Abstract
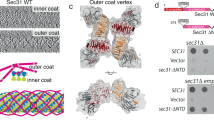
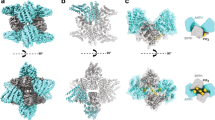
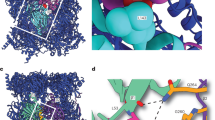
Endomembranes of eukaryotic cells are dynamic structures that are in continuous communication through the activity of specialized cellular machineries1, such as the coat protein complex II (COPII), which mediates cargo export from the endoplasmic reticulum (ER)2,3. COPII consists of the Sar1 GTPase, Sec23 and Sec24 (Sec23/24), where Sec23 is a Sar1-specific GTPase-activating protein and Sec24 functions in cargo selection, and Sec13 and Sec31 (Sec13/31), which has a structural role3. Whereas recent results have shown that Sec23/24 and Sec13/31 can self-assemble to form COPII cage-like particles4, we now show that Sec13/31 can self-assemble to form minimal cages in the absence of Sec23/24. We present a three-dimensional reconstruction of these Sec13/31 cages at 30 Å resolution using cryo-electron microscopy and single particle analysis. These results reveal a novel cuboctahedron geometry with the potential to form a flexible lattice and to generate a diverse range of containers. Our data are consistent with a model for COPII coat complex assembly in which Sec23/24 has a non-structural role as a multivalent ligand localizing the self-assembly of Sec13/31 to form a cage lattice driving ER cargo export.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Gurkan, C. et al. Large-scale profiling of Rab GTPase trafficking networks: The membrome. Mol. Biol. Cell 16, 3847–3864 (2005)
Bonifacino, J. S. & Glick, B. S. The mechanisms of vesicle budding and fusion. Cell 116, 153–166 (2004)
Lee, M. C., Miller, E. A., Goldberg, J., Orci, L. & Schekman, R. Bi-directional protein transport between the ER and Golgi. Annu. Rev. Cell Dev. Biol. 20, 87–123 (2004)
Antonny, B., Gounon, P., Schekman, R. & Orci, L. Self-assembly of minimal COPII cages. EMBO Rep. 4, 419–424 (2003)
Gurkan, C. & Balch, W. E. Recombinant production in baculovirus-infected insect cells and purification of the mammalian Sec13/Sec31 complex. Methods Enzymol. 404, 58–66 (2005)
Salama, N. R., Chuang, J. S. & Schekman, R. W. Sec31 encodes an essential component of the COPII coat required for transport vesicle budding from the endoplasmic reticulum. Mol. Biol. Cell 8, 205–217 (1997)
Lederkremer, G. Z. et al. Structure of the Sec23p/24p and Sec13p/31p complexes of COPII. Proc. Natl Acad. Sci. USA 98, 10704–10709 (2001)
Kim, J., Hamamoto, S., Ravazzola, M., Orci, L. & Schekman, R. Uncoupled packaging of amyloid precursor protein and presenilin 1 into COPII vesicles. J. Biol. Chem. 280, 7758–7768 (2005)
Shugrue, C. A. et al. Identification of the putative mammalian orthologue of Sec31P, a component of the COPII coat. J. Cell Sci. 112, 4547–4556 (1999)
Salama, N. R., Yeung, T. & Schekman, R. W. The Sec13p complex and reconstitution of vesicle budding from the ER with purified cytosolic proteins. EMBO J. 12, 4073–4082 (1993)
Tang, B. L. et al. Mammalian homologues of yeast sec31p. An ubiquitously expressed form is localized to endoplasmic reticulum (ER) exit sites and is essential for ER-Golgi transport. J. Biol. Chem. 275, 13597–13604 (2000)
Matsuoka, K. et al. COPII-coated vesicle formation reconstituted with purified coat proteins and chemically defined liposomes. Cell 93, 263–275 (1998)
Matsuoka, K., Schekman, R., Orci, L. & Heuser, J. E. Surface structure of the COPII-coated vesicle. Proc. Natl Acad. Sci. USA 98, 13705–13709 (2001)
Aridor, M., Bannykh, S. I., Rowe, T. & Balch, W. E. Cargo can modulate COPII vesicle formation from the endoplasmic reticulum. J. Biol. Chem. 274, 4389–4399 (1999)
Ludtke, S. J., Baldwin, P. R. & Chiu, W. EMAN: semiautomated software for high-resolution single-particle reconstructions. J. Struct. Biol. 128, 82–97 (1999)
Belden, W. J. & Barlowe, C. Purification of functional Sec13p-Sec31p complex, a subunit of COPII coat. Methods Enzymol. 329, 438–443 (2001)
Shimoni, Y. & Schekman, R. Vesicle budding from endoplasmic reticulum. Methods Enzymol. 351, 258–278 (2002)
Pryer, N. K., Salama, N. R., Schekman, R. & Kaiser, C. A. Cytosolic Sec13p complex is required for vesicle formation from the endoplasmic reticulum in vitro. J. Cell Biol. 120, 865–875 (1993)
Devos, D. et al. Components of coated vesicles and nuclear pore complexes share a common molecular architecture. PLoS Biol. 2, e380 (2004)
Saxena, K. et al. Analysis of the physical properties and molecular modeling of Sec13: A WD repeat protein involved in vesicular traffic. Biochemistry 35, 15215–15221 (1996)
Musacchio, A. et al. Functional organization of clathrin in coats: combining electron cryomicroscopy and X-ray crystallography. Mol. Cell 3, 761–770 (1999)
Fotin, A. et al. Molecular model for a complete clathrin lattice from electron cryomicroscopy. Nature 432, 573–579 (2004)
Shaywitz, D. A., Espenshade, P. J., Gimeno, R. E. & Kaiser, C. A. COPII subunit interactions in the assembly of the vesicle coat. J. Biol. Chem. 272, 25413–25416 (1997)
Smith, C. J., Grigorieff, N. & Pearse, B. M. Clathrin coats at 21 Å resolution: a cellular assembly designed to recycle multiple membrane receptors. EMBO J. 17, 4943–4953 (1998)
Jones, B. et al. Mutations in a Sar1 GTPase of COPII vesicles are associated with lipid absorption disorders. Nature Genet. 34, 29–31 (2003)
Mironov, A. A. et al. ER-to-Golgi carriers arise through direct en bloc protrusion and multistage maturation of specialized ER exit domains. Dev. Cell 5, 583–594 (2003)
Suloway, C. et al. Automated molecular microscopy: The new Leginon system. J. Struct. Biol. 151, 41–60 (2005)
Zhu, Y., Carragher, B. & Potter, C. S. Improving Template Matching Based Particle Selection for Cryo-electron Microscopy (IEEE ISBI04 Conference, Arlington, Virginia, 2004)
Mallick, S. P., Carragher, B., Potter, C. S. & Kriegman, D. J. ACE: Automated CTF Estimation. Ultramicroscopy 104, 8–29 (2005)
Acknowledgements
These studies are supported by NIH grants to W.E.B., C.S.P. and B.C. S.M.S. is a recipient of an NIH Postdoctoral Fellowship. C.G. is a recipient of a Cystic Fibrosis Foundation Postdoctoral Research Fellowship. We thank G. Cantin and J. Venable for LC-MS/MS analysis, and J. T. Weissman, K. Straley and S. J. Lloyd for setting up or helping with recombinant expression of the human Sec13/31 complex in baculovirus-infected insect cells. This is TSRI manuscript number 17546-CB.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Notes
This file contains Supplementary Methods and Supplementary Figure Legends. (DOC 44 kb)
Supplementary Figure 1
Further initial analyses of the self-assembled Sec13/31 cages. (PDF 537 kb)
Supplementary Figure 2
Single particle analysis of the Sec13/31 cages in vitreous ice. (PDF 1587 kb)
Supplementary Figure 3
Further potential geometries consistent with a Sec13/31 lattice. (PDF 467 kb)
Supplementary Video
The reconstructed cage structure has a diameter of 600 A ° along its longest diagonal, the length of an edge is 300 Å, and the width of an edge is 40 Å. (MOV 3354 kb)
Rights and permissions
About this article
Cite this article
Stagg, S., Gürkan, C., Fowler, D. et al. Structure of the Sec13/31 COPII coat cage. Nature 439, 234–238 (2006). https://doi.org/10.1038/nature04339
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1038/nature04339
This article is cited by
-
Repeat length of C9orf72-associated glycine–alanine polypeptides affects their toxicity
Acta Neuropathologica Communications (2023)
-
Vesicle trafficking and vesicle fusion: mechanisms, biological functions, and their implications for potential disease therapy
Molecular Biomedicine (2022)
-
Acetyl-CoA flux from the cytosol to the ER regulates engagement and quality of the secretory pathway
Scientific Reports (2021)
-
Structure of the complete, membrane-assembled COPII coat reveals a complex interaction network
Nature Communications (2021)
-
Interactive GPU active contours for segmenting inhomogeneous objects
Journal of Real-Time Image Processing (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.