Abstract
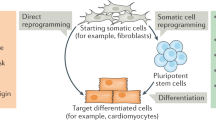
The ability to produce stem cells by induced pluripotency (iPS reprogramming) has rekindled an interest in earlier studies showing that transcription factors can directly convert specialized cells from one lineage to another. Lineage reprogramming has become a powerful tool to study cell fate choice during differentiation, akin to inducing mutations for the discovery of gene functions. The lessons learnt provide a rubric for how cells may be manipulated for therapeutic purposes.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
References
Blau, H. M. How fixed is the differentiated state? Lessons from heterokaryons. Trends Genet. 5, 268–272 (1989)
Davis, R. L., Weintraub, H. & Lassar, A. B. Expression of a single transfected cDNA converts fibroblasts to myoblasts. Cell 51, 987–1000 (1987)
Kulessa, H., Frampton, J. & Graf, T. GATA-1 reprograms avian myelomonocytic cell lines into eosinophils, thromboblasts, and erythroblasts. Genes Dev. 9, 1250–1262 (1995)This paper, together with refs 19 and 20, established the principle of transcription factor cross-antagonisms.
Gurdon, J. B. & Byrne, J. A. The first half-century of nuclear transplantation. Proc. Natl Acad. Sci. USA 100, 8048–8052 (2003)
Wilmut, I., Schnieke, A. E., McWhir, J., Kind, A. J. & Campbell, K. H. Viable offspring derived from fetal and adult mammalian cells. Nature 385, 810–813 (1997)
Gurdon, J. B. & Melton, D. A. Nuclear reprogramming in cells. Science 322, 1811–1815 (2008)
Hochedlinger, K. & Jaenisch, R. Monoclonal mice generated by nuclear transfer from mature B and T donor cells. Nature 415, 1035–1038 (2002)
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006)
Slack, J. M. Metaplasia and transdifferentiation: from pure biology to the clinic. Nature Rev. Mol. Cell Biol. 8, 369–378 (2007)
Yang, J. & Weinberg, R. A. Epithelial-mesenchymal transition: at the crossroads of development and tumor metastasis. Dev. Cell 14, 818–829 (2008)
Kragl, M. et al. Cells keep a memory of their tissue origin during axolotl limb regeneration. Nature 460, 60–65 (2009)
Chen, M. J., Yokomizo, T., Zeigler, B. M., Dzierzak, E. & Speck, N. A. Runx1 is required for the endothelial to haematopoietic cell transition but not thereafter. Nature 457, 887–891 (2009)
Lancrin, C. et al. The haemangioblast generates haematopoietic cells through a haemogenic endothelium stage. Nature 457, 892–895 (2009)
Dzierzak, E. & Speck, N. A. Of lineage and legacy: the development of mammalian hematopoietic stem cells. Nature Immunol. 9, 129–136 (2008)
Eilken, H. M., Nishikawa, S. & Schroeder, T. Continuous single-cell imaging of blood generation from haemogenic endothelium. Nature 457, 896–900 (2009)An example of ‘transdifferentiation’ in the context of normal lineage progression; also highlights how real-time visualization may show cell fate conversions that are otherwise hard to document.
Adamo, L. et al. Biomechanical forces promote embryonic haematopoiesis. Nature 459, 1131–1135 (2009)
North, T. E. et al. Hematopoietic stem cell development is dependent on blood flow. Cell 137, 736–748 (2009)
Zhou, Q. & Melton, D. A. Extreme makeover: converting one cell into another. Cell Stem Cell 3, 382–388 (2008)
Visvader, J. E., Elefanty, A. G., Strasser, A. & Adams, J. M. GATA-1 but not SCL induces megakaryocytic differentiation in an early myeloid line. EMBO J. 11, 4557–4564 (1992)
Nerlov, C. & Graf, T. PU.1 induces myeloid lineage commitment in multipotent hematopoietic progenitors. Genes Dev. 12, 2403–2412 (1998)
Heyworth, C., Pearson, S., May, G. & Enver, T. Transcription factor-mediated lineage switching reveals plasticity in primary committed progenitor cells. EMBO J. 21, 3770–3781 (2002)
Zhang, P. et al. Enhancement of hematopoietic stem cell repopulating capacity and self-renewal in the absence of the transcription factor C/EBPα. Immunity 21, 853–863 (2004)
Xie, H., Ye, M., Feng, R. & Graf, T. Stepwise reprogramming of B cells into macrophages. Cell 117, 663–676 (2004)
Laiosa, C. V., Stadtfeld, M., Xie, H., de Andres-Aguayo, L. & Graf, T. Reprogramming of committed T cell progenitors to macrophages and dendritic cells by C/EBPα and PU.1 transcription factors. Immunity 25, 731–744 (2006)
Arinobu, Y. et al. Reciprocal activation of GATA-1 and PU.1 marks initial specification of hematopoietic stem cells into myeloerythroid and myelolymphoid lineages. Cell Stem Cell 1, 416–427 (2007)
Iwasaki, H. & Akashi, K. Myeloid lineage commitment from the hematopoietic stem cell. Immunity 26, 726–740 (2007)
Okuno, Y. et al. Potential autoregulation of transcription factor PU.1 by an upstream regulatory element. Mol. Cell. Biol. 25, 2832–2845 (2005)
Yu, C. et al. Targeted deletion of a high-affinity GATA-binding site in the GATA-1 promoter leads to selective loss of the eosinophil lineage in vivo . J. Exp. Med. 195, 1387–1395 (2002)
Ptashne, M. A Genetic Switch. Phage Lambda Revisited 3rd edn (Cold Spring Harbor Laboratory Press, 2004)
Cantor, A. B. & Orkin, S. H. Hematopoietic development: a balancing act. Curr. Opin. Genet. Dev. 11, 513–519 (2001)
Graf, T. Differentiation plasticity of hematopoietic cells. Blood 99, 3089–3101 (2002)
Orkin, S. H. & Zon, L. I. Hematopoiesis: an evolving paradigm for stem cell biology. Cell 132, 631–644 (2008)
Zhang, P. et al. Negative cross-talk between hematopoietic regulators: GATA proteins repress PU.1. Proc. Natl Acad. Sci. USA 96, 8705–8710 (1999)
Stopka, T., Amanatullah, D. F., Papetti, M. & Skoultchi, A. I. PU.1 inhibits the erythroid program by binding to GATA-1 on DNA and creating a repressive chromatin structure. EMBO J. 24, 3712–3723 (2005)
Rhodes, J. et al. Interplay of Pu.1 and Gata1 determines myelo-erythroid progenitor cell fate in zebrafish. Dev. Cell 8, 97–108 (2005) In vivo evidence for the importance of GATA1:PU.1 interplay in lineage specification.
Galloway, J. L., Wingert, R. A., Thisse, C., Thisse, B. & Zon, L. I. Loss of Gata1 but not Gata2 converts erythropoiesis to myelopoiesis in zebrafish embryos. Dev. Cell 8, 109–116 (2005)
Warga, R. M., Kane, D. A. & Ho, R. K. Fate mapping embryonic blood in zebrafish: multi- and unipotential lineages are segregated at gastrulation. Dev. Cell 16, 744–755 (2009)
Nutt, S. L., Heavey, B., Rolink, A. G. & Busslinger, M. Commitment to the B-lymphoid lineage depends on the transcription factor Pax5. Nature 401, 556–562 (1999)
Cobaleda, C., Jochum, W. & Busslinger, M. Conversion of mature B cells into T cells by dedifferentiation to uncommitted progenitors. Nature 449, 473–477 (2007)
Rothenberg, E. V. Cell lineage regulators in B and T cell development. Nature Immunol. 8, 441–444 (2007)
Davidson, E. H. & Levine, M. S. Properties of developmental gene regulatory networks. Proc. Natl Acad. Sci. USA 105, 20063–20066 (2008)
Zhou, L. et al. TGF-β-induced Foxp3 inhibits TH17 cell differentiation by antagonizing RORγt function. Nature 453, 236–240 (2008)
Rieger, M. A., Hoppe, P. S., Smejkal, B. M., Eitelhuber, A. C. & Schroeder, T. Hematopoietic cytokines can instruct lineage choice. Science 325, 217–218 (2009)
Sarrazin, S. et al. MafB restricts M-CSF-dependent myeloid commitment divisions of hematopoietic stem cells. Cell 138, 300–313 (2009)An example of how extrinsic signals may act through intrinsic regulators to specify lineage fates; ref. 57 addresses a similar issue from a mathematical modelling perspective.
Smith, J., Wardle, F., Loose, M., Stanley, E. & Patient, R. Germ layer induction in ESC–following the vertebrate roadmap. Curr. Protocols Stem Cell Biol. 1, 1D.1.1–1D.1.22 (2007)
Iwasaki, H. et al. The order of expression of transcription factors directs hierarchical specification of hematopoietic lineages. Genes Dev. 20, 3010–3021 (2006)Showed that the order of transcription factor expression can induce different cell fates.
Sieweke, M. H. & Graf, T. A transcription factor party during blood cell differentiation. Curr. Opin. Genet. Dev. 8, 545–551 (1998)
Waddington, C. H. The Strategy of the Genes (Allen & Unwin, 1957)
Kauffman, S. Metabolic stability and epigenesis in randomly constructed genetic nets. J. Theor. Biol. 22, 437–467 (1969)
Kauffman, S. Origins of Order: Self-organization and Selection in Evolution (Oxford Univ. Press, 1993)
Enver, T., Pera, M., Peterson, C. & Andrews, P. W. Stem cell states, fates, and the rules of attraction. Cell Stem Cell 4, 387–397 (2009)
Hu, M. et al. Multilineage gene expression precedes commitment in the hemopoietic system. Genes Dev. 11, 774–785 (1997)
Miyamoto, T. et al. Myeloid or lymphoid promiscuity as a critical step in hematopoietic lineage commitment. Dev. Cell 3, 137–147 (2002)
Månsson, R. et al. Molecular evidence for hierarchical transcriptional lineage priming in fetal and adult stem cells and multipotent progenitors. Immunity 26, 407–419 (2007)
Enver, T., Heyworth, C. M. & Dexter, T. M. Do stem cells play dice? Blood 92, 348–351,–352 (1998)
Graf, T. & Stadtfeld, M. Heterogeneity of embryonic and adult stem cells. Cell Stem Cell 3, 480–483 (2008)
Chambers, I. et al. Nanog safeguards pluripotency and mediates germline development. Nature 450, 1230–1234 (2007)
Chickarmane, V., Enver, T. & Peterson, C. Computational modeling of the hematopoietic erythroid-myeloid switch reveals insights into cooperativity, priming, and irreversibility. PLoS Comput. Biol. 5, e1000268 (2009)
Huang, S., Guo, Y. P., May, G. & Enver, T. Bifurcation dynamics in lineage-commitment in bipotent progenitor cells. Dev. Biol. 305, 695–713 (2007)Refs 57, 58 and 59 highlight how mathematical modelling of cross-antagonistic circuits illuminates their dynamic behaviour and capacity to effect stable lineage choice decisions.
Roeder, I. & Glauche, I. Towards an understanding of lineage specification in hematopoietic stem cells: a mathematical model for the interaction of transcription factors GATA-1 and PU.1. J. Theor. Biol. 241, 852–865 (2006)
Swiers, G., Patient, R. & Loose, M. Genetic regulatory networks programming hematopoietic stem cells and erythroid lineage specification. Dev. Biol. 294, 525–540 (2006)
Laslo, P. et al. Multilineage transcriptional priming and determination of alternate hematopoietic cell fates. Cell 126, 755–766 (2006)An example of sequential cross-antagonistic switches in the specification of cell lineage.
Frontelo, P. et al. Novel role for EKLF in megakaryocyte lineage commitment. Blood 110, 3871–3880 (2007)
Hwang, E. S., Szabo, S. J., Schwartzberg, P. L. & Glimcher, L. H. T helper cell fate specified by kinase-mediated interaction of T-bet with GATA-3. Science 307, 430–433 (2005)
Yechoor, V. et al. Neurogenin3 is sufficient for transdetermination of hepatic progenitor cells into neo-islets in vivo but not transdifferentiation of hepatocytes. Dev. Cell 16, 358–373 (2009)
Zhou, Q., Brown, J., Kanarek, A., Rajagopal, J. & Melton, D. A. In vivo reprogramming of adult pancreatic exocrine cells to β-cells. Nature 455, 627–632 (2008)Showed that expression in the pancreas of a combination of three key regulators re-specifies one somatic cell type into another functional cell type, in vivo.
Starck, J. et al. Functional cross-antagonism between transcription factors FLI-1 and EKLF. Mol. Cell. Biol. 23, 1390–1402 (2003)
Querfurth, E. et al. Antagonism between C/EBPβ and FOG in eosinophil lineage commitment of multipotent hematopoietic progenitors. Genes Dev. 14, 2515–2525 (2000)
Kajimura, S. et al. Regulation of the brown and white fat gene programs through a PRDM16/CtBP transcriptional complex. Genes Dev. 22, 1397–1409 (2008)
Heins, N. et al. Glial cells generate neurons: the role of the transcription factor Pax6. Nature Neurosci. 5, 308–315 (2002)
Jessberger, S., Toni, N., Clemenson, G. D., Ray, J. & Gage, F. H. Directed differentiation of hippocampal stem/progenitor cells in the adult brain. Nature Neurosci. 11, 888–893 (2008)
Gubbels, S. P., Woessner, D. W., Mitchell, J. C., Ricci, A. J. & Brigande, J. V. Functional auditory hair cells produced in the mammalian cochlea by in utero gene transfer. Nature 455, 537–541 (2008)
Horb, M. E., Shen, C. N., Tosh, D. & Slack, J. M. Experimental conversion of liver to pancreas. Curr. Biol. 13, 105–115 (2003)
Niwa, H. et al. Interaction between Oct3/4 and Cdx2 determines trophectoderm differentiation. Cell 123, 917–929 (2005)
Ralston, A. & Rossant, J. Genetic regulation of stem cell origins in the mouse embryo. Clin. Genet. 68, 106–112 (2005)
Aoi, T. et al. Generation of pluripotent stem cells from adult mouse liver and stomach cells. Science 321, 699–702 (2008)
Stadtfeld, M., Brennand, K. & Hochedlinger, K. Reprogramming of pancreatic β cells into induced pluripotent stem cells. Curr. Biol. 18, 890–894 (2008)
Hochedlinger, K. & Plath, K. Epigenetic reprogramming and induced pluripotency. Development 136, 509–523 (2009)
Yamanaka, S. Elite and stochastic models for induced pluripotent stem cell generation. Nature 460, 49–52 (2009)
Kim, J. B. et al. Oct4-induced pluripotency in adult neural stem cells. Cell 136, 411–419 (2009)
Loh, Y. H., Zhang, W., Chen, X., George, J. & Ng, H. H. Jmjd1a and Jmjd2c histone H3 Lys 9 demethylases regulate self-renewal in embryonic stem cells. Genes Dev. 21, 2545–2557 (2007)
Bernstein, B. E. et al. A bivalent chromatin structure marks key developmental genes in embryonic stem cells. Cell 125, 315–326 (2006)
Ying, Q. L. et al. The ground state of embryonic stem cell self-renewal. Nature 453, 519–523 (2008)
Alon, U. An Introduction to Systems Biology. Design Principles of Biological Circuits (Chapman and Hall/CRC, 2006)
Chickarmane, V., Troein, C., Nuber, U. A., Sauro, H. M. & Peterson, C. Transcriptional dynamics of the embryonic stem cell switch. PLoS Comput. Biol. 2, e123 (2006)
Chickarmane, V. & Peterson, C. A computational model for understanding stem cell, trophectoderm and endoderm lineage determination. PLoS One 3, e3478 (2008)
Chang, H. H., Hemberg, M., Barahona, M., Ingber, D. E. & Huang, S. Transcriptome-wide noise controls lineage choice in mammalian progenitor cells. Nature 453, 544–547 (2008)
Boukamp, P., Chen, J., Gonzales, F., Jones, P. A. & Fusenig, N. E. Progressive stages of “transdifferentiation” from epidermal to mesenchymal phenotype induced by MyoD1 transfection, 5-aza-2′-deoxycytidine treatment, and selection for reduced cell attachment in the human keratinocyte line HaCaT. J. Cell Biol. 116, 1257–1271 (1992)
Feng, R. et al. PU.1 and C/EBPα/β convert fibroblasts into macrophage-like cells. Proc. Natl Acad. Sci. USA 105, 6057–6062 (2008)
Palermo, A. et al. Nuclear reprogramming in heterokaryons is rapid, extensive, and bidirectional. FASEB J. 23, 1431–1440 (2009)
Singh, H., Medina, K. L. & Pongubala, J. M. Contingent gene regulatory networks and B cell fate specification. Proc. Natl Acad. Sci. USA 102, 4949–4953 (2005)
Kitajima, K., Zheng, J., Yen, H., Sugiyama, D. & Nakano, T. Multipotential differentiation ability of GATA-1-null erythroid-committed cells. Genes Dev. 20, 654–659 (2006)
Judson, R. L., Babiarz, J. E., Venere, M. & Blelloch, R. Embryonic stem cell-specific microRNAs promote induced pluripotency. Nature Biotechnol. 27, 459–461 (2009)
Takeuchi, J. K. & Bruneau, B. G. Directed transdifferentiation of mouse mesoderm to heart tissue by defined factors. Nature 459, 708–711 (2009)
Viswanathan, S. R., Daley, G. Q. & Gregory, R. I. Selective blockade of microRNA processing by Lin28. Science 320, 97–100 (2008)
Feng, B., Ng, J. H., Heng, J. C. & Ng, H. H. Molecules that promote or enhance reprogramming of somatic cells to induced pluripotent stem cells. Cell Stem Cell 4, 301–312 (2009)
Collombat, P. et al. Opposing actions of Arx4 and Pax4 in endocrine pancreas development. Genes Dev. 15, 2591–2603 (2003)
Lagha, M. et al. Pax3/7:Foxc2 reciprocal repression in the somite modulates multipotent cell fates. Dev. Cell (in the press)
Acknowledgements
We would like to thank J. Sharpe, C. Peterson, J. Brickman and D. Thieffry for feedback and suggestions. T.G. is an ICREA professor and T.E. is supported by an LRF specialist programme.
Author Contributions T.G. and T.E. together conceived the ideas encapsulated in the article and also drafted it jointly. Most of the figures were conceived by T.G. and modified by T.E.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Graf, T., Enver, T. Forcing cells to change lineages . Nature 462, 587–594 (2009). https://doi.org/10.1038/nature08533
Issue Date:
DOI: https://doi.org/10.1038/nature08533
This article is cited by
-
Logical modeling of thymus and natural killer lymphocyte differentiation
Journal of Biological Physics (2021)
-
Small-molecule-mediated reprogramming: a silver lining for regenerative medicine
Experimental & Molecular Medicine (2020)
-
Microfibril-associated glycoprotein 4 (Mfap4) regulates haematopoiesis in zebrafish
Scientific Reports (2020)
-
Do cells use passwords in cell-state transitions? Is cell signaling sometimes encrypted?
Theory in Biosciences (2020)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.