Abstract
The development of immunologic interventions that can target the viral reservoir in HIV-1-infected individuals is a major goal of HIV-1 research1,2. However, little evidence exists that the viral reservoir can be sufficiently targeted to improve virologic control following discontinuation of antiretroviral therapy. Here we show that therapeutic vaccination with Ad26/MVA (recombinant adenovirus serotype 26 (Ad26) prime, modified vaccinia Ankara (MVA) boost)3,4 and stimulation of TLR7 (Toll-like receptor 7) improves virologic control and delays viral rebound following discontinuation of antiretroviral therapy in SIV-infected rhesus monkeys that began antiretroviral therapy during acute infection. Therapeutic vaccination with Ad26/MVA resulted in a marked increase in the magnitude and breadth of SIV-specific cellular immune responses in virologically suppressed, SIV-infected monkeys. TLR7 agonist administration led to innate immune stimulation and cellular immune activation. The combination of Ad26/MVA vaccination and TLR7 stimulation resulted in decreased levels of viral DNA in lymph nodes and peripheral blood, and improved virologic control and delayed viral rebound following discontinuation of antiretroviral therapy. The breadth of cellular immune responses correlated inversely with set point viral loads and correlated directly with time to viral rebound. These data demonstrate the potential of therapeutic vaccination combined with innate immune stimulation as a strategy aimed at a functional cure for HIV-1 infection.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Barouch, D. H. & Deeks, S. G. Immunologic strategies for HIV-1 remission and eradication. Science 345, 169–174 (2014)
Deeks, S. G. et al. International AIDS Society global scientific strategy: towards an HIV cure 2016. Nat. Med. 22, 839–850 (2016)
Barouch, D. H. et al. Protective efficacy of a global HIV-1 mosaic vaccine against heterologous SHIV challenges in rhesus monkeys. Cell 155, 531–539 (2013)
Barouch, D. H. et al. Vaccine protection against acquisition of neutralization-resistant SIV challenges in rhesus monkeys. Nature 482, 89–93 (2012)
Finzi, D. et al. Identification of a reservoir for HIV-1 in patients on highly active antiretroviral therapy. Science 278, 1295–1300 (1997)
Persaud, D., Zhou, Y., Siliciano, J. M. & Siliciano, R. F. Latency in human immunodeficiency virus type 1 infection: no easy answers. J. Virol. 77, 1659–1665 (2003)
Chun, T. W. et al. Presence of an inducible HIV-1 latent reservoir during highly active antiretroviral therapy. Proc. Natl Acad. Sci. USA 94, 13193–13197 (1997)
Ho, Y. C. et al. Replication-competent noninduced proviruses in the latent reservoir increase barrier to HIV-1 cure. Cell 155, 540–551 (2013)
Finzi, D. et al. Latent infection of CD4+ T cells provides a mechanism for lifelong persistence of HIV-1, even in patients on effective combination therapy. Nat. Med. 5, 512–517 (1999)
Chun, T. W., Davey, R. T., Jr, Engel, D., Lane, H. C. & Fauci, A. S. Re-emergence of HIV after stopping therapy. Nature 401, 874–875 (1999)
Shan, L. et al. Stimulation of HIV-1-specific cytolytic T lymphocytes facilitates elimination of latent viral reservoir after virus reactivation. Immunity 36, 491–501 (2012)
Deng, K. et al. Broad CTL response is required to clear latent HIV-1 due to dominance of escape mutations. Nature 517, 381–385 (2015)
Liu, J. et al. Immune control of an SIV challenge by a T-cell-based vaccine in rhesus monkeys. Nature 457, 87–91 (2009)
Whitney, J. B. et al. Rapid seeding of the viral reservoir prior to SIV viraemia in rhesus monkeys. Nature 512, 74–77 (2014)
Abbink, P. et al. Comparative seroprevalence and immunogenicity of six rare serotype recombinant adenovirus vaccine vectors from subgroups B and D. J. Virol. 81, 4654–4663 (2007)
Kawai, T. et al. Interferon-α induction through Toll-like receptors involves a direct interaction of IRF7 with MyD88 and TRAF6. Nat. Immunol. 5, 1061–1068 (2004)
Hemmi, H. et al. Small anti-viral compounds activate immune cells via the TLR7 MyD88-dependent signaling pathway. Nat. Immunol. 3, 196–200 (2002)
Henrich, T. J. et al. Antiretroviral-free HIV-1 remission and viral rebound after allogeneic stem cell transplantation: report of 2 cases. Ann. Intern. Med. 161, 319–327 (2014)
Persaud, D. et al. Absence of detectable HIV-1 viremia after treatment cessation in an infant. N. Engl. J. Med. 369, 1828–1835 (2013)
Banga, R. et al. PD-1+ and follicular helper T cells are responsible for persistent HIV-1 transcription in treated aviremic individuals. Nat. Med. 22, 754–761 (2016)
Hel, Z. et al. Viremia control following antiretroviral treatment and therapeutic immunization during primary SIV251 infection of macaques. Nat. Med. 6, 1140–1146 (2000)
Rosenberg, E. S. et al. Safety and immunogenicity of therapeutic DNA vaccination in individuals treated with antiretroviral therapy during acute/early HIV-1 infection. PLoS One 5, e10555 (2010)
Li, J. Z. et al. Factors associated with viral rebound in HIV-1-infected individuals enrolled in a therapeutic HIV-1 gag vaccine trial. J. Infect. Dis. 203, 976–983 (2011)
Liu, J. et al. Low-dose mucosal simian immunodeficiency virus infection restricts early replication kinetics and transmitted virus variants in rhesus monkeys. J. Virol. 84, 10406–10412 (2010)
Burg, D., Rong, L., Neumann, A. U. & Dahari, H. Mathematical modeling of viral kinetics under immune control during primary HIV-1 infection. J. Theor. Biol. 259, 751–759 (2009)
Wodarz, D. et al. A new theory of cytotoxic T-lymphocyte memory: implications for HIV treatment. Philos. Trans. R. Soc. B Biol. Sci. 355, 329–343 (2000)
Luzyanina, T., Engelborghs, K., Ehl, S., Klenerman, P. & Bocharov, G. Low level viral persistence after infection with LCMV: a quantitative insight through numerical bifurcation analysis. Math. Biosci. 173, 1–23 (2001)
Althaus, C. L. & De Boer, R. J. Dynamics of immune escape during HIV/SIV infection. PLOS Comput. Biol. 4, e1000103 (2008)
Elemans, M. et al. Why don’t CD8+ T cells reduce the lifespan of SIV-infected cells in vivo? PLOS Comput. Biol. 7, e1002200 (2011)
Gadhamsetty, S., Coorens, T. & Boer, R. J. de. Notwithstanding circumstantial alibis, cytotoxic T cells can be major killers of HIV-1 infected cells. J. Virol. 306–316 (2016)
Chen, H. Y., Di Mascio, M., Perelson, A. S., Ho, D. D. & Zhang, L. Determination of virus burst size in vivo using a single-cycle SIV in rhesus macaques. Proc. Natl Acad. Sci. USA 104, 19079–19084 (2007)
De Boer, R. J., Ribeiro, R. M. & Perelson, A. S. Current estimates for HIV-1 production imply rapid viral clearance in lymphoid tissues. PLOS Comput. Biol. 6, e1000906 (2010)
Ramratnam, B. et al. Rapid production and clearance of HIV-1 and hepatitis C virus assessed by large volume plasma apheresis. Lancet 354, 1782–1785 (1999)
Chivers, C. MHadaptive: General Markov Chain Monte Carlo for Bayesian Inference using adaptive Metropolis-Hastings sampling. https://cran.r-project.org/web/packages/MHadaptive/MHadaptive.pdf (2012)
Soetaert, K., Petzoldt, T. & Setzer, R. W. deSolve: Solvers for Initial Value Problems of Differential Equations (ODE, DAE, DDE). https://cran.r-project.org/web/packages/deSolve/vignettes/deSolve.pdf (2016)
Acknowledgements
We thank C. Linde, T. Broge, T. Barnes, D. van Manen, F. Wegmann, C. Shaver, W. Wagner, M. Boyd, R. Nityanandam, K. Smith, S. Blackmore, L. Parenteau, P. Giglio, M. Shetty, S. Levin, J. Shields, G. Neubauer, and F. Stephens for generous advice, assistance, and reagents. We acknowledge support from the US Army Medical Research and Materiel Command and the Military HIV Research Program, Walter Reed Army Institute of Research through its cooperative agreement with the Henry M. Jackson Foundation (W81XWH-11-2-0174); the National Institutes of Health (AI096040, AI124377, AI126603, OD019851); the Ragon Institute of MGH, MIT, and Harvard. Mathematical model fitting was performed on the Orchestra High Performance Compute Cluster at Harvard Medical School. The views expressed in this manuscript are those of the authors and do not represent the official views of the Department of the Army or the Department of Defense.
Author information
Authors and Affiliations
Contributions
D.H.B, N.L.M., J.H.K., M.L.R., M.G.P., H.S., and R.G. designed the studies. J.H. and R.G. developed the ART formulation and TLR7 agonist. E.N.B., C.C., K.E.S., J.L., J.P.N., A.L.B., L.P., B.C.L., J.J., D.J., J.M., S.M., A.C., K.M., and G.A. performed the immunologic assays. P.A. and D.N. conducted the virologic assays. J.M.G. and A.L.H. performed the viral dynamics modelling. M.G.L. led the clinical care of the rhesus monkeys. D.H.B. wrote the paper with all co-authors.
Corresponding author
Ethics declarations
Competing interests
M.G.P. and H.S. are employees of Janssen Infectious Diseases and Vaccines. J.H. and R.G. are employees of Gilead Sciences.
Additional information
Reviewer Information
Nature thanks S. Lewin and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Extended data figures and tables
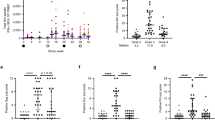
Extended Data Figure 1 Activation of CD8+ T cells following GS-986 administration.
Representative data on days 0, 1, and 2 following GS-986 administration. Activation was assessed by CD69 expression on CD3+CD8+ T cells. Red bars represent mean values for each group.
Extended Data Figure 2 Activation of CD4+ T cells following GS-986 administration.
Representative data on days 0, 1, and 2 following GS-986 administration. Activation was assessed by CD69 expression on CD3+CD4+ T cells.
Extended Data Figure 3 Innate immune stimulation following GS-986 administration.
Plasma IFN-α (pg ml−1) levels are shown on day 1 following GS-986 administration. Red bars represent mean values for each group. Data points reflect all animals following all GS-986 administrations combined with pre-dose levels subtracted.
Extended Data Figure 4 CD8+ T cells following Ad26/MVA vaccination.
SIVmac239 Gag-, Pol- and Env-specific, IFN-γ+CD3+CD8+ central memory T cells were assessed by multiparameter intracellular cytokine staining assays.
Extended Data Figure 5 CD4+ T cells following Ad26/MVA vaccination.
SIVmac239 Gag-, Pol- and Env-specific, IFN-γ+CD3+CD4+ central memory T cells were assessed by multiparameter intracellular cytokine staining assays.
Extended Data Figure 6 Cellular immune breadth.
Responses to subpools of 10 peptides spanning SIVmac239 Gag, Pol, and Env are shown before vaccination (week 20, blue), after Ad26 prime (week 29, red), and after MVA boost (week 50, green). Coloured squares indicate positive responses. ‘x’ indicates missing data as a result of insufficient PBMCs.
Extended Data Figure 7 Correlations of cellular immune breadth to day 7 SIV RNA.
Correlations are shown for the breadth of Gag, Pol, Env and total (Gag+Pol+Env) cellular immune responses at the time of ART discontinuation at week 72 and pre-ART day 7 log SIV RNA. P value indicates two-sided Spearman rank-correlation test. R value indicates correlation coefficient.
Extended Data Figure 8 Correlations of cellular immune breadth to set point viral loads.
Correlations are shown for the breadth of Gag, Pol, Env and total (Gag+Pol+Env) cellular immune responses at peak immunity at week 50 and set point log SIV RNA following ART discontinuation. P values indicate two-sided Spearman rank-correlation tests. R values indicate correlation coefficients.
Extended Data Figure 9 Correlations of cellular immune breadth to time to viral rebound.
Correlations are shown for the breadth of Gag, Pol, Env and total (Gag+Pol+Env) cellular immune responses at peak immunity at week 50 and time to viral rebound following ART discontinuation. P values indicate two-sided Spearman rank-correlation tests. R values indicate correlation coefficients.
Extended Data Figure 10 Rebound kinetic parameters estimated from viral dynamics modelling.
Viral load values following ART discontinuation in each animal were fit to a viral dynamics model using a Bayesian framework. a–d, Plots show the median and 95% credible intervals for estimations of the rate of reactivation of cells from the latent reservoir (a), the initial exponential growth rate (b), the immune proliferation rate (c), and the time at which viral load reaches the detection threshold of 200 copies per ml for each treatment group (d). Monkeys treated with both the vaccine and TLR7 agonist exhibited slower viral growth rates and stronger immune responses than all other groups (P < 0.01 for each comparison). These monkeys and monkeys treated with only the vaccine exhibited a lower reservoir exit rate than untreated monkeys (P < 0.05 for each comparison).
Rights and permissions
About this article
Cite this article
Borducchi, E., Cabral, C., Stephenson, K. et al. Ad26/MVA therapeutic vaccination with TLR7 stimulation in SIV-infected rhesus monkeys. Nature 540, 284–287 (2016). https://doi.org/10.1038/nature20583
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature20583
This article is cited by
-
Immune targeting of HIV-1 reservoir cells: a path to elimination strategies and cure
Nature Reviews Microbiology (2024)
-
More than the Infinite Monkey Theorem: NHP Models in the Development of a Pediatric HIV Cure
Current HIV/AIDS Reports (2024)
-
Differential immunophenotype of circulating monocytes from pregnant women in response to viral ligands
BMC Pregnancy and Childbirth (2023)
-
Prevention, treatment and cure of HIV infection
Nature Reviews Microbiology (2023)
-
Immunogenic arenavirus vector SIV vaccine reduces setpoint viral load in SIV-challenged rhesus monkeys
npj Vaccines (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.