–601.
Laemmli, U.K. 1989. Cleavage of structral proteins during the assembly of the head of bacteriophage T4. Nature 227: 680–685.
Hochstrasser, D.F., Harrington, M.G., Hochstrasser, A.-C., Miller, M.J. and Merril, C.R. 1988. Methods for increasing the resolution of two-dimensional protein electrophoresis. Anal. Biochem. 173: 424–435.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Rinas, U., Tsai, L., Lyons, D. et al. Cysteine to Serine Substitutions in Basic Fibroblast Growth Factor: Effect on Inclusion Body Formation and Proteolytic Susceptibility During in Vitro Refolding. Nat Biotechnol 10, 435–440 (1992). https://doi.org/10.1038/nbt0492-435
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nbt0492-435
This article is cited by
-
Recombinant protein production associated growth inhibition results mainly from transcription and not from translation
Microbial Cell Factories (2020)
-
Post-production protein stability: trouble beyond the cell factory
Microbial Cell Factories (2011)
-
High level soluble expression, one-step purification and characterization of HIV-1 p24 protein
Virology Journal (2011)
-
High-level expression of the xylanase from Thermomyces lanuginosus in Escherichia coli
World Journal of Microbiology and Biotechnology (2008)
-
Aggregation as bacterial inclusion bodies does not imply inactivation of enzymes and fluorescent proteins
Microbial Cell Factories (2005)